Revolutionizing Lasmiditan Intermediate Production with Advanced Palladium Catalysis for Commercial Scale-up
Revolutionizing Lasmiditan Intermediate Production with Advanced Palladium Catalysis for Commercial Scale-up
The pharmaceutical landscape for migraine treatment has been significantly altered by the approval of Lasmiditan, a selective 5-HT1F receptor agonist. As demand for this novel therapeutic grows, the efficiency of its supply chain becomes paramount, particularly regarding the synthesis of its critical precursors. Patent CN115947718A introduces a transformative methodology for preparing the key intermediate, 2-bromo (or chloro)-6-(1-methylpiperidin-4-yl acyl)-pyridine. This innovation shifts the paradigm from hazardous cryogenic chemistry to a robust, palladium-catalyzed cross-coupling strategy. By leveraging stable boronic acid starting materials, this approach addresses long-standing bottlenecks in yield, safety, and scalability that have plagued earlier synthetic routes. For global procurement teams and R&D directors, understanding this technological leap is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent quality and volume requirements.
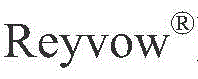
Lasmiditan represents a significant clinical advancement, yet its complex molecular architecture poses challenges for efficient manufacturing. The core structure relies heavily on the precise assembly of a substituted pyridine ring linked to a methylated piperidine moiety. Historically, constructing this bond has required aggressive reagents and extreme conditions. However, the methodology disclosed in CN115947718A simplifies this construction through a direct coupling reaction. This not only streamlines the synthetic pathway but also enhances the overall impurity profile of the resulting material. For stakeholders focused on cost reduction in API manufacturing, this patent offers a viable pathway to optimize production economics while maintaining the high purity standards required for regulatory approval.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthetic routes for Lasmiditan intermediates are fraught with operational hazards and inefficiencies that hinder industrial adoption. Traditional methods often rely on the use of n-butyllithium for metal-halogen exchange at ultra-low temperatures, typically around -78°C. This requirement necessitates specialized cryogenic equipment and rigorous safety protocols to manage the pyrophoric nature of organolithium reagents. Furthermore, these lithiation steps frequently suffer from poor selectivity and low yields, often reported around 10% in early discovery phases. Alternative approaches involving Grignard reagents or Goldberg amidation reactions introduce additional complexities, such as the need for expensive phosphine ligands like BINAP and noble metal catalysts, which drive up raw material costs significantly. The cumulative effect of these factors is a fragile supply chain vulnerable to disruptions and quality deviations.
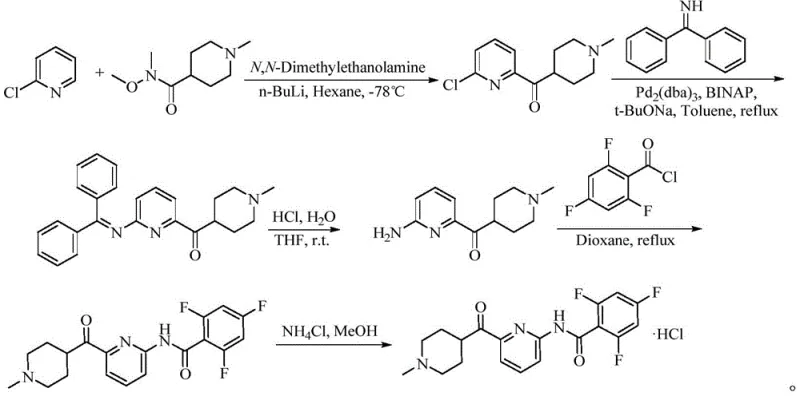
The Novel Approach
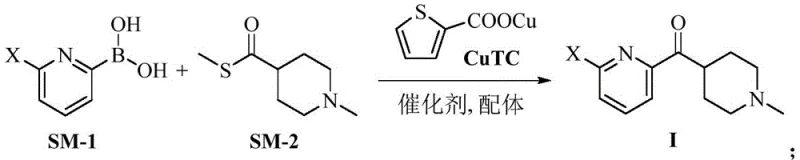
In stark contrast, the novel approach detailed in the patent utilizes a palladium-catalyzed coupling between a pyridyl boronic acid and a thioester derivative. This method operates under remarkably mild conditions, with reaction temperatures ranging from 40°C to 83°C, eliminating the need for energy-intensive cooling systems. The use of copper thiophene-2-carboxylate (CuTC) as a co-catalyst facilitates the acylation step with high efficiency, bypassing the need for unstable organometallic intermediates. This shift in chemistry allows for a dramatic improvement in process safety and operational simplicity. By replacing hazardous lithiation with a stable cross-coupling protocol, manufacturers can achieve yields exceeding 96% with purity levels approaching 99.7%. This robustness makes the process ideally suited for commercial scale-up of complex pharmaceutical intermediates, ensuring consistent supply continuity.
Mechanistic Insights into Pd-Catalyzed Cross-Coupling Acylation
The core of this technological breakthrough lies in the mechanistic elegance of the palladium-catalyzed cross-coupling acylation. Unlike traditional nucleophilic substitutions that require highly reactive organometallic species, this reaction proceeds through a catalytic cycle involving oxidative addition, transmetallation, and reductive elimination. The pyridyl boronic acid serves as a stable nucleophile source, which, in the presence of the palladium catalyst and CuTC, effectively couples with the thioester functionality of the piperidine derivative. The choice of ligand, such as triphenylphosphine, plays a critical role in stabilizing the active catalytic species and promoting turnover. This mechanism avoids the formation of side products commonly associated with over-lithiation or competing nucleophilic attacks, thereby simplifying the downstream purification process. Understanding this catalytic cycle is vital for R&D teams aiming to replicate these results or further optimize reaction parameters for specific plant configurations.
Impurity control is another critical aspect where this mechanism excels. In conventional routes, the harsh conditions often lead to decomposition of the sensitive pyridine ring or the piperidine nitrogen, generating difficult-to-remove byproducts. The mild thermal profile of the new coupling reaction preserves the integrity of these functional groups. Additionally, the workup procedure described involves a straightforward acid-base extraction sequence that effectively removes metal residues and unreacted starting materials. The result is an intermediate with a clean impurity profile, reducing the burden on analytical quality control laboratories. For procurement managers, this translates to a lower risk of batch rejection and a more predictable manufacturing timeline. The ability to consistently produce high-purity material without extensive chromatographic purification is a significant competitive advantage in the generic pharmaceutical market.
How to Synthesize 2-Bromo-6-(1-methylpiperidin-4-yl acyl)-pyridine Efficiently
The implementation of this synthesis route requires careful attention to reagent stoichiometry and reaction monitoring to maximize yield and purity. The process begins with the preparation of the reaction mixture under an inert atmosphere to prevent catalyst deactivation. Key parameters such as temperature control and the order of addition are optimized to ensure smooth progression of the catalytic cycle. While the general procedure is robust, scaling this reaction from laboratory to pilot plant requires validation of mixing efficiency and heat transfer capabilities. The following guide outlines the standardized steps derived from the patent examples to assist technical teams in adopting this superior methodology. Detailed operational parameters and safety data should always be verified against local regulatory guidelines before execution.
- Combine (6-bromopyridin-2-yl)boronic acid, S-(1-methylpiperidin-4-yl) ethanethioate, CuTC, Pd catalyst, and ligand in THF under inert gas.
- Heat the reaction mixture to 50-55°C and maintain until conversion is complete, avoiding cryogenic conditions.
- Filter through Celite, extract with organic solvent after pH adjustment, and concentrate to obtain the high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the adoption of this novel synthetic route offers tangible strategic benefits beyond mere chemical efficiency. The transition away from cryogenic chemistry and hazardous reagents fundamentally alters the cost structure and risk profile of manufacturing this key intermediate. By simplifying the process flow and utilizing commercially available starting materials, companies can achieve substantial cost savings and enhance their supply chain resilience. The following points detail how this technology aligns with broader business objectives regarding cost, reliability, and sustainability.
- Cost Reduction in Manufacturing: The elimination of n-butyllithium and the associated cryogenic infrastructure results in significant operational expenditure reductions. Traditional methods require specialized reactors capable of maintaining -78°C, which are energy-intensive and costly to maintain. In contrast, the new process operates at near-ambient temperatures, allowing the use of standard glass-lined or stainless steel reactors. Furthermore, the high yield of over 96% minimizes raw material waste, directly improving the cost of goods sold (COGS). The removal of expensive ligands like BINAP further contributes to a leaner cost structure, making the final API more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on hazardous or specialty reagents that have limited suppliers. Boronic acids and thioesters used in this new method are stable, shelf-stable commodities with a broad supplier base. This diversification reduces the risk of supply disruptions caused by regulatory restrictions on hazardous chemicals or logistical challenges in transporting pyrophoric materials. Additionally, the simplified workup procedure shortens the overall cycle time per batch, allowing manufacturers to respond more agilely to fluctuations in market demand. This reliability is crucial for maintaining uninterrupted production schedules for the final drug product.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this methodology mitigates those concerns effectively. The absence of volatile, toxic solvents and the reduction in hazardous waste generation align with green chemistry principles. The mild reaction conditions reduce the potential for thermal runaways, a critical safety consideration when moving from kilogram to ton-scale production. Moreover, the simplified purification steps reduce solvent consumption and waste disposal costs. This environmental compliance not only meets regulatory standards but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. They serve to clarify the practical advantages and operational considerations for stakeholders evaluating this technology for adoption.
Q: Why is the new Pd-catalyzed route safer than traditional lithiation methods?
A: Traditional methods require n-butyllithium at -78°C, posing severe thermal runaway and fire risks. The new patent CN115947718A utilizes stable boronic acids at 50-55°C, drastically reducing operational hazards.
Q: How does this process impact the cost of Lasmiditan API manufacturing?
A: By eliminating expensive cryogenic equipment and hazardous reagents like n-BuLi, and achieving yields over 96%, the process significantly lowers both CAPEX and OPEX for large-scale production.
Q: What is the purity profile of the intermediate produced via this method?
A: The patented method consistently delivers intermediates with purity exceeding 99%, minimizing downstream purification burdens and ensuring robust quality control for the final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-6-(1-methylpiperidin-4-yl acyl)-pyridine Supplier
As the pharmaceutical industry continues to evolve, the need for efficient and scalable synthetic routes becomes increasingly critical. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced catalytic technologies to deliver high-quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global API manufacturers. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required for clinical and commercial use. Our expertise in process optimization allows us to translate laboratory innovations like CN115947718A into robust industrial realities.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can drive efficiency and reliability in the production of life-saving migraine treatments, ensuring that patients worldwide have access to high-quality medication.
