Advanced Nickel-Catalyzed Synthesis of Benzofuranones for Industrial Scale Manufacturing
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic routes. Patent CN109503533B introduces a groundbreaking methodology for the synthesis of benzofuranone compounds, a class of molecules critical to the pharmaceutical, agrochemical, and polymer industries. This innovation leverages nickel trifluoromethanesulfonate as a highly effective Lewis acid catalyst to facilitate the condensation of mandelic acid derivatives with phenols. Unlike traditional processes that often rely on corrosive mineral acids or volatile organic solvents, this novel approach operates under solvent-free vacuum conditions. The technical breakthrough lies in its ability to achieve high conversion rates at moderate temperatures ranging from 100°C to 160°C, thereby minimizing energy consumption and thermal degradation of sensitive functional groups. For R&D directors and process chemists, this represents a pivotal shift towards sustainable catalysis that does not compromise on yield or purity.
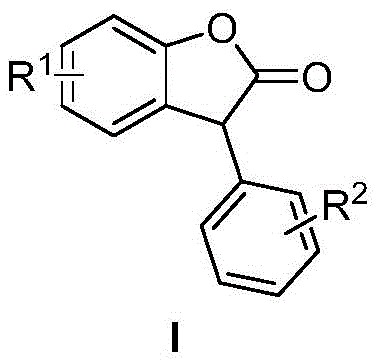
Furthermore, the versatility of this synthetic pathway is demonstrated by its tolerance to a wide array of substituents, including alkyl, alkoxy, halogen, and nitro groups on both the mandelic acid and phenol rings. This broad substrate scope is essential for the rapid development of diverse libraries of bioactive molecules, particularly for drug discovery programs targeting antitumor activities or antioxidant properties. The patent explicitly details the successful synthesis of various 3-arylbenzofuranones, proving the robustness of the catalytic system. By eliminating the need for complex workup procedures associated with liquid acid catalysts, this method streamlines the downstream processing, directly addressing key pain points in commercial scale-up such as waste treatment and operator safety. As a reliable benzofuranone supplier, understanding these mechanistic advantages is crucial for optimizing supply chains and ensuring consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of benzofuranone derivatives has been plagued by significant technical and environmental challenges. Conventional synthetic routes frequently employ strong protic acids like sulfuric acid or polyphosphoric acid, which necessitate rigorous neutralization steps and generate substantial amounts of acidic wastewater. These harsh conditions often lead to poor selectivity, resulting in complex impurity profiles that are difficult and costly to remove, especially when scaling up to multi-ton quantities. Additionally, many traditional methods require the use of large volumes of organic solvents to dissolve reactants and manage heat transfer, which introduces severe safety hazards related to flammability and volatility. The removal of these solvents post-reaction typically involves energy-intensive distillation processes, further inflating the carbon footprint and operational expenditure of the manufacturing process. Moreover, the reliance on stoichiometric amounts of dehydrating agents creates a massive burden on waste management systems, making compliance with increasingly stringent environmental regulations difficult for manufacturers.
The Novel Approach
In stark contrast, the method disclosed in CN109503533B offers a paradigm shift by utilizing a catalytic amount of nickel trifluoromethanesulfonate in a solvent-free environment. This approach fundamentally alters the reaction thermodynamics and kinetics, allowing the condensation to proceed efficiently under vacuum. The absence of solvent not only eliminates the risks associated with VOC emissions but also drastically increases the effective concentration of reactants, driving the equilibrium towards the desired lactone product. The mild reaction conditions, typically maintained between 100°C and 160°C, preserve the integrity of sensitive functional groups that might otherwise decompose under the extreme conditions of classical Friedel-Crafts type cyclizations. 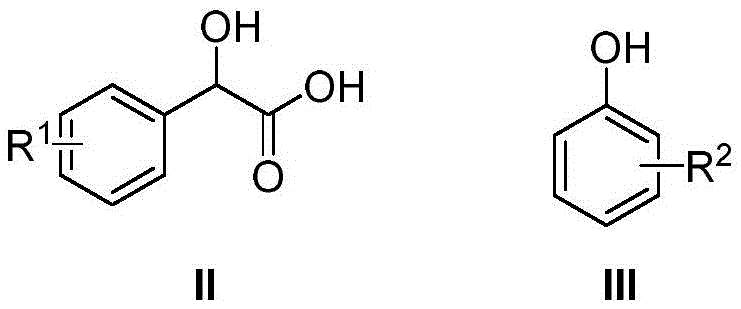
This green chemistry protocol significantly simplifies the operational workflow, transforming a multi-step, hazardous process into a streamlined one-pot synthesis. The ability to recycle the nickel catalyst further enhances the economic viability of this route, reducing the dependency on expensive metal reagents. For procurement managers, this translates to a more stable cost structure, as the process is less susceptible to fluctuations in solvent prices and waste disposal fees. The high selectivity observed in this catalytic system ensures that the final product meets stringent purity specifications required for pharmaceutical applications, reducing the need for extensive recrystallization or chromatographic purification steps.
Mechanistic Insights into Ni(OTf)2-Catalyzed Cyclization
The efficacy of nickel trifluoromethanesulfonate in this transformation stems from its potent Lewis acidity, which activates the carbonyl group of the mandelic acid derivative towards nucleophilic attack by the phenol. In the proposed mechanism, the nickel center coordinates with the oxygen atoms of the carboxylic acid moiety, increasing the electrophilicity of the carbonyl carbon. This activation lowers the energy barrier for the initial esterification or direct C-C bond formation step, depending on the specific pathway taken. Under vacuum conditions, the water molecule generated during the condensation is continuously removed from the reaction mixture. This continuous removal of the byproduct shifts the chemical equilibrium towards the product side according to Le Chatelier's principle, ensuring high conversion even without a large excess of reagents. The stability of the nickel catalyst under these thermal conditions allows it to maintain its activity throughout the 12-hour reaction window, facilitating the subsequent intramolecular cyclization that forms the furanone ring.
From an impurity control perspective, the mildness of the nickel catalyst is a distinct advantage over stronger Bronsted acids. Strong acids often promote side reactions such as polymerization of the phenol ring or rearrangement of the mandelic acid backbone, leading to tarry byproducts that are difficult to separate. The specific coordination geometry of the nickel complex likely imposes a degree of steric control, favoring the formation of the 3-arylbenzofuranone scaffold over other isomeric lactones. This inherent selectivity is critical for maintaining a clean impurity profile, which is a top priority for R&D directors overseeing the development of API intermediates. Furthermore, the solvent-free nature of the reaction minimizes the risk of solvent-induced side reactions or the formation of solvates, which can complicate the crystallization behavior of the final product. Understanding these mechanistic nuances allows process engineers to fine-tune parameters such as temperature and vacuum pressure to maximize yield and minimize trace impurities.
How to Synthesize 3-Arylbenzofuranones Efficiently
The implementation of this catalytic protocol requires precise control over reaction parameters to ensure reproducibility and safety on a larger scale. The process begins with the careful weighing of mandelic acid derivatives and phenol derivatives, typically in a molar ratio that favors the limiting reagent to drive conversion, although the patent suggests a specific stoichiometry for optimal results. The nickel trifluoromethanesulfonate catalyst is then introduced, and the mixture is subjected to a vacuum to remove any adsorbed moisture before heating commences. Maintaining the temperature within the 100-160°C range is critical; temperatures that are too low may result in incomplete conversion, while excessive heat could lead to decomposition. Detailed standardized synthesis steps, including specific workup procedures and purification protocols, are outlined in the technical guide below to assist process chemists in replicating these high-yield results.
- Combine mandelic acid derivatives and phenol derivatives with 10 mol% nickel trifluoromethanesulfonate catalyst in a reaction vessel.
- Heat the mixture to 100-160°C under vacuum conditions without any additional solvent for approximately 12 hours.
- Upon completion, isolate the target benzofuranone product through column chromatography separation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed solvent-free technology offers profound strategic benefits beyond mere technical performance. The elimination of organic solvents removes a major variable from the supply chain, mitigating risks associated with solvent shortages, price volatility, and hazardous material transportation. Since the reaction does not require specialized corrosion-resistant equipment needed for handling strong mineral acids, existing stainless steel reactors can often be utilized, reducing capital expenditure for facility upgrades. The simplicity of the one-pot process reduces the overall cycle time per batch, effectively increasing the throughput of existing manufacturing assets without the need for new construction. This efficiency gain is vital for meeting tight delivery schedules and responding rapidly to market demand fluctuations for high-value intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic reduction in raw material and waste management costs. By operating without solvents, the facility saves significantly on the purchase of bulk solvents and the energy required for their recovery and distillation. The ability to recycle the nickel catalyst multiple times further amortizes the cost of the metal reagent over many batches, lowering the cost of goods sold (COGS). Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts, improving the overall atom economy. These factors combine to create a leaner, more cost-effective manufacturing model that enhances competitiveness in the global market for fine chemicals.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals like mandelic acid and phenol derivatives ensures a robust and resilient supply chain. These starting materials are produced on a massive scale globally, reducing the risk of supply disruptions compared to exotic or proprietary reagents. The simplified process flow also reduces the dependency on complex utility support systems, such as large-scale solvent recovery units or specialized acid scrubbing towers. This operational simplicity translates to higher plant availability and fewer unplanned shutdowns due to equipment maintenance or regulatory inspections. Consequently, suppliers can offer more reliable lead times and consistent volume availability to their downstream customers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of solvent-related heat transfer limitations. The exothermic nature of the reaction is easier to manage in a concentrated, solvent-free melt, allowing for safer scale-up to multi-ton reactors. From an environmental standpoint, the near-zero emission of VOCs and the absence of acidic wastewater align perfectly with modern green chemistry principles and strict environmental regulations. This compliance reduces the administrative burden of permitting and reporting, while also enhancing the corporate sustainability profile. Companies adopting this technology can market their products as 'green' or 'sustainably manufactured,' adding value for environmentally conscious end-users in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzofuranone synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production portfolios. The answers cover aspects ranging from catalyst handling to product isolation, ensuring a comprehensive overview of the process capabilities.
Q: What are the primary advantages of using nickel trifluoromethanesulfonate for benzofuranone synthesis?
A: The use of nickel trifluoromethanesulfonate allows for a solvent-free, one-pot reaction under vacuum, significantly reducing waste generation and simplifying the purification process compared to traditional methods requiring harsh acids or toxic solvents.
Q: Can the catalyst be reused in this synthesis process?
A: Yes, the patent highlights that the nickel catalyst can be recycled and reused multiple times efficiently, which contributes to lower operational costs and improved environmental compliance for large-scale production.
Q: What is the typical yield range for this catalytic method?
A: Depending on the specific substituents on the mandelic acid and phenol starting materials, yields can vary significantly, with optimized conditions achieving high yields up to 97% for certain derivatives like 3,5-di-tert-butylphenol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylbenzofuranone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN109503533B for the production of high-value benzofuranone derivatives. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless and efficient. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the exacting standards required for pharmaceutical and agrochemical applications. Our state-of-the-art facilities are equipped to handle solvent-free and vacuum-based processes safely, allowing us to leverage the full cost and efficiency benefits of this green chemistry route for our clients.
We invite you to collaborate with us to optimize your supply chain for benzofuranone intermediates. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and operational excellence can drive value and reliability in your manufacturing operations.
