Advanced Palladium Catalyzed Decarboxylation Technology For Commercial Scale-Up Of Complex Pharmaceutical Intermediates
Advanced Palladium Catalyzed Decarboxylation Technology For Commercial Scale-Up Of Complex Pharmaceutical Intermediates
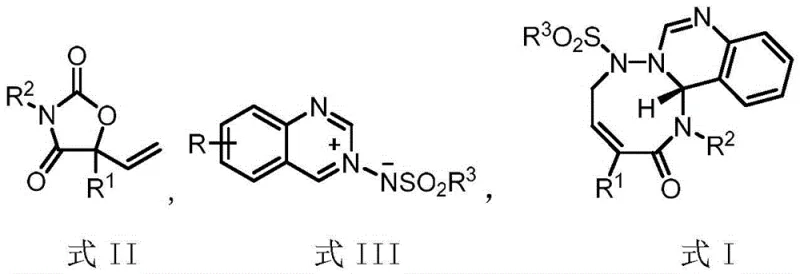
The landscape of organic synthesis for complex heterocyclic scaffolds is undergoing a significant transformation driven by the need for more efficient, stereoselective, and environmentally benign methodologies. A pivotal advancement in this domain is detailed in patent CN116041354A, which discloses a novel palladium-catalyzed decarboxylative [5+3] cycloaddition reaction. This technology leverages 5-vinyloxazolidine-2,4-dione as a versatile allylic zwitterionic precursor to construct medium-ring heterocyclic compounds of Formula I with exceptional precision. For R&D directors and process chemists, this represents a critical breakthrough in accessing structurally diverse nitrogen-containing heterocycles that are often challenging to synthesize via conventional routes. The methodology not only addresses the long-standing issue of regioselectivity in cycloadditions but also provides a robust platform for generating high-value intermediates used in the development of next-generation active pharmaceutical ingredients (APIs).
The core innovation lies in the strategic design of the reactant system, where the decarboxylation event serves as the thermodynamic driving force for the generation of the reactive palladium species. By utilizing readily available starting materials and operating under remarkably mild conditions, this process aligns perfectly with the modern principles of green chemistry and sustainable manufacturing. The ability to achieve high yields and excellent enantiomeric excess (ee) values without the need for cryogenic temperatures or hazardous reagents makes this technology particularly attractive for large-scale production. As we delve deeper into the technical specifics, it becomes evident that this patent offers a reliable pathway for the commercial scale-up of complex pharmaceutical intermediates, addressing both the purity requirements of regulatory bodies and the cost-efficiency demands of global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing medium-ring heterocycles, particularly those containing nitrogen atoms within an eight-membered ring framework, have historically been plagued by significant synthetic hurdles. Conventional thermal cycloaddition reactions often necessitate harsh reaction conditions, including elevated temperatures and prolonged reaction times, which can lead to the degradation of sensitive functional groups and the formation of complex mixtures of regioisomers. Furthermore, achieving high levels of stereocontrol in these thermal processes is notoriously difficult, frequently requiring cumbersome resolution steps that drastically reduce overall material throughput and increase waste generation. The reliance on stoichiometric amounts of organometallic reagents or highly reactive dipolarophiles in older methodologies also introduces safety concerns and complicates the purification process, making them less ideal for the rigorous standards of high-purity pharmaceutical intermediate manufacturing. These limitations often result in low atom economy and prohibitive costs when attempting to translate laboratory discoveries into viable commercial processes.
The Novel Approach
In stark contrast, the methodology described in patent CN116041354A introduces a paradigm shift by employing a transition-metal catalyzed strategy that operates under ambient thermal conditions. The use of 5-vinyloxazolidine-2,4-dione derivatives as latent allylmetal equivalents allows for the in situ generation of reactive intermediates through a clean decarboxylation pathway. This approach effectively bypasses the need for pre-formed, unstable organometallic species, thereby enhancing the operational safety and simplicity of the procedure. The reaction proceeds smoothly in common organic solvents such as dichloromethane at temperatures ranging from 20°C to 30°C, demonstrating remarkable functional group tolerance. This mildness is crucial for preserving the integrity of complex molecular architectures often found in drug candidates. Moreover, the integration of chiral phosphine ligands enables precise control over the stereochemical outcome, delivering products with enantiomeric excess values frequently exceeding 90%, which significantly reduces the burden on downstream purification processes.
Mechanistic Insights into Palladium-Catalyzed Decarboxylative Cycloaddition
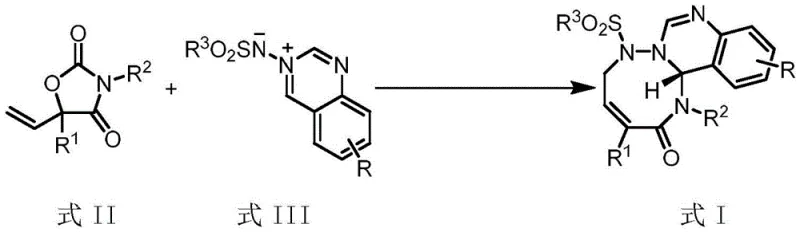
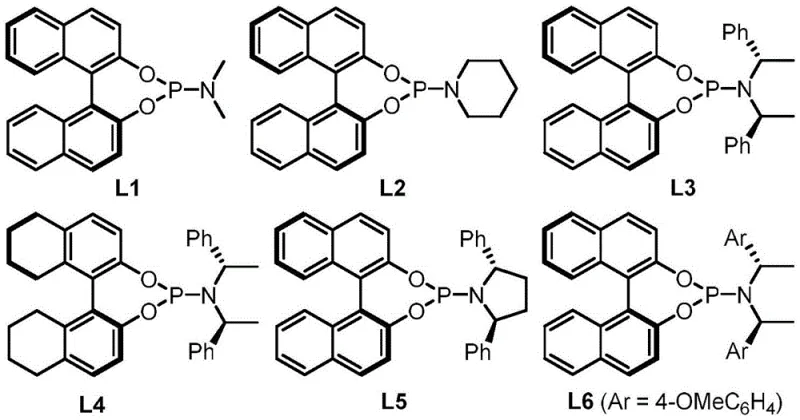
To fully appreciate the sophistication of this technology, one must understand the intricate mechanistic pathway that governs the transformation. The reaction initiates with the coordination of the palladium catalyst, specifically Pd2dba3·CHCl3, to the vinyl group of the oxazolidinedione precursor. This coordination facilitates the oxidative addition and subsequent decarboxylation, generating a key allylpalladium zwitterionic intermediate. This transient species is highly electrophilic and reacts rapidly with the azomethine imine dipole (Formula III) in a concerted [5+3] cycloaddition manifold. The beauty of this mechanism lies in the self-regulating nature of the decarboxylation step, which ensures a steady supply of the reactive allyl species without overwhelming the system, thus minimizing side reactions such as polymerization or oligomerization. The choice of ligand plays a pivotal role in this orchestration; chiral phosphine ligands, particularly the binaphthyl-based structures like L3 shown in the patent data, create a sterically defined environment around the palladium center. This chiral pocket dictates the facial selectivity of the attack on the dipole, ensuring that the newly formed stereocenters are established with high fidelity.
Furthermore, the mechanistic pathway offers inherent advantages regarding impurity control. Because the reaction is catalytic and driven by the irreversible loss of carbon dioxide, the equilibrium is strongly shifted towards the product side, preventing the accumulation of unreacted starting materials that could complicate isolation. The specific interaction between the palladium center and the nitrogen atoms of the azomethine imine also helps to orient the reactants in a way that favors the formation of the desired eight-membered ring over potential [3+2] cycloaddition byproducts. This level of mechanistic control is essential for R&D teams aiming to define a robust design space for regulatory filings. Understanding these nuances allows process chemists to fine-tune parameters such as catalyst loading and ligand-to-metal ratios to optimize the reaction profile for specific substrate classes, ensuring consistent quality across different batches of pharmaceutical intermediates.
How to Synthesize 5-Vinyloxazolidine-2,4-dione Derivatives Efficiently
The successful implementation of this cycloaddition strategy relies heavily on the efficient preparation of the key 5-vinyloxazolidine-2,4-dione precursor (Formula II). The patent outlines a concise two-step sequence starting from readily available alpha-keto esters. Initially, a Grignard addition using vinyl magnesium bromide introduces the necessary vinyl functionality while generating a tertiary alcohol intermediate. This step is critical as it sets the stage for the subsequent ring closure. The second step involves a cyclization reaction with an appropriate isocyanate in the presence of an organic base, which constructs the oxazolidinedione core. This modular approach allows for the easy diversification of the R1 and R2 substituents, enabling the rapid generation of a library of analogues for structure-activity relationship (SAR) studies. The detailed standardized synthesis steps for optimizing this precursor preparation and the subsequent cycloaddition are provided in the guide below.
- Preparation of the 5-vinyloxazolidine-2,4-dione precursor (Formula II) via Grignard addition to alpha-keto esters followed by cyclization with isocyanates.
- Reaction setup involving the mixing of Formula II and azomethine imine (Formula III) with Pd2dba3·CHCl3 catalyst and chiral phosphine ligand L3 in dichloromethane.
- Stirring the mixture at ambient temperature (20-30°C) under inert atmosphere for 12 hours, followed by silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this palladium-catalyzed decarboxylative cycloaddition offers substantial benefits that extend far beyond the laboratory bench. For procurement managers and supply chain heads, the primary value proposition lies in the drastic simplification of the manufacturing workflow and the associated reduction in operational risks. The ability to run reactions at ambient temperatures eliminates the need for energy-intensive heating or cooling infrastructure, directly translating to lower utility costs and a smaller carbon footprint. Additionally, the use of stable, commercially available starting materials mitigates the risk of supply chain disruptions often associated with exotic or custom-synthesized reagents. The high selectivity of the reaction means that fewer purification steps are required, which not only saves time but also reduces the consumption of solvents and chromatography media, leading to significant cost reduction in API manufacturing.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the use of catalytic amounts of palladium rather than stoichiometric metals fundamentally alters the cost structure of producing these heterocycles. By avoiding the need for cryogenic reagents and expensive protecting group strategies often required in alternative routes, the overall cost of goods sold (COGS) is significantly optimized. The high yields reported in the patent data suggest that material throughput is maximized, reducing the amount of raw material needed per kilogram of final product. Furthermore, the simplified workup procedures minimize labor hours and waste disposal costs, creating a leaner and more economically viable production process that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: The robustness of this chemical process contributes directly to supply chain stability. Since the reaction tolerates a wide range of functional groups and substituents, it allows for the use of diverse feedstock sources without compromising product quality. This flexibility is crucial for maintaining continuity of supply in the face of raw material volatility. The scalability of the protocol, demonstrated by its effectiveness in standard solvents like dichloromethane and toluene, ensures that technology transfer from pilot plant to commercial scale can be executed smoothly. This reliability reduces lead time for high-purity pharmaceutical intermediates, allowing downstream partners to plan their production schedules with greater confidence and accuracy.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental impact intensify, this technology offers a compliant pathway forward. The atom economy of the decarboxylative process is superior to many traditional methods, as the only byproduct is carbon dioxide, which is easily managed. The reduction in solvent usage and the avoidance of toxic heavy metal waste streams simplify the environmental permitting process for manufacturing facilities. This alignment with green chemistry principles not only future-proofs the supply chain against tightening regulations but also enhances the brand reputation of companies adopting sustainable practices. The ease of scaling this reaction ensures that demand surges can be met without the need for extensive capital investment in specialized reactor hardware.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity for stakeholders evaluating this synthetic route for their own pipelines. Understanding these details is essential for making informed decisions about process adoption and resource allocation.
Q: What are the key advantages of this palladium-catalyzed method over traditional thermal cycloadditions?
A: This method operates under mild conditions (20-30°C) compared to the high temperatures often required for thermal variants, resulting in superior enantioselectivity (up to 97% ee) and avoiding thermal decomposition of sensitive functional groups.
Q: Is the chiral ligand system scalable for industrial production?
A: Yes, the protocol utilizes commercially available chiral phosphine ligands (such as L3) and standard palladium sources like Pd2dba3·CHCl3, which are well-established in industrial catalysis, ensuring robust scalability without exotic reagents.
Q: How does the decarboxylation mechanism improve atom economy?
A: The reaction utilizes CO2 loss as a driving force to generate the reactive allylpalladium zwitterion in situ, eliminating the need for pre-formed organometallic nucleophiles and reducing waste generation associated with stoichiometric metal byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Vinyloxazolidine-2,4-dione Derivative Supplier
The technological potential of palladium-catalyzed decarboxylative cycloaddition is immense, offering a streamlined route to valuable heterocyclic scaffolds that are integral to modern drug discovery. At NINGBO INNO PHARMCHEM, we recognize the complexities involved in translating such advanced academic and patent literature into robust commercial processes. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from gram-scale optimization to multi-ton manufacturing. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to leverage our expertise to accelerate your development timelines and reduce your overall project costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecular targets, identifying opportunities for efficiency that might otherwise be overlooked. We encourage you to contact our technical procurement team today to request specific COA data for similar intermediates and to discuss comprehensive route feasibility assessments. Let us help you navigate the complexities of modern synthesis and secure a reliable supply chain for your critical pharmaceutical intermediates.
