Advanced Aqueous Chlorination Strategy for High-Purity 2-Amino-5-Chloropyridine Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates, particularly those serving high-demand therapeutic areas such as sedative-hypnotics. Patent CN108675955B introduces a transformative preparation method for 2-amino-5-chloropyridine, a pivotal building block in the synthesis of zopiclone. This innovation addresses longstanding challenges in halogenation chemistry by shifting from hazardous organic solvent systems to a controlled aqueous environment. By utilizing 2-aminopyridine as the starting material and employing a saturated sodium chlorate solution coupled with a phase transfer catalyst, the process achieves exceptional selectivity. The technical breakthrough lies in the precise regulation of chlorine atom release, which effectively suppresses the formation of polychlorinated byproducts that typically plague traditional electrophilic substitution reactions. This approach not only enhances the chemical integrity of the final product but also aligns with modern green chemistry principles by significantly reducing the reliance on volatile organic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-5-chloropyridine has been fraught with operational hazards and efficiency bottlenecks that complicate supply chain stability for pharmaceutical manufacturers. Traditional protocols often involve the direct chlorination of 2-aminopyridine using chlorine gas in concentrated sulfuric acid, a method that poses severe safety risks due to the handling of toxic gases and corrosive acids. Furthermore, the reaction kinetics in these acidic media are difficult to modulate, frequently leading to over-chlorination and the generation of stubborn impurities like 3,5-dichloro-2-aminopyridine. Alternative methods utilizing potassium chlorate in ethanol have shown improvement but still suffer from the inherent drawbacks of using large volumes of flammable organic solvents, which escalate fire risks and necessitate expensive solvent recovery infrastructure. The cumulative effect of these inefficiencies is a process with lower overall yields, higher waste disposal costs, and a final product that requires extensive, multi-step purification to meet the stringent purity standards required for API intermediates.
The Novel Approach
In stark contrast to these legacy techniques, the novel methodology disclosed in the patent leverages a sophisticated aqueous phase transfer catalysis system to redefine the chlorination landscape. The core of this innovation involves dissolving the substrate in dilute hydrochloric acid and introducing it into a saturated sodium chlorate solution under strictly controlled low-temperature conditions ranging from 0°C to -5°C. The addition of TEBA (benzyltriethylammonium chloride) acts as a crucial mediator, facilitating the transport of reactive species across phase boundaries and ensuring a slow, steady release of active chlorine. This controlled release mechanism is the key to minimizing side reactions, allowing the reaction to proceed with high regioselectivity towards the desired 5-position. The result is a crude product with a purity approaching 98%, which can be further refined to over 99.8% through a simple decolorization and crystallization process using methanol and activated carbon.
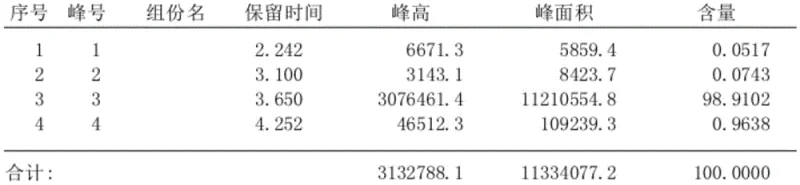
Mechanistic Insights into TEBA-Catalyzed Aqueous Chlorination
To fully appreciate the technical superiority of this route, one must examine the mechanistic role of the phase transfer catalyst within the aqueous matrix. In a standard biphasic system without a catalyst, the interaction between the organic-soluble aminopyridine salt and the inorganic chlorate oxidant would be kinetically limited by the interface area. TEBA functions by forming lipophilic ion pairs that shuttle the chlorate or active chlorinating species into the organic-like microenvironment surrounding the protonated amine. This proximity effect dramatically increases the effective concentration of reactants at the reaction site without requiring bulk organic solvents. Moreover, the saturation of the sodium chlorate solution ensures a thermodynamic driving force that maintains a consistent potential for oxidation, while the low temperature serves as a kinetic brake to prevent runaway exothermic events. This delicate balance allows for the mono-chlorination to dominate, effectively shutting down the pathways that lead to di- or tri-chlorinated impurities which are energetically accessible at higher temperatures or uncontrolled addition rates.
From an impurity control perspective, the mechanism inherently favors the formation of the target molecule by exploiting the electronic properties of the pyridine ring under acidic conditions. The protonation of the amino group and the ring nitrogen deactivates the ring towards electrophilic attack, necessitating a potent yet controlled chlorinating agent to achieve substitution. The in situ generation of active chlorine from the chlorate-hydrochloride system provides just the right oxidative power to overcome this deactivation at the 5-position, which is electronically favored over the 3 or 6 positions due to resonance and inductive effects. Any unreacted starting material remains largely in the form of the hydrochloride salt, which exhibits different solubility characteristics compared to the neutral chlorinated product. This difference is exploited during the workup phase, where freezing the reaction mixture causes the product to precipitate while leaving soluble salts and minor impurities in the mother liquor, thereby achieving a preliminary purification that simplifies downstream processing significantly.
How to Synthesize 2-Amino-5-Chloropyridine Efficiently
Implementing this synthesis protocol requires strict adherence to the specified stoichiometric ratios and thermal profiles to ensure reproducibility and safety on a commercial scale. The process begins with the preparation of the acidified amine solution, followed by the critical addition step into the chilled oxidant bath where the TEBA catalyst is already dispersed. Operators must monitor the temperature vigilantly to prevent local hot spots that could trigger side reactions. Following the reaction completion, the isolation strategy relies on physical property differences rather than complex chemical extractions, utilizing freezing and filtration to separate the solid crude product. For detailed operational parameters, stoichiometry, and safety precautions, please refer to the standardized synthesis guide below.
- Dissolve 2-aminopyridine in a 5-10% hydrochloric acid solution to form reaction solution A.
- At low temperature (0°C to -5°C), drip solution A into a saturated sodium chlorate aqueous solution containing 1-3% TEBA catalyst.
- Freeze the reaction mixture, filter to obtain crude product, and refine with methanol and activated carbon to achieve >99.8% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this aqueous-based synthesis represents a strategic opportunity to optimize cost structures and mitigate supply risks associated with volatile organic solvent markets. The elimination of ethanol or other organic solvents from the reaction medium drastically reduces the raw material procurement burden and removes the logistical complexities associated with storing and transporting large quantities of flammable liquids. Furthermore, the simplified workup procedure, which avoids energy-intensive distillation steps for solvent recovery, translates directly into lower utility consumption and reduced processing time per batch. This efficiency gain allows manufacturing facilities to increase throughput without expanding physical footprint, thereby enhancing the overall agility of the supply chain to respond to fluctuating market demands for zopiclone and related pharmaceutical products.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and hazardous organic solvents with water, which is universally available and inexpensive. By removing the need for complex solvent recovery systems and reducing the volume of hazardous waste generated, facilities can achieve substantial savings in both operational expenditure and waste disposal fees. The high selectivity of the reaction means that less raw material is wasted on forming byproducts, effectively increasing the mass efficiency of the process and lowering the cost per kilogram of the active pharmaceutical ingredient intermediate. Additionally, the simplicity of the purification step using methanol and activated carbon is far less capital intensive than the chromatographic or multi-step recrystallization processes often required for lower-purity crude materials.
- Enhanced Supply Chain Reliability: Relying on stable, inorganic reagents like sodium chlorate and hydrochloric acid insulates the production schedule from the price volatility and supply disruptions common in the petrochemical-derived solvent market. The robustness of the aqueous system ensures consistent batch-to-batch quality, reducing the incidence of out-of-specification results that can delay shipments and strain customer relationships. Because the reaction conditions are mild and easily controllable, the risk of catastrophic batch failure due to thermal runaway is minimized, providing a more predictable and reliable production timeline. This stability is crucial for maintaining continuous supply lines to downstream API manufacturers who operate on tight just-in-time inventory models.
- Scalability and Environmental Compliance: The environmental profile of this method offers a distinct advantage in regions with increasingly stringent regulations on industrial emissions and effluent discharge. By generating significantly less organic waste and avoiding the release of chlorinated volatile organic compounds, the process simplifies the permitting process and reduces the liability associated with environmental compliance. The scalability is further enhanced by the fact that heat management in aqueous systems is generally more efficient than in organic solvents, allowing for safer scale-up from pilot plant to full commercial production volumes. This alignment with green chemistry principles not only future-proofs the manufacturing asset but also appeals to end-clients who are prioritizing sustainability in their supplier selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced chlorination technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear picture of the operational realities and benefits. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios or for procurement specialists assessing the long-term viability of suppliers utilizing this method.
Q: What are the primary impurities in conventional 2-amino-5-chloropyridine synthesis?
A: Conventional methods often produce significant amounts of 3,5-dichloro-2-aminopyridine and 3,5,6-trichloro-2-aminopyridine due to uncontrolled chlorination, requiring complex purification steps.
Q: How does the aqueous TEBA-catalyzed method improve yield?
A: By using saturated sodium chlorate in water with TEBA, chlorine atoms are released slowly and controllably, minimizing side reactions and boosting yield to approximately 93%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the elimination of organic solvents and the use of simple freezing filtration for separation make this method highly scalable and environmentally compliant for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-Chloropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of innovative synthetic routes like the one described in CN108675955B requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to this high-efficiency process is seamless and risk-free. Our facilities are equipped with state-of-the-art reactors capable of maintaining the precise low-temperature conditions required for this chlorination, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards of the global pharmaceutical industry. We are committed to delivering not just a chemical product, but a comprehensive solution that enhances your supply chain resilience.
We invite you to engage with our technical team to explore how this optimized synthesis can drive value for your specific projects. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this aqueous-based method for your operations. We encourage you to contact our technical procurement team today to obtain specific COA data from our recent batches and to discuss detailed route feasibility assessments tailored to your production volume requirements. Let us collaborate to secure a sustainable and cost-effective supply of high-purity 2-amino-5-chloropyridine for your critical drug development programs.
