Advanced Palladium-Catalyzed Decarboxylation for Scalable Pharmaceutical Intermediate Manufacturing
Advanced Palladium-Catalyzed Decarboxylation for Scalable Pharmaceutical Intermediate Manufacturing
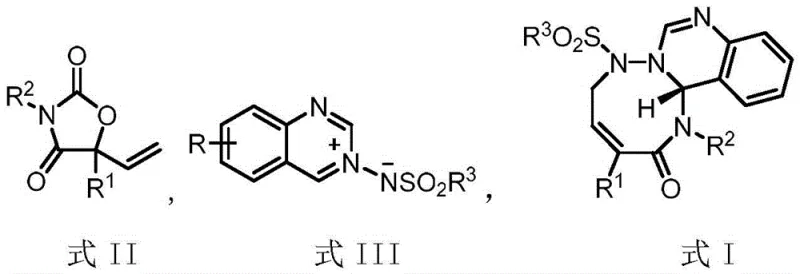
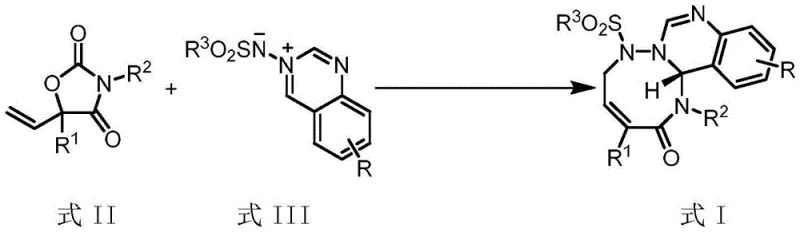
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical industry, particularly in the construction of complex heterocyclic scaffolds. A significant breakthrough in this domain is documented in patent CN116041354A, which discloses a highly efficient palladium-catalyzed decarboxylative [5+3] cycloaddition reaction. This innovative methodology utilizes 5-vinyloxazolidine-2,4-dione as a novel allylic zwitterionic precursor to construct medium-ring heterocyclic compounds of Formula I. For R&D directors and procurement specialists, this technology represents a paradigm shift, offering a pathway to access chemically challenging structures with exceptional stereocontrol. The ability to generate these complex cores under mild conditions addresses long-standing synthetic bottlenecks, positioning this reaction as a critical tool for the development of next-generation active pharmaceutical ingredients (APIs) and high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of medium-ring heterocycles via cycloaddition reactions has been plagued by significant challenges that hinder efficient commercial manufacturing. Conventional palladium-catalyzed decarboxylative cycloadditions often rely on allylic precursors that exhibit poor reactivity or require harsh reaction conditions, such as elevated temperatures and strong bases, which can compromise the integrity of sensitive functional groups. Furthermore, achieving high levels of enantioselectivity in these transformations has historically been difficult, often necessitating complex chiral auxiliaries or stoichiometric amounts of expensive chiral reagents. These limitations result in low overall yields, difficult purification processes, and substantial waste generation, all of which drive up the cost of goods sold (COGS) and extend lead times for reliable pharmaceutical intermediate suppliers. The lack of robust, generalizable methods for constructing these specific ring systems has forced many organizations to rely on linear, step-intensive synthetic routes that are inherently inefficient.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN116041354A introduces a streamlined approach utilizing 5-vinyloxazolidine-2,4-dione derivatives as the key allylpalladium zwitterion precursors. This specific structural motif allows for the generation of reactive intermediates under remarkably mild conditions, typically between 20°C and 30°C, thereby preserving sensitive functionalities and minimizing side reactions. The reaction proceeds with high atom economy and exceptional regioselectivity, directly coupling the precursor with azomethine imine intermediates (Formula III) to form the desired seven-membered ring systems. By employing a catalytic system based on Pd2dba3·CHCl3 and specialized chiral phosphine ligands, this novel approach achieves enantiomeric excess (ee) values exceeding 90% in many cases, with some examples reaching up to 97% ee. This leap in efficiency not only simplifies the synthetic workflow but also drastically reduces the environmental footprint associated with the production of these complex heterocycles.
Mechanistic Insights into Palladium-Catalyzed Decarboxylative Cycloaddition
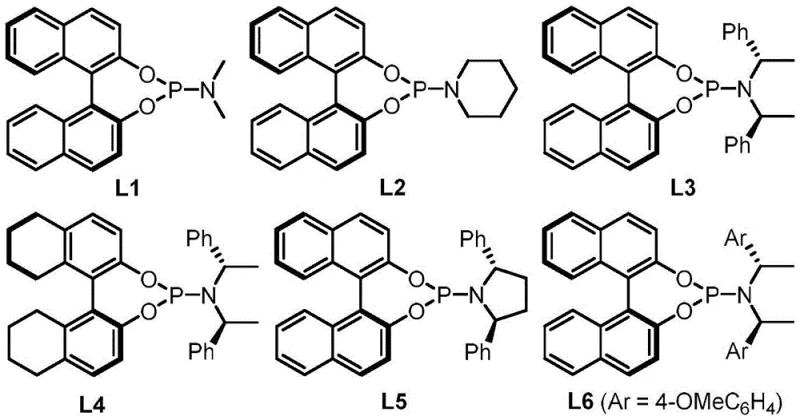
The success of this transformation hinges on the precise interplay between the palladium catalyst and the chiral ligand environment, which dictates both the reactivity and the stereochemical outcome of the reaction. The mechanism initiates with the oxidative addition of the palladium(0) species to the 5-vinyloxazolidine-2,4-dione precursor, triggering a decarboxylation event that generates a transient allylpalladium zwitterion intermediate. This highly reactive species is then intercepted by the electrophilic azomethine imine dipole in a concerted [5+3] cycloaddition manifold. The use of bulky, electron-rich binaphthyl-derived phosphine ligands, such as L3 and L6, creates a well-defined chiral pocket around the metal center. This steric environment effectively shields one face of the reacting species, ensuring that the nucleophilic attack occurs with high facial selectivity. The result is the formation of the new carbon-carbon and carbon-nitrogen bonds with rigorous control over the absolute configuration of the newly generated stereocenters, a critical parameter for the biological activity of the final drug substance.
Beyond stereocontrol, the mechanistic design of this process inherently supports high purity profiles, which is a primary concern for quality assurance teams. The decarboxylation step serves as a driving force for the reaction, pushing the equilibrium towards product formation and minimizing the accumulation of unreacted starting materials. Furthermore, the mild reaction temperatures prevent thermal degradation pathways that often lead to the formation of difficult-to-remove polymeric impurities or regioisomers. The choice of solvent, such as dichloromethane or toluene, further optimizes the solubility of the intermediates while maintaining the stability of the catalytic cycle. By understanding these mechanistic nuances, process chemists can fine-tune reaction parameters—such as the molar ratio of catalyst to substrate (optimized at 1:1 to 1:6) and ligand loading—to maximize yield and minimize the presence of trace metal residues. This level of control is essential for meeting the stringent impurity specifications required for clinical-grade pharmaceutical intermediates.
How to Synthesize 5-Vinyloxazolidine-2,4-dione Derivatives Efficiently
The preparation of the key starting material, the 5-vinyloxazolidine-2,4-dione (Formula II), is itself a robust and scalable two-step process that leverages commodity chemicals. The sequence begins with the addition of vinyl magnesium bromide to an alpha-keto ester (S1) at low temperatures (-78°C to -10°C) to form the tertiary alcohol intermediate (S2). This is followed by a cyclization step involving reaction with an isocyanate (R2NCO) in the presence of an organic base like triethylamine. This precursor synthesis is highly modular, allowing for the easy introduction of diverse aryl and heteroaryl substituents at the R1 and R2 positions. The detailed standardized synthesis steps for the final cycloaddition reaction are outlined below, providing a clear roadmap for laboratory and pilot plant execution.
- Preparation of the 5-vinyloxazolidine-2,4-dione precursor (Formula II) by reacting alpha-keto esters with vinyl magnesium bromide followed by cyclization with isocyanates.
- Reaction of Formula II with azomethine imine intermediates (Formula III) in the presence of Pd2dba3·CHCl3 and chiral phosphine ligands (L1-L6).
- Purification of the resulting cycloadduct (Formula I) via silica gel column chromatography to achieve high enantiomeric excess (ee) values up to 97%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this palladium-catalyzed technology offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the significant simplification of the supply chain for complex heterocyclic building blocks. By replacing multi-step, low-yielding sequences with a single, high-efficiency cycloaddition step, manufacturers can drastically reduce the number of unit operations required. This consolidation translates directly into reduced operational overhead, lower energy consumption, and a smaller physical footprint for production facilities. Furthermore, the reliance on commercially available and inexpensive starting materials, such as ethyl benzoylformate and vinyl magnesium bromide, mitigates the risk of raw material shortages and price volatility. This stability is crucial for maintaining continuous supply lines for long-term commercial projects.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive chiral reagents and the reduction of waste disposal costs. Since the reaction utilizes a catalytic amount of palladium and achieves high conversion rates, the cost per kilogram of the final intermediate is significantly lowered compared to stoichiometric approaches. Additionally, the high enantioselectivity reduces or eliminates the need for costly chiral resolution steps, such as preparative chiral HPLC or recrystallization, which are often the most expensive part of asymmetric synthesis. The mild conditions also allow for the use of standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment, further lowering capital expenditure requirements for scale-up.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a key metric for supply chain reliability. The tolerance of the catalytic system to various functional groups means that a single set of reaction conditions can be applied to a wide library of substrates, simplifying inventory management and reducing the need for specialized reagents for different analogues. This flexibility allows suppliers to respond rapidly to changing demand patterns or design modifications from pharmaceutical partners. Moreover, the use of common organic solvents like dichloromethane and toluene ensures that solvent supply chains remain stable and cost-effective, avoiding dependencies on exotic or regulated solvents that might face logistical bottlenecks.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process aligns well with green chemistry principles. The decarboxylative nature of the reaction releases carbon dioxide as the only byproduct, avoiding the generation of toxic heavy metal waste or halogenated byproducts common in other cross-coupling reactions. The ability to run the reaction at near-ambient temperatures reduces the energy load associated with heating and cooling, contributing to a lower carbon footprint. As regulatory pressures on pharmaceutical manufacturing increase, having a synthetic route that inherently minimizes hazardous waste and energy usage provides a competitive advantage in regulatory filings and sustainability reporting, facilitating faster approval times for new drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed decarboxylative cycloaddition technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for process development teams. Understanding these details is critical for assessing the feasibility of integrating this chemistry into existing manufacturing workflows.
Q: What are the key advantages of using 5-vinyloxazolidine-2,4-dione as a precursor?
A: According to patent CN116041354A, this precursor acts as a highly effective allylpalladium zwitterion source, enabling mild reaction conditions (20-30°C) and excellent enantioselectivity compared to traditional harsh methods.
Q: Which chiral ligands provide the best enantioselectivity in this reaction?
A: The patent identifies binaphthyl-based chiral phosphine ligands, specifically L3 and L6, as providing superior stereocontrol, achieving ee values as high as 97% in dichloromethane solvents.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes readily available starting materials and operates at near-ambient temperatures without requiring extreme pressure, making it highly amenable to commercial scale-up for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Vinyloxazolidine-2,4-dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN116041354A for accelerating drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to verify enantiomeric excess and impurity profiles. We are committed to delivering high-purity heterocyclic compounds that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new targets, our experts are ready to assist. Please contact our technical procurement team today to discuss your needs and discover how we can support your journey from molecule to medicine with reliable, cost-effective, and scalable solutions.
