Advanced One-Pot Synthesis of Bioactive Spiro-Heterocyclic Indole Derivatives for Commercial Scale-Up
Introduction to Novel Spiro-Heterocyclic Synthesis
The pharmaceutical industry continuously seeks efficient pathways to access complex heterocyclic scaffolds that serve as privileged structures in drug discovery. Patent CN102584860A introduces a significant advancement in this domain by disclosing a robust preparation method for spiro-heterocyclic compounds containing indole structures. These compounds, specifically dihydro-spiro[indole-3,4'-pyrazolo[3,4-e][1,4]thiazepine] diketones, represent a class of molecules with profound biological potential, mirroring the activity of natural alkaloids like Spirotryprostatin A and Pteropodine. The innovation lies not just in the molecular architecture but in the operational simplicity of the synthesis, which addresses long-standing challenges in multicomponent reactions.
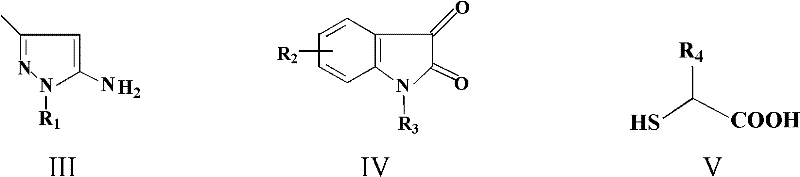
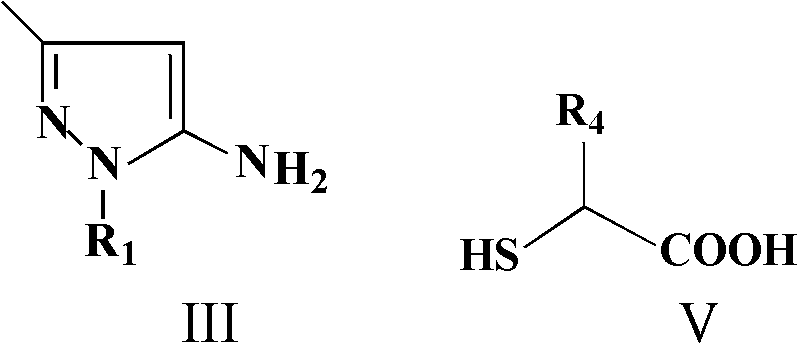
Historically, the construction of spiro-oxindole frameworks has required intricate multi-step sequences or harsh conditions that compromise functional group tolerance. This patent delineates a streamlined approach where isatin or acenaphthylenedione reacts with 5-aminopyrazoles and mercapto carboxylic acids in a single pot. By leveraging acid catalysis at moderate temperatures ranging from 65°C to 95°C, the method achieves high conversion rates while maintaining exceptional purity profiles. For R&D teams focused on high-purity pharmaceutical intermediates, this technology offers a direct route to diverse chemical libraries without the burden of extensive downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in the synthesis of indole-based spiro compounds often relied on methodologies that were operationally cumbersome and economically inefficient. For instance, earlier reports described the use of anhydrous zinc chloride to catalyze the reaction between isatin and o-aminothiophenol, a process that necessitates strict anhydrous conditions and generates heavy metal waste streams. Other approaches involved multi-step sequences where intermediates had to be isolated and purified before proceeding to the next stage, significantly increasing the overall production time and material loss. Furthermore, methods utilizing reflux in toluene or other high-boiling solvents often resulted in difficult product isolation, requiring column chromatography which is notoriously difficult to scale for commercial manufacturing.
The complexity of these traditional routes poses significant risks for supply chain stability. The reliance on specialized reagents and the generation of complex impurity profiles mean that batch-to-batch consistency is hard to guarantee. For procurement managers evaluating cost reduction in pharmaceutical intermediate manufacturing, these legacy methods represent a liability due to high solvent consumption, expensive purification media, and prolonged cycle times. The need for transition metal catalysts also introduces regulatory hurdles regarding residual metal limits in final API products, adding another layer of quality control complexity that slows down time-to-market for new drug candidates.
The Novel Approach
In stark contrast, the methodology described in CN102584860A utilizes a green chemistry-inspired multicomponent reaction that converges three distinct building blocks into the final spiro-cycle in a single operation. The reaction employs readily available starting materials—isatin derivatives, 5-aminopyrazoles, and mercaptoacetic or 2-mercaptopropionic acid—dissolved in common solvents like acetonitrile or ethanol. The use of p-toluenesulfonic acid (PTSA) as a catalyst eliminates the need for toxic heavy metals, simplifying the environmental compliance profile of the process. The reaction proceeds smoothly at temperatures between 65°C and 95°C, typically reaching completion within 8 to 24 hours as monitored by thin-layer chromatography (TLC).
The most compelling advantage of this novel approach is the drastic simplification of the workup procedure. Unlike previous methods requiring chromatographic purification, the products in this invention often precipitate directly from the reaction mixture or can be isolated simply by evaporating the solvent and washing the residue with anhydrous ethanol. This "filter-and-dry" capability is a game-changer for commercial scale-up of complex pharmaceutical intermediates. It reduces the dependency on specialized equipment and skilled labor for purification, thereby lowering the barrier to entry for large-scale production. The patent reports yields consistently exceeding 70%, with specific examples achieving up to 89%, demonstrating both high efficiency and robustness across a variety of substrate substitutions.
Mechanistic Insights into Acid-Catalyzed Multicomponent Cyclization
The formation of the dihydro-spiro[indole-3,4'-pyrazolo[3,4-e][1,4]thiazepine] core proceeds through a concerted cascade mechanism facilitated by Brønsted acid catalysis. Initially, the acid catalyst activates the carbonyl group of the isatin or acenaphthylenedione, enhancing its electrophilicity towards nucleophilic attack by the amino group of the 5-aminopyrazole. This condensation step forms an imine or enamine intermediate, which is crucial for setting the stereochemistry at the spiro-center. Subsequently, the thiol group of the mercapto carboxylic acid attacks the activated carbonyl or the imine carbon, initiating the ring-closing sequence that constructs the seven-membered thiazepine ring fused to the pyrazole moiety.
This mechanistic pathway is highly sensitive to the electronic nature of the substituents on the aromatic rings, yet the protocol demonstrates remarkable tolerance. Whether the isatin bears electron-withdrawing groups like fluorine or bromine, or electron-donating groups like methyl, the reaction proceeds efficiently. This suggests that the rate-determining step is likely the initial condensation or the final cyclization, both of which are sufficiently accelerated by the acidic environment. Understanding this mechanism allows chemists to fine-tune reaction parameters, such as catalyst loading (optimized at a molar ratio of 1:0.3 relative to the aminopyrazole), to maximize throughput. The ability to predictably manage impurity formation through controlled acid catalysis ensures that the final high-purity pharmaceutical intermediates meet stringent quality specifications without extensive recrystallization.
How to Synthesize Spiro-Heterocyclic Indole Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to stoichiometry and thermal management to ensure optimal yields. The process is designed to be user-friendly, utilizing standard glassware and heating mantles commonly found in chemical facilities. The key to success lies in the precise mixing of the three components—isatin derivative, aminopyrazole, and mercapto acid—followed by the addition of the catalyst and solvent. Maintaining a steady reflux temperature is critical to drive the equilibrium towards the spiro-product while minimizing side reactions. Detailed standard operating procedures for this synthesis are outlined below to guide technical teams in replicating the high yields reported in the patent literature.
- Dissolve isatin or acenaphthylenedione, a 5-aminopyrazole compound, and mercapto carboxylic acid in a suitable solvent such as acetonitrile or ethanol.
- Add an organic acid catalyst, preferably p-toluenesulfonic acid, to the reaction mixture to initiate the cyclization process.
- Heat the mixture to reflux at temperatures between 65°C and 95°C for 8 to 24 hours, monitoring progress via TLC until completion.
- Upon completion, evaporate the solvent, wash the resulting solid with anhydrous ethanol, and filter to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations focused on supply chain resilience and cost efficiency, the adoption of this synthetic route offers tangible strategic benefits. The elimination of transition metal catalysts removes the need for expensive scavenging resins and complex analytical testing for heavy metal residues, which directly translates to lower operational expenditures. Furthermore, the use of commodity solvents like ethanol and acetonitrile ensures that raw material sourcing is stable and unaffected by geopolitical fluctuations that often impact specialty solvents. The simplicity of the isolation process means that production cycles are significantly shortened, allowing for faster turnover of manufacturing suites and increased overall capacity utilization without capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of chromatographic purification steps. In traditional fine chemical synthesis, chromatography can account for a substantial portion of the total manufacturing cost due to silica gel consumption and solvent volume. By enabling isolation via simple filtration and washing, this method drastically reduces solvent waste disposal costs and consumable expenses. Additionally, the high atom economy of the multicomponent reaction ensures that a larger proportion of the starting mass ends up in the final product, minimizing raw material waste and improving the overall cost-per-kilogram metric for the intermediate.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis—isatins, aminopyrazoles, and mercapto acids—are bulk chemicals produced by numerous global suppliers. This diversity in the supply base mitigates the risk of single-source dependency, ensuring that production schedules are not disrupted by vendor shortages. The robustness of the reaction conditions (65-95°C) also means that the process can be transferred between different manufacturing sites with minimal re-validation effort. This flexibility is crucial for reducing lead time for high-purity pharmaceutical intermediates, as it allows for dynamic allocation of production capacity across a global network of contract manufacturing organizations.
- Scalability and Environmental Compliance: Scaling a chemical process often introduces heat transfer and mixing challenges that can alter product quality. However, the exothermic profile of this reaction is manageable under reflux conditions, making it inherently safer and easier to scale from gram to ton quantities. The absence of hazardous reagents and the use of recyclable solvents align with modern green chemistry principles, facilitating easier permitting and environmental compliance. This sustainability angle is increasingly important for pharmaceutical companies aiming to reduce their carbon footprint, making this technology a preferred choice for long-term supply agreements and strategic partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this spiro-heterocyclic synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production pipelines or R&D programs.
Q: What catalysts are suitable for this spiro-heterocyclic synthesis?
A: The patent specifies the use of organic or mineral acids as catalysts. Preferred embodiments utilize p-toluenesulfonic acid (PTSA) due to its solubility and effectiveness in promoting the multicomponent condensation without requiring harsh transition metals.
Q: What yields can be expected from this preparation method?
A: Experimental data within the patent indicates that the reaction consistently achieves yields greater than 70%. Specific examples demonstrate yields ranging from 74% to 89%, depending on the specific substituents on the isatin and pyrazole rings.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is highly scalable. It utilizes standard reflux conditions (65-95°C) and common solvents like ethanol or acetonitrile. The workup involves simple filtration and washing, eliminating the need for complex chromatographic purification which is often a bottleneck in scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro-Heterocyclic Compound Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating innovative academic research into commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped to handle the specific requirements of acid-catalyzed multicomponent reactions, featuring rigorous QC labs that enforce stringent purity specifications for every batch released. We understand that the integrity of your drug development pipeline depends on the quality of your intermediates, which is why we prioritize process robustness and impurity control in every project we undertake.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next-generation therapeutic programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with NINGBO INNO PHARMCHEM, you gain access to deep process expertise and a reliable supply chain capable of supporting your growth from clinical trials to full-scale commercialization. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for these valuable spiro-heterocyclic scaffolds.
