Scalable Manufacturing of High-Purity Butylphthalide via Mild Hydrogenation
Scalable Manufacturing of High-Purity Butylphthalide via Mild Hydrogenation
The pharmaceutical industry continuously seeks robust synthetic routes for neuroprotective agents, particularly for the production of butylphthalide, a critical active pharmaceutical ingredient (API) used in treating ischemic stroke. Patent CN107216298B introduces a transformative preparation method that addresses the longstanding challenges of yield, purity, and scalability inherent in traditional synthesis. This innovative approach leverages a mild Raney nickel-catalyzed hydrogenation followed by a sophisticated hydrolysis-precipitation purification strategy, effectively eliminating the need for hazardous high-temperature rectification. By shifting from energy-intensive distillation to crystallization-based purification, this technology offers a compelling value proposition for reliable API intermediate suppliers aiming to optimize their manufacturing footprint. The structural integrity and therapeutic efficacy of the final product are paramount, necessitating a process that rigorously controls stereochemistry and impurity profiles.
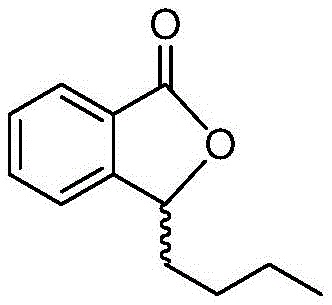
Furthermore, the adoption of this methodology aligns with modern green chemistry principles by utilizing safer solvents like ethanol and dichloromethane instead of highly volatile ethers. The process is designed to be inherently scalable, moving away from laboratory-scale column chromatography towards unit operations suitable for multi-ton production. For procurement managers and supply chain directors, understanding the mechanistic advantages of this route is essential for evaluating long-term supply security. The ability to produce high-purity butylphthalide without complex fractional distillation not only reduces capital expenditure on equipment but also minimizes the thermal degradation of sensitive intermediates. This report analyzes the technical depth of this patent to demonstrate its viability for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of butylphthalide has been plagued by inefficient purification steps and hazardous reaction conditions that hinder industrial application. Traditional methods, such as those described in early academic literature, often rely on heating phthalic anhydride and valeric anhydride at extreme temperatures exceeding 300°C, followed by tedious silica gel column chromatography which yields a mere 25% recovery. Such low efficiency is economically unsustainable for large-scale manufacturing. Other existing protocols utilize diethyl ether as a solvent for hydrogenation with Pd/C catalysts; however, the high volatility and flammability of ether pose significant safety risks in a plant environment. Moreover, these conventional routes typically require high-vacuum rectification to separate the product from isomers and by-products, a process that is both energy-intensive and prone to causing thermal decomposition of the lactone ring. The reliance on precious metal catalysts like palladium further inflates the raw material costs, creating a bottleneck for cost reduction in pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a batch-wise addition of sodium valerate during the precursor synthesis to control exothermic gas release, enhancing safety and reaction consistency. As illustrated in the reaction scheme below, the preferred embodiment employs sodium valerate rather than sodium acetate, which lowers the reaction temperature and mitigates environmental pollution from acetic acid vapors. The core innovation lies in the downstream processing: instead of distilling the crude hydrogenated product, the method employs an alkaline hydrolysis to convert the crude oil into a water-soluble salt, followed by selective acid precipitation. This phase separation technique effectively strips away non-acidic impurities and unreacted starting materials without the need for thermal stress. By isolating the intermediate as a solid hydroxypentylbenzoic acid, the process ensures that the final cyclization step proceeds with a highly purified substrate, guaranteeing exceptional final product quality.
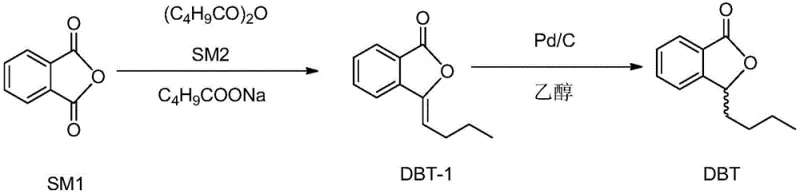
Mechanistic Insights into Raney Nickel-Catalyzed Hydrogenation and Purification
The heart of this synthetic route is the heterogeneous catalysis step using Raney nickel, which offers distinct kinetic advantages over homogeneous or precious metal catalysts. The hydrogenation of the butenyl double bond in butenylphthalide proceeds efficiently at mild temperatures between 15°C and 50°C and moderate pressures of 1 to 10 atm. Raney nickel's high surface area facilitates rapid hydrogen adsorption and transfer, ensuring complete conversion of the alkene to the alkane without over-reduction of the aromatic ring or the lactone carbonyl. This selectivity is crucial for maintaining the structural fidelity of the phthalide core. Furthermore, the use of ethanol as the solvent creates a polar environment that stabilizes the transition state while remaining easy to recover and recycle. The mild conditions prevent the formation of polymeric by-products that often complicate downstream purification in high-temperature processes, thereby simplifying the impurity profile significantly.
Following hydrogenation, the purification mechanism relies on the differential solubility of the intermediate species. The crude reaction mixture is treated with sodium hydroxide, which hydrolyzes the lactone ring to form the corresponding hydroxy-carboxylate salt, soluble in the aqueous phase. Washing this aqueous phase with dichloromethane removes neutral organic impurities, such as unreacted butenylphthalide or fully reduced non-acidic side products. Subsequent acidification with glacial acetic acid to a precise pH of 4-6 triggers the re-precipitation of the hydroxy-acid intermediate. This crystallization step acts as a powerful purification barrier, excluding ionic impurities and residual catalysts. Finally, the purified acid undergoes acid-catalyzed cyclization in an organic solvent, reforming the lactone ring under controlled conditions to yield the target molecule with minimal racemization or degradation.
How to Synthesize Butylphthalide Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory benchtop to pilot plant production. The process is divided into three critical stages: catalytic hydrogenation, hydrolytic purification, and final cyclization. Each step is optimized for maximum yield and minimal waste generation, utilizing common industrial solvents and reagents. The following guide summarizes the operational parameters required to achieve the high purity standards demanded by regulatory bodies. Operators should note that strict temperature control during the acid precipitation phase is vital to ensure the formation of well-defined crystals, which facilitates efficient filtration and washing.
- Perform hydrogenation of butenyl phthalide using Raney nickel catalyst in an alcohol solvent at mild temperatures (15-50°C) and low pressure (1-10 atm).
- Conduct alkaline hydrolysis of the crude intermediate with sodium hydroxide, followed by dichloromethane extraction and acid precipitation to isolate pure hydroxypentylbenzoic acid.
- Execute cyclization of the purified acid intermediate in an acidic organic solvent environment to close the lactone ring and obtain the final butylphthalide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this patented process are profound, offering a pathway to substantial cost optimization without compromising quality. The elimination of high-vacuum rectification columns reduces both the capital investment required for plant infrastructure and the ongoing energy consumption associated with maintaining deep vacuums and high temperatures. By replacing column chromatography with crystallization, the process becomes amenable to continuous or large-batch processing, drastically increasing throughput capacity. The switch from expensive palladium catalysts to Raney nickel represents a direct reduction in raw material costs, while the use of ethanol and dichloromethane ensures that solvent recovery systems can be easily integrated for recycling. These factors collectively contribute to a more resilient and cost-effective supply chain for this critical neuroprotective intermediate.
- Cost Reduction in Manufacturing: The replacement of energy-intensive distillation with crystallization-based purification significantly lowers utility costs. The use of Raney nickel instead of precious metals reduces catalyst expenditure, and the ability to recycle solvents like ethanol and dichloromethane further minimizes variable production costs. This lean manufacturing approach allows for competitive pricing strategies in the global API market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as sodium hydroxide, glacial acetic acid, and Raney nickel ensures that raw material sourcing is not subject to the volatility of the precious metals market. The robustness of the process against minor fluctuations in reaction conditions means that batch-to-batch consistency is high, reducing the risk of production delays due to out-of-specification results. This stability is critical for maintaining uninterrupted supply to downstream drug manufacturers.
- Scalability and Environmental Compliance: The process avoids the use of highly toxic or ozone-depleting solvents, aligning with increasingly stringent environmental regulations. The absence of high-temperature steps reduces the carbon footprint of the manufacturing process. Furthermore, the simplicity of the unit operations—hydrogenation, extraction, filtration, and crystallization—allows for straightforward scale-up from pilot plants to multi-ton commercial reactors without complex engineering modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this synthesis route. These answers are derived directly from the experimental data and process descriptions found in the patent documentation. Understanding these nuances helps technical teams evaluate the feasibility of adopting this method for their own production lines.
Q: How does this purification method avoid the need for high-vacuum rectification?
A: The process utilizes a unique alkaline hydrolysis followed by selective acid precipitation. By converting the crude oil into a solid hydroxy-acid intermediate (hydroxypentylbenzoic acid) through pH control and crystallization, impurities are removed via filtration rather than energy-intensive distillation, significantly lowering operational costs.
Q: Why is Raney Nickel preferred over Palladium on Carbon (Pd/C) for this hydrogenation?
A: Raney Nickel offers superior catalytic activity for this specific alkene reduction under milder conditions compared to precious metal catalysts. It allows the reaction to proceed efficiently at lower pressures (1-10 atm) and ambient temperatures, reducing safety risks associated with high-pressure hydrogenation while drastically cutting catalyst procurement costs.
Q: What represents the main advantage regarding impurity control in this route?
A: The intermediate isolation step acts as a powerful purification barrier. By precipitating the specific hydroxy-acid derivative at a controlled pH of 4-6, unreacted starting materials and side products remain in the mother liquor or organic wash layers, ensuring the final cyclization step begins with a highly pure substrate, resulting in >99% final assay.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Butylphthalide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of butylphthalide meets the highest international standards. Our commitment to technical excellence allows us to deliver consistent quality, supporting our partners in their regulatory filings and clinical trials with confidence.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's success. Let us be your partner in delivering high-purity pharmaceutical solutions to the global market.
