Revolutionizing 2-Amino-5-Halopyridine Production: A Green, Scalable Route for Global API Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking more efficient, safer, and cost-effective pathways for critical heterocyclic intermediates. Patent CN110092746B introduces a groundbreaking methodology for the preparation of 2-amino-5-halogenated pyridines, a class of compounds essential for synthesizing active pharmaceutical ingredients (APIs) such as betrixaban and zopiclone. Unlike traditional multi-step syntheses that rely on expensive starting materials and hazardous reagents, this innovation utilizes 4-cyano-1-butyne and elemental halogens in a streamlined, acid-catalyzed process. The technical breakthrough lies in its ability to achieve high selectivity and yields under mild conditions, effectively bypassing the environmental and safety pitfalls associated with classical nitration and diazotization chemistries. For global supply chain leaders, this represents a pivotal shift towards greener manufacturing protocols that do not compromise on purity or scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-amino-5-halogenated pyridines has been plagued by significant technical and economic inefficiencies. Conventional routes typically commence with 2-aminopyridine, a relatively high-cost precursor, which undergoes a series of cumbersome transformations including nitration, acetylation, reduction, and diazotization. As illustrated in the legacy synthetic pathway, the nitration step using mixed acids generates substantial volumes of acidic wastewater, posing severe environmental disposal challenges. Furthermore, the selectivity of electrophilic substitution on the pyridine ring is often poor, leading to difficult-to-separate isomers like 2-amino-3-nitropyridine, which drastically reduces overall yield. The subsequent formation of diazonium salts introduces critical safety hazards, as these intermediates are thermally unstable and prone to explosive decomposition if temperature control is not meticulously maintained, making large-scale operations inherently risky.
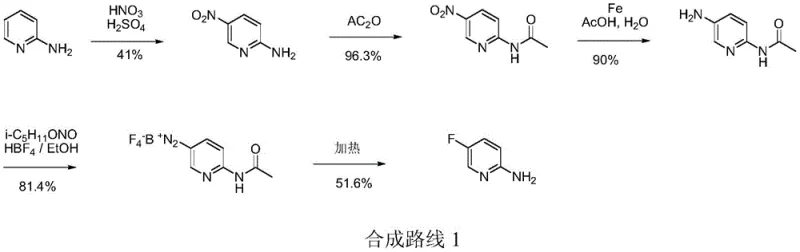
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent data重构 s the synthetic logic by building the pyridine ring from an aliphatic precursor rather than modifying an existing aromatic system. This approach initiates with the acid-catalyzed addition of elemental halogens to 4-cyano-1-butyne, forming a tetrahalogenated n-valeronitrile intermediate with exceptional selectivity. This intermediate then undergoes a direct cyclization reaction with ammonia to construct the pyridine core, effectively installing the amino and halogen groups in their correct positions simultaneously. By eliminating the need for protecting group strategies (like acetylation) and avoiding the generation of unstable diazonium species, this route simplifies the process flow significantly. The ability to potentially execute this as a one-pot reaction further enhances its appeal, reducing solvent usage, energy consumption, and labor costs while delivering product purities exceeding 99%.
Mechanistic Insights into Acid-Catalyzed Halogenation and Cyclization
The core of this technological advancement relies on a precise mechanistic sequence that ensures high regioselectivity and minimal byproduct formation. In the first stage, the carbon-carbon triple bond of 4-cyano-1-butyne acts as a nucleophile, attacking the halogen molecule activated by a Lewis or Brønsted acid catalyst such as ferric trichloride or hydrobromic acid. This electrophilic addition proceeds with high fidelity to generate the 4,4,5,5-tetrahalogenated species, where the electron-withdrawing cyano group helps stabilize the transition state and directs the addition. The choice of catalyst and the molar ratio of halogen to alkyne are critical parameters; the patent specifies a catalyst loading of merely 0.005 to 0.03 molar equivalents, demonstrating high catalytic efficiency. This step is conducted at moderate temperatures (0-80°C), preventing side reactions such as polymerization or over-halogenation that could complicate downstream purification.
Following the halogenation, the mechanism shifts to a nucleophilic cyclization driven by ammonia. The terminal dihalogenated moiety of the intermediate is highly reactive towards nucleophilic attack by ammonia, leading to the formation of an imine or amidine-like species which subsequently undergoes intramolecular condensation. This cyclization step effectively aromatizes the six-membered ring while expelling hydrogen halide, resulting in the stable 2-amino-5-halogenated pyridine structure. The use of aqueous ammonia or ammonia-alcohol solutions facilitates this transformation under relatively mild thermal conditions (20-100°C). Crucially, the high selectivity (>98%) observed in gas phase detection indicates that the reaction pathway is kinetically favored towards the target isomer, minimizing the formation of regioisomers or polymeric tars that typically burden crude product streams in traditional aromatic substitutions.
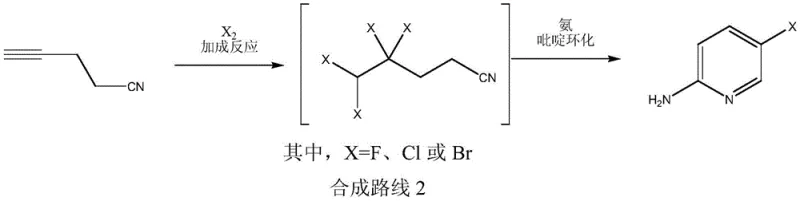
How to Synthesize 2-Amino-5-Halogenated Pyridine Efficiently
Implementing this synthesis requires careful control of reaction parameters to maximize the benefits of the one-pot design. The process begins by dissolving 4-cyano-1-butyne in a chlorinated solvent such as 1,2-dichloroethane or dichloromethane, followed by the addition of a catalytic amount of acid. Halogen gas is then introduced at a controlled rate to manage exotherms, ensuring the formation of the tetrahalo-intermediate. Once the addition is complete, residual acid gases are purged with nitrogen, and an ammonia source is introduced directly to the same vessel to trigger cyclization. For detailed operational parameters, stoichiometry, and work-up procedures, please refer to the standardized synthesis guide below.
- Perform an addition reaction between 4-cyano-1-butyne and a halogen simple substance (Cl2, Br2, or F2) in a solvent like dichloromethane, catalyzed by an acid such as ferric trichloride, to form a tetrahalogenated n-valeronitrile intermediate.
- Without isolating the intermediate, introduce an ammonia source (e.g., aqueous ammonia or ammonia alcohol solution) to the reaction mixture to initiate pyridine cyclization.
- Maintain the cyclization temperature between 20-100°C for 2-10 hours, then separate the organic layer, wash, dry, and recover the solvent to obtain high-purity 2-amino-5-halogenated pyridine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative benefits that extend beyond simple yield improvements. The shift from expensive, specialized aromatic starting materials to commodity aliphatic nitriles fundamentally alters the cost structure of production. By removing the dependency on 2-aminopyridine, which is subject to volatile market pricing and limited supplier bases, manufacturers can secure a more stable and predictable raw material supply. Furthermore, the elimination of hazardous unit operations such as diazotization reduces the regulatory burden and insurance costs associated with handling energetic materials, directly contributing to a lower total cost of ownership for the manufacturing asset.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the drastic simplification of the synthetic sequence. By combining halogenation and cyclization into a streamlined workflow, potentially within a single reactor, the requirement for intermediate isolation, drying, and purification steps is removed. This reduction in unit operations translates to significant savings in energy consumption, solvent recovery costs, and labor hours. Additionally, the use of inexpensive catalysts like ferric chloride instead of precious metals or stoichiometric reagents further lowers the variable cost per kilogram, enabling competitive pricing for high-purity pharmaceutical intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step syntheses where a failure in any single stage can halt production. This robust two-step protocol minimizes points of failure and utilizes widely available bulk chemicals like chlorine, bromine, and ammonia, which are produced on a massive industrial scale globally. The high selectivity of the reaction means that crude product quality is consistently high, reducing the need for extensive reprocessing or recycling of off-spec material. This reliability allows supply chain planners to forecast lead times with greater accuracy and maintain leaner inventory levels while ensuring uninterrupted delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the ability to manufacture with a reduced ecological footprint is a strategic imperative. This method generates significantly less acidic wastewater compared to nitration-based routes and avoids the creation of heavy metal waste or explosive diazonium byproducts. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for specialized explosion-proof infrastructure, facilitating easier scale-up from pilot plant to commercial tonnage. This alignment with green chemistry principles not only ensures long-term regulatory compliance but also enhances the brand reputation of the supplier among environmentally conscious multinational clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy processes in terms of safety, yield, and operational simplicity. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary safety advantages of this new synthesis route compared to traditional nitration methods?
A: The new route eliminates the use of hazardous diazonium salts and strong mixed acids (nitric/sulfuric), significantly reducing the risk of explosion and the generation of acidic wastewater, thereby enhancing operational safety and environmental compliance.
Q: Can this process be adapted for different halogens like fluorine, chlorine, and bromine?
A: Yes, the method is highly versatile and serves as a general platform technology; by simply switching the halogen simple substance (F2, Cl2, or Br2) used in the initial addition step, manufacturers can produce 2-amino-5-fluoropyridine, 2-amino-5-chloropyridine, or 2-amino-5-bromopyridine with high selectivity.
Q: How does the atom economy of this method impact production costs?
A: By utilizing cheap and readily available 4-cyano-1-butyne as a starting material and employing a one-pot strategy that avoids intermediate isolation, the process maximizes atom economy and minimizes unit operations, leading to substantial reductions in raw material and processing costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-Halogenated Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to innovative synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN110092746B are fully realized in practical application. We operate stringent purity specifications and maintain rigorous QC labs equipped to verify the absence of critical impurities, guaranteeing that every batch of 2-amino-5-halogenated pyridine meets the exacting standards required for pharmaceutical synthesis.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value and security for your global operations.
