Revolutionizing Boronic Ester Synthesis: A Cost-Effective n-Butyllithium Catalytic Route for Industrial Scale-Up
The landscape of organoboron chemistry is undergoing a significant transformation driven by the urgent need for more efficient and cost-effective synthetic routes in the pharmaceutical industry. Patent CN109438493B introduces a groundbreaking methodology that leverages the ubiquitous reagent n-butyllithium to catalyze the hydroboration of aromatic carboxylic acids with pinacolborane. This innovation addresses critical bottlenecks in the production of boronic esters, which are indispensable building blocks for Suzuki-Miyaura cross-coupling reactions widely used in API synthesis. By shifting away from expensive transition metal complexes and harsh reducing agents, this technology offers a pathway to high-purity intermediates with exceptional atom economy. For R&D directors and procurement managers alike, the implications are profound, promising not only simplified purification protocols but also a drastic reduction in the cost of goods sold for complex fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the conversion of carboxylic acids to boronic esters has been plagued by significant technical and economic hurdles that hinder scalable manufacturing. Traditional hydroboration methods often rely on hazardous reagents such as lithium aluminum hydride (LiAlH4) or sodium borohydride (NaBH4), which pose severe safety risks due to their pyrophoric nature and vigorous reactivity with moisture. Furthermore, advanced catalytic systems utilizing transition metal complexes frequently demand rigorous operating conditions, including elevated temperatures around 60°C and extended reaction times exceeding 24 hours. These energy-intensive processes not only inflate operational expenditures but also complicate thermal management in large-scale reactors. Additionally, many prior art catalysts, such as the complex lithium 2,4-bis(2,6-diisopropylanilino)-2-pentenyl structure shown below, require multi-step synthesis themselves, adding layers of cost and supply chain vulnerability before the main reaction even begins.
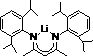
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in the patent utilizes commercially available n-butyllithium as a highly efficient catalyst, fundamentally altering the economic equation of boronic ester production. This method operates under remarkably mild conditions, proceeding smoothly at room temperature and reaching completion within a mere 40 to 50 minutes. The reaction scheme is elegantly simple, involving the direct mixing of the carboxylic acid and pinacolborane followed by the addition of the catalyst, as illustrated in the general reaction pathway below. This simplicity translates directly into operational efficiency, eliminating the need for specialized high-pressure equipment or prolonged heating cycles. Moreover, the system demonstrates exceptional versatility, accommodating a wide range of aromatic substrates including those with electron-withdrawing or electron-donating groups, thereby serving as a robust platform for diverse intermediate synthesis.
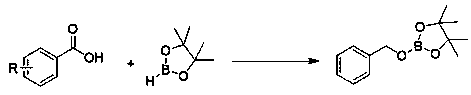
Mechanistic Insights into n-Butyllithium Catalyzed Hydroboration
The efficacy of this system lies in the unique ability of n-butyllithium to activate the carboxylic acid moiety without inducing unwanted side reactions typically associated with strong bases. Mechanistically, the catalyst likely facilitates the deprotonation of the carboxylic acid to form a reactive carboxylate species, which then undergoes nucleophilic attack on the boron center of the pinacolborane. This activation lowers the energy barrier for the hydroboration step, allowing the reaction to proceed rapidly at ambient temperatures. Unlike aldehydes or ketones where the carbonyl carbon is the primary electrophilic site, carboxylic acids present a challenge due to the resonance stabilization of the carbonyl group and the presence of the acidic proton. The n-butyllithium catalyst effectively navigates these electronic constraints, ensuring that the reduction occurs selectively at the carbonyl functionality to yield the desired alcohol-derived boronate without compromising other sensitive functional groups on the aromatic ring.
From an impurity control perspective, this catalytic cycle offers distinct advantages over stoichiometric reduction methods. Because the catalyst loading is exceptionally low, ranging from 0.4% to 0.6% molar equivalent, the introduction of extraneous metallic impurities is minimized. This is critical for pharmaceutical applications where residual metal limits are strictly regulated. The reaction terminates simply upon exposure to air, which quenches any remaining reactive lithium species, followed by a straightforward solvent removal process. This clean workup profile significantly reduces the burden on downstream purification units, such as chromatography or recrystallization, leading to higher overall recovery rates. The ability to achieve yields up to 99% across various substrates, including sterically hindered naphthoic acids and halogenated benzoic acids, underscores the robustness of this mechanistic pathway against structural variations.
How to Synthesize Boronic Esters Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to standard inert atmosphere techniques to ensure optimal catalyst performance and safety. The process begins with the preparation of anhydrous and oxygen-free reaction vessels, typically achieved through nitrogen or argon purging, which is standard practice in fine chemical manufacturing. The aromatic carboxylic acid substrate is first mixed with pinacolborane in a suitable solvent such as tetrahydrofuran, ensuring a homogeneous suspension before catalyst addition. The precise dosing of the n-butyllithium solution is the critical control point, where maintaining the molar ratio between 0.4% and 0.6% is essential for maximizing turnover frequency while minimizing reagent waste. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are provided in the technical documentation below.
- Under an inert gas atmosphere, mix the aromatic carboxylic acid substrate with pinacolborane in a dry reaction vessel.
- Add the n-butyllithium catalyst (0.4-0.6 mol%) to the mixture and stir at room temperature for 40-50 minutes.
- Terminate the reaction by exposure to air, remove the solvent under reduced pressure, and purify to obtain the high-yield boronic ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this n-butyllithium catalyzed process represents a strategic opportunity to optimize the cost structure of key intermediate production. The shift from bespoke, difficult-to-synthesize catalysts to a commodity chemical like n-butyllithium drastically simplifies the raw material sourcing strategy. Since n-butyllithium is produced on a massive industrial scale globally, its supply is stable and its price point is significantly lower than that of specialized transition metal complexes or rare earth catalysts. This transition effectively decouples the production cost from the volatility of the precious metals market, providing a more predictable financial model for long-term manufacturing contracts. Furthermore, the elimination of expensive ligand synthesis steps reduces the overall number of unit operations required, directly translating to lower capital expenditure and reduced utility consumption per batch.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven primarily by the drastic reduction in catalyst cost and the simplification of the reaction workflow. By utilizing a catalyst loading of less than 1 mol%, the process minimizes the consumption of high-value reagents, which is a major cost driver in traditional catalytic systems. Additionally, the ability to run the reaction at room temperature eliminates the energy costs associated with heating large reactor volumes for extended periods, such as the 24-hour cycles seen in older methods. The simplified post-processing, which avoids complex metal scavenging steps often required for transition metal catalysts, further reduces the consumption of purification media and solvents, contributing to substantial overall cost savings in fine chemical manufacturing.
- Enhanced Supply Chain Reliability: Relying on commercially available reagents like n-butyllithium and pinacolborane significantly mitigates supply chain risks associated with proprietary or custom-synthesized catalysts. In the event of supply disruptions for specialized ligands, production lines can face costly downtime; however, the commoditized nature of the reagents in this process ensures a continuous and reliable flow of materials. This reliability is crucial for maintaining just-in-time inventory levels and meeting tight delivery schedules for downstream API manufacturers. The robustness of the reaction across different substrates also means that a single standardized protocol can be applied to multiple products, increasing facility flexibility and reducing the need for specialized training or equipment changes.
- Scalability and Environmental Compliance: The mild reaction conditions and high atom economy of this process make it inherently suitable for commercial scale-up of complex organoboron compounds. Operating at room temperature reduces the thermal load on cooling systems and minimizes the risk of thermal runaway, a critical safety consideration when scaling exothermic reactions. From an environmental standpoint, the high yields and selectivity reduce the generation of chemical waste, aligning with green chemistry principles and easing the burden on waste treatment facilities. The absence of heavy metals in the catalyst system simplifies effluent treatment and ensures that the final product meets stringent regulatory standards for residual impurities without requiring extensive remediation steps.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this n-butyllithium catalyzed hydroboration technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: What are the advantages of using n-butyllithium over transition metal catalysts for boronic ester synthesis?
A: Unlike transition metal complexes which often require high temperatures (60°C), long reaction times (24 hours), and expensive ligands, n-butyllithium operates efficiently at room temperature within 40-50 minutes. It is a commercially available reagent that eliminates the need for complex catalyst synthesis, significantly reducing raw material costs and simplifying the supply chain.
Q: Is this method compatible with sensitive functional groups like halogens?
A: Yes, the patent data demonstrates excellent tolerance for various substituents. Substrates such as 4-bromobenzoic acid, 4-fluorobenzoic acid, and 4-iodobenzoic acid all achieved high yields (90-99%), indicating that the mild conditions preserve sensitive carbon-halogen bonds essential for downstream cross-coupling reactions.
Q: How does the catalyst loading compare to traditional methods?
A: The process utilizes an extremely low catalyst loading of only 0.4% to 0.6% molar ratio relative to the carboxylic acid. This high catalytic activity contrasts sharply with older methods that often require stoichiometric amounts of reducing agents or higher loadings of precious metal catalysts, leading to substantial cost savings in large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boronic Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalytic technology in delivering high-value intermediates to the global market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing environments. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of boronic ester meets the exacting standards required for pharmaceutical synthesis. We are committed to leveraging such innovative chemistries to provide our partners with a competitive edge through superior quality and consistent supply.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact on your specific projects. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a seamless transition to this next-generation manufacturing platform.
