Scalable Synthesis of Nitric Oxide Donor Prostaglandin Analogues via Optimized Nitration
Introduction to Advanced Prostaglandin Analogue Synthesis
The pharmaceutical landscape for ophthalmic treatments, particularly for lowering intraocular pressure (IOP), demands increasingly stringent purity profiles for active pharmaceutical ingredients (APIs) and their intermediates. Patent CN114174261A discloses a groundbreaking method for preparing hexanoic acid, 6-(nitrooxy)-, (1S,2E)-3-[(1R,2R,3S,5R)-2-[(2Z)-7-(ethylamino)-7-oxo-2-hepten-1-yl]-3,5-dihydroxycyclopentyl]-1-(2-phenylethyl)-2-propen-1-yl ester, commonly referred to as Compound (I). This nitric oxide donor type prostaglandin analogue represents a significant evolution in glaucoma therapy, combining the efficacy of bimatoprost with the vasodilatory properties of nitric oxide. The core innovation lies not merely in the final coupling but in the rigorous control of the synthetic pathway leading to the nitric oxide donor chain, specifically addressing the critical issue of genotoxic impurities that have plagued previous manufacturing routes.
Traditional methods for synthesizing such complex esters often rely on halogenated precursors and heavy metal substitutions, which introduce significant regulatory and environmental burdens. The disclosed technology offers a robust alternative by utilizing a direct nitration strategy on a hydroxy-acid precursor derived from caprolactone. This approach fundamentally shifts the impurity profile, ensuring that the final API meets the rigorous safety standards required for chronic ophthalmic use. For R&D directors and procurement specialists, understanding this shift is vital, as it translates directly into reduced risk of batch rejection and streamlined regulatory filings. The ability to produce this high-value intermediate with a chemical purity exceeding 98% and controlled levels of specific by-products positions this methodology as a new benchmark for reliable pharmaceutical intermediate supplier capabilities in the niche of ophthalmic drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
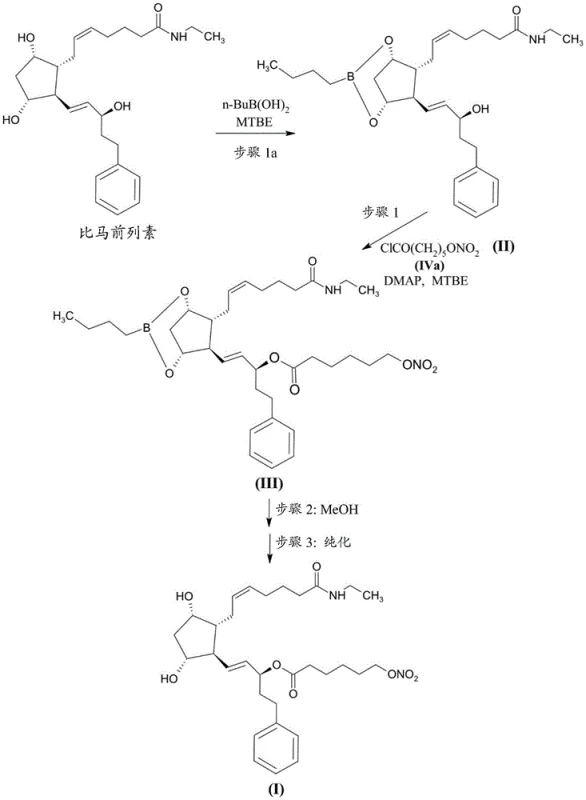
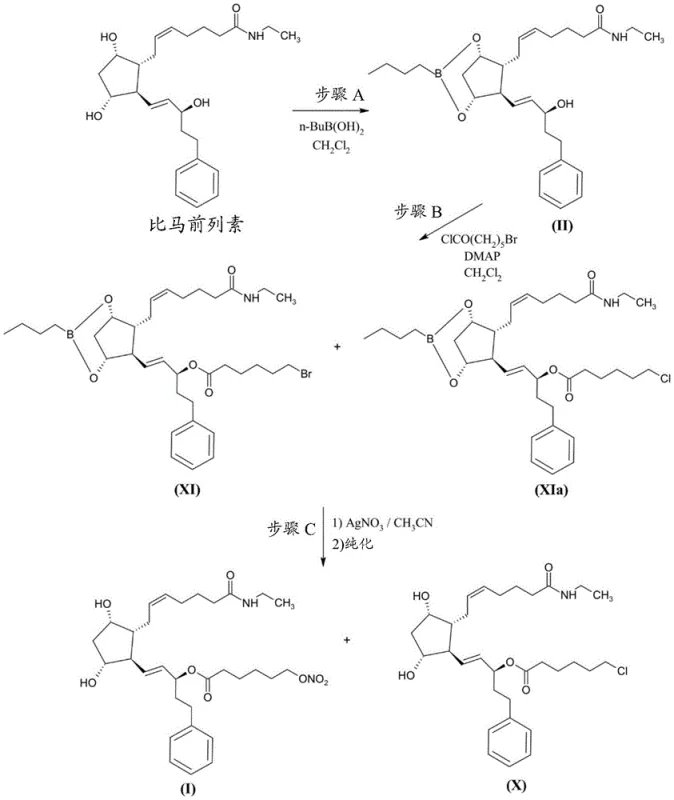
Prior art processes, such as those disclosed in WO 2009/136281, typically rely on a multi-step sequence involving the reaction of a boronate-protected bimatoprost derivative with 6-bromohexanoyl chloride. This initial esterification is followed by a substitution reaction using silver nitrate to convert the bromide into the desired nitrate ester. While chemically feasible, this route presents severe drawbacks for commercial scale-up. The use of stoichiometric amounts of silver nitrate generates substantial quantities of silver-containing waste streams, creating significant environmental disposal costs and complicating wastewater treatment protocols. Furthermore, the reliance on a brominated intermediate introduces the risk of halogen exchange reactions.
Specifically, the presence of chloride ions, often originating from reagents like DMAP hydrochloride used in the esterification step, can lead to the formation of the 15-(6-chlorohexanoyl) ester of bimatoprost, identified as Compound (X). Regulatory assessments predict this chloro-analogue to be positive for bacterial in vitro mutagenicity, classifying it as a genotoxic impurity of high concern. Removing Compound (X) is exceptionally difficult due to its similar polarity and lipophilicity to the target product, often requiring repeated chromatographic purifications that drastically reduce overall yield. Additionally, incomplete conversion of the bromo-intermediate can lead to the persistence of the bromo-ester impurity, further complicating the purification landscape and threatening the safety profile of the final drug substance.
The Novel Approach
The methodology described in CN114174261A circumvents these pitfalls by reversing the logic of the synthesis. Instead of installing a halogen and substituting it later, the process begins with the preparation of high-purity 6-(nitrooxy)hexanoic acid (Compound VIIa). This key building block is synthesized via the ring-opening of 2-caprolactone followed by direct nitration of the resulting 6-hydroxycaproic acid salt. By establishing the nitrate functionality early in the synthesis of the side chain, the process entirely eliminates the need for silver nitrate substitution and avoids the formation of halogenated mutagenic impurities at the source. The subsequent conversion of this purified acid to its acid chloride (Compound IVa) allows for a clean coupling reaction with the protected prostaglandin core.
This strategic pivot ensures that the coupling reaction proceeds with high fidelity, minimizing the formation of the dreaded chloro-impurity (Compound X) and the dimeric impurity (Compound XII). The novel route leverages the stability of the nitrate ester under the specific acidic and coupling conditions employed, provided that the starting acid is sufficiently pure. By decoupling the nitration step from the sensitive prostaglandin core, the process allows for rigorous purification of the side chain before it ever encounters the valuable API precursor. This results in a final product with significantly improved safety margins and a much simpler downstream purification process, representing a substantial advancement in cost reduction in API manufacturing for complex ophthalmic agents.
Mechanistic Insights into Nitration and Impurity Control
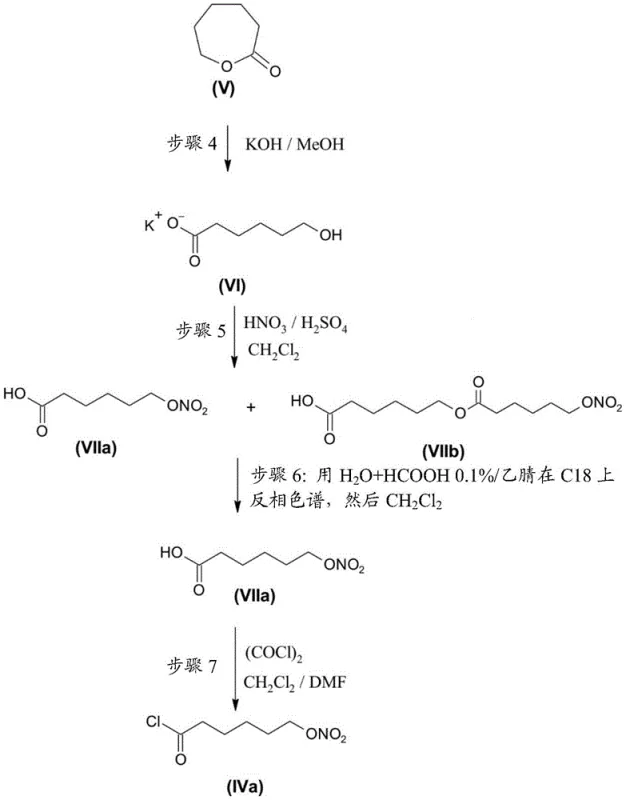
The heart of this technological breakthrough lies in the meticulous preparation and purification of the 6-(nitrooxy)hexanoic acid intermediate. The process initiates with the ring-opening of 2-caprolactone (Compound V) using an inorganic base such as potassium hydroxide in methanol. This yields the potassium salt of 6-hydroxycaproic acid (Compound VI) in high yield. The subsequent nitration step involves treating this salt with a mixture of nitric and sulfuric acids in dichloromethane at controlled low temperatures (0°C to 10°C). While this reaction efficiently installs the nitrooxy group, it inherently produces a dimeric by-product, 6-{[6-(nitrooxy)hexanoyl]oxy}hexanoic acid (Compound VIIb), formed through the esterification of the hydroxy group of one molecule with the carboxylic acid of another under acidic conditions.
If left unchecked, this dimeric acid would be converted to its corresponding acid chloride and subsequently couple with the bimatoprost core to form the dimeric impurity Compound (XII). Because Compound (XII) shares similar chromatographic properties with the target Compound (I), its removal at the final stage is inefficient and yield-destructive. The patent discloses a critical purification intervention: the crude nitrated acid mixture is subjected to reverse phase chromatography (e.g., C18 flash cards) using aqueous formic acid and acetonitrile. This step selectively isolates the monomeric 6-(nitrooxy)hexanoic acid (VIIa), reducing the dimer content to below the HPLC detection limit of 0.05%. Only after this purification is the acid converted to the acid chloride using oxalyl chloride, ensuring that the coupling partner introduced to the bimatoprost scaffold is of exceptional chemical integrity.
How to Synthesize High-Purity Bimatoprost Nitrate Efficiently
The synthesis of Compound (I) requires a disciplined adherence to the optimized reaction parameters outlined in the patent to ensure the suppression of genotoxic impurities. The process flows logically from the preparation of the side chain to the final assembly of the prostaglandin analogue. Operators must pay close attention to temperature controls during the nitration and chlorination steps, as well as the stoichiometry of the coupling reagents. The use of boronate protection on the bimatoprost diol system is essential to prevent acylation at the wrong positions and to facilitate the final deprotection. Detailed standard operating procedures for each unit operation, from the initial caprolactone ring opening to the final silica gel chromatography, are critical for reproducibility.
- Ring-opening of 2-caprolactone with potassium hydroxide to form potassium 6-hydroxycaproate.
- Nitration of the hydroxycaproate salt using HNO3/H2SO4 followed by C18 reverse phase chromatography to remove dimer impurities.
- Conversion to acid chloride and coupling with boronate-protected bimatoprost, followed by deprotection and final purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling strategic advantages beyond mere technical elegance. The most significant impact is the elimination of silver nitrate from the process. Silver is a precious metal with volatile pricing, and its use in stoichiometric quantities represents a substantial variable cost in the traditional route. By removing this reagent, the new process achieves significant cost reduction in pharmaceutical intermediate manufacturing, stabilizing the bill of materials against commodity price fluctuations. Furthermore, the avoidance of heavy metal waste simplifies environmental compliance and reduces the overhead associated with hazardous waste disposal, contributing to a more sustainable and economically predictable production model.
Supply chain reliability is also markedly enhanced by the robustness of the impurity profile. In the legacy process, the formation of mutagenic Compound (X) posed a constant risk of batch failure, potentially leading to supply disruptions if purification limits were exceeded. The new method, by designing out the formation of this impurity, ensures a consistent supply of high-quality material that meets stringent regulatory specifications without the need for excessive reprocessing. This reliability is crucial for maintaining continuous production schedules for downstream API manufacturing. Additionally, the scalability of the caprolactone-based route utilizes readily available bulk chemicals, reducing dependency on specialized halogenated reagents that may face supply constraints, thereby securing the long-term continuity of supply for this critical ophthalmic ingredient.
- Cost Reduction in Manufacturing: The elimination of expensive silver salts and the reduction in chromatographic purification steps directly lower the cost of goods sold (COGS). By preventing the formation of difficult-to-remove impurities early in the synthesis, the process minimizes yield losses associated with aggressive purification, leading to substantial overall cost savings.
- Enhanced Supply Chain Reliability: Utilizing common feedstocks like caprolactone and avoiding specialized halogenated intermediates reduces supply risk. The robust nature of the synthesis ensures consistent batch-to-batch quality, minimizing the likelihood of production delays caused by out-of-specification results related to genotoxic impurities.
- Scalability and Environmental Compliance: The process avoids the generation of heavy metal waste streams, aligning with green chemistry principles and simplifying regulatory environmental reporting. The use of standard organic solvents and reagents facilitates easy scale-up from pilot plant to commercial tonnage without requiring specialized corrosion-resistant equipment for handling aggressive halogenation chemistries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of nitric oxide donor prostaglandin analogues. These insights are derived directly from the experimental data and comparative examples provided in the patent literature. Understanding these nuances is essential for partners evaluating the feasibility of integrating this intermediate into their own supply chains.
Q: How does this process reduce mutagenic impurities compared to prior art?
A: By avoiding the use of 6-bromohexanoyl chloride and silver nitrate substitution, the process eliminates the formation of the 15-(6-chlorohexanoyl) ester impurity (Compound X), which is predicted to be mutagenic.
Q: What is the key purification step for the nitric oxide donor chain?
A: The crude 6-(nitrooxy)hexanoic acid is purified using reverse phase chromatography (C18) with aqueous formic acid and acetonitrile to reduce the dimer impurity (Compound VIIb) to below 0.05%.
Q: Why is the boronate protection strategy used for bimatoprost?
A: Boronate protection of the cis-diol system on the cyclopentane ring prevents side reactions during the esterification step and allows for mild deprotection conditions using methanol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bimatoprost Nitrate Intermediate Supplier
As the demand for advanced ophthalmic therapies continues to grow, the need for high-purity, safely manufactured intermediates has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in complex organic synthesis to deliver solutions that meet the rigorous demands of global pharmaceutical companies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of our prostaglandin intermediates adheres to the highest international standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. Whether you are looking to mitigate regulatory risks associated with genotoxic impurities or seeking to optimize the cost structure of your API production, we are ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to quality and innovation can support your long-term strategic goals in the competitive ophthalmic market.
