Advanced Fmoc-Based Synthesis Strategy for High-Purity Lefenacin Intermediates
The pharmaceutical industry is constantly seeking robust, scalable, and safe synthetic routes for complex active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is detailed in patent CN114573500A, which discloses a novel preparation method for a key lefenacin intermediate. Lefenacin, a long-acting muscarinic antagonist (LAMA), is critical for the maintenance treatment of Chronic Obstructive Pulmonary Disease (COPD). The traditional synthetic pathways for this molecule have long been plagued by safety hazards associated with high-pressure hydrogenation and the use of toxic reagents. This new methodology fundamentally shifts the paradigm by employing Fmoc-Cl (9-fluorenylmethoxycarbonyl chloride) as an amino protecting group, thereby circumventing the need for hazardous Pd/C hydrogenation steps. For R&D directors and process chemists, this represents a pivotal opportunity to enhance purity profiles while simultaneously addressing the stringent safety protocols required in modern GMP manufacturing environments.
From a supply chain perspective, the adoption of this technology signals a move towards more resilient manufacturing processes. The reliance on bulk, commercially available reagents such as sodium triacetoxyborohydride and morpholine, combined with the elimination of specialized high-pressure equipment, drastically simplifies the production landscape. This report analyzes the technical nuances of patent CN114573500A, providing a comprehensive evaluation of its mechanistic advantages and its potential to drive substantial cost reduction in API manufacturing. By understanding the specific solvent optimizations and stoichiometric controls detailed in the patent, procurement managers can better assess the feasibility of scaling this route for commercial production, ensuring a steady supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN114573500A, the synthesis of lefenacin intermediates predominantly relied on routes that introduced significant operational risks and inefficiencies. The original patent CN1930125A and subsequent improvements like U.S. Pat. No. 2012/0016130A1 utilized biphenyl-2-isocyanate and 4-hydroxy-N-benzyl piperidine as starting materials. A critical bottleneck in these legacy processes was the necessity of removing benzyl or benzyloxycarbonyl protecting groups via high-pressure hydrogenation using palladium on carbon (Pd/C) catalysts. This step is inherently dangerous due to the involvement of hydrogen gas under pressure, classifying it as a high-risk reaction that requires specialized infrastructure and rigorous safety monitoring. Furthermore, the use of heterogeneous metal catalysts introduces the persistent challenge of residual metal removal, necessitating additional purification steps that erode overall yield and increase production costs. The overall molar yields for these conventional routes hover around 40% to 45%, indicating substantial material loss and inefficiency that is unacceptable for large-scale commercial operations.
The Novel Approach
The methodology presented in CN114573500A offers a transformative solution by replacing the hazardous hydrogenation steps with a mild, homogeneous chemical sequence centered around Fmoc chemistry. As illustrated in the reaction scheme below, the process initiates with the protection of methylamino acetaldehyde dimethyl acetal using Fmoc-Cl, followed by hydrolysis, reductive amination, and finally, deprotection using morpholine. This approach completely avoids the use of high-pressure hydrogenation, rendering the process significantly safer and more amenable to standard reactor setups found in most fine chemical facilities. The substitution of the benzyl group with the Fmoc group allows for deprotection under mild basic conditions using morpholine, a reagent that is easy to handle and remove. This strategic shift not only mitigates safety risks but also streamlines the downstream processing, as there is no need for complex filtration systems to remove spent metal catalysts. The result is a cleaner reaction profile with improved throughput, positioning this method as a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
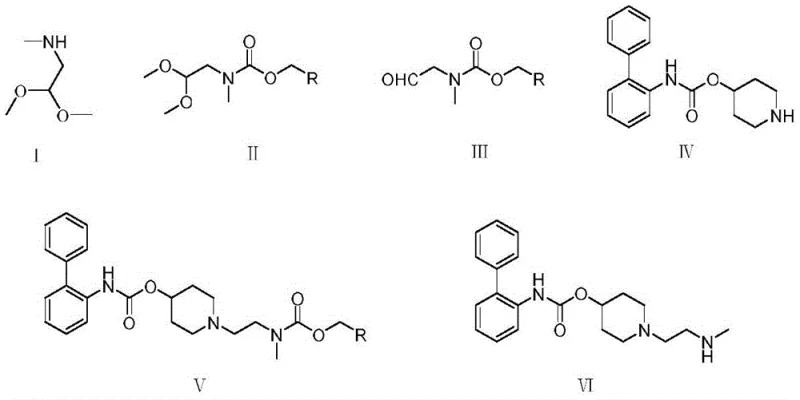
Mechanistic Insights into Fmoc Protection and Reductive Amination
The success of this synthetic route hinges on the precise control of reaction conditions, particularly during the protection and reductive amination stages. The initial protection of the secondary amine in methylamino acetaldehyde dimethyl acetal with Fmoc-Cl is conducted under alkaline conditions, typically using inorganic bases like sodium carbonate or sodium hydroxide. The choice of solvent here is paramount; experimental data within the patent indicates that 2-methyltetrahydrofuran (2-MeTHF) is vastly superior to methanol, ethyl acetate, or dichloromethane. In 2-MeTHF, the reaction proceeds to completion within 5 hours at ambient temperature, whereas other solvents leave unreacted starting materials. This suggests that 2-MeTHF provides an optimal polarity environment that stabilizes the transition state for the nucleophilic attack of the amine on the chloroformate, while effectively solubilizing both the organic substrate and the inorganic base interface. Following protection, the acetal is hydrolyzed using a specific concentration of hydrochloric acid (preferably 3 mol/L) to reveal the reactive aldehyde functionality in Formula III, which is essential for the subsequent coupling step.
The core bond-forming event occurs in the third step, a reductive amination between the aldehyde (Formula III) and the piperidine carbamate (Formula IV). This reaction utilizes sodium triacetoxyborohydride as the reducing agent, a choice that offers distinct advantages over sodium cyanoborohydride in terms of safety and selectivity. The mechanism involves the initial formation of an iminium ion intermediate between the amine and the aldehyde, which is then selectively reduced by the hydride source. The patent highlights the critical importance of stoichiometry here; using 1.2 equivalents of sodium triacetoxyborohydride ensures complete conversion without excessive waste. Furthermore, the use of tetrahydrofuran (THF) as the solvent for this step is crucial, as it minimizes the formation of impurities compared to ethyl acetate. The final deprotection step leverages the unique susceptibility of the Fmoc group to nucleophilic attack by secondary amines like morpholine. This mechanism proceeds via an E1cB-like elimination pathway, releasing the free amine (Formula VI) and a dibenzofulvene-morpholine adduct, which is easily separated during workup, ensuring the high purity required for pharmaceutical applications.
How to Synthesize Lefenacin Intermediate Efficiently
The synthesis of the lefenacin intermediate described in patent CN114573500A is a meticulously optimized four-step process designed for maximum efficiency and safety. To achieve the reported yields and purity levels, strict adherence to the specified solvent systems and reagent ratios is essential. The process begins with the Fmoc protection in 2-methyltetrahydrofuran, followed by acid hydrolysis to generate the key aldehyde intermediate. The subsequent reductive amination couples this aldehyde with the biphenyl-piperidine fragment, and the sequence concludes with a mild morpholine-mediated deprotection. Each step has been parameterized to avoid common pitfalls such as incomplete reactions or impurity buildup. For process chemists looking to implement this route, the following standardized guide outlines the critical operational parameters derived directly from the patent examples.
- Perform protection reaction on methylamino acetaldehyde dimethyl acetal with Fmoc-Cl in 2-methyltetrahydrofuran under alkaline conditions to obtain the protected intermediate.
- Carry out hydrolysis reaction on the protected compound using a hydrochloric acid solution to generate the aldehyde intermediate (Formula III).
- Execute reductive amination between the aldehyde intermediate, piperidine-4-yl [1,1-biphenyl]-2-carbamate, and sodium triacetoxyborohydride in tetrahydrofuran.
- Conduct deprotection reaction on the resulting amine using morpholine in tetrahydrofuran to yield the final lefenacin intermediate (Formula VI).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the technical improvements outlined in this patent translate directly into tangible business benefits, primarily driven by risk mitigation and process simplification. The elimination of high-pressure hydrogenation is perhaps the most significant advantage, as it removes the requirement for specialized, high-CAPEX reactor vessels and the associated safety infrastructure. This reduction in capital expenditure allows for more flexible manufacturing options and lowers the barrier to entry for production. Additionally, the avoidance of palladium catalysts eliminates the costly and time-consuming steps associated with metal scavenging and residual metal testing, which are critical quality control checkpoints in API manufacturing. By streamlining these unit operations, the overall production cycle time is effectively shortened, enhancing the responsiveness of the supply chain to market demands.
- Cost Reduction in Manufacturing: The shift to Fmoc chemistry significantly lowers operational costs by removing the need for expensive noble metal catalysts and high-pressure equipment. The reagents used, such as Fmoc-Cl and sodium triacetoxyborohydride, are bulk commodities with stable supply chains, ensuring consistent pricing and availability. Furthermore, the higher efficiency of the reaction sequence reduces the consumption of raw materials per kilogram of product, driving down the variable cost of goods sold. The simplified workup procedures, which do not require complex filtration or metal removal resins, also contribute to substantial cost savings in terms of labor and consumables.
- Enhanced Supply Chain Reliability: By relying on widely available organic reagents rather than specialized catalytic systems, the supply chain becomes more robust against disruptions. The solvents identified as optimal, such as 2-methyltetrahydrofuran and tetrahydrofuran, are standard industrial chemicals with multiple global suppliers, reducing the risk of single-source dependency. The mild reaction conditions (ambient temperature and pressure) also mean that the process can be transferred between different manufacturing sites with greater ease, ensuring continuity of supply even if one facility faces operational challenges. This flexibility is crucial for maintaining the uninterrupted production of critical respiratory medications.
- Scalability and Environmental Compliance: The process is inherently greener and more scalable due to the absence of heavy metals and high-energy inputs. The waste stream is simpler to treat, as it lacks toxic metal residues, aligning with increasingly stringent environmental regulations. The use of morpholine for deprotection generates byproducts that are easier to manage compared to the waste from hydrogenation processes. This environmental compatibility facilitates smoother regulatory approvals and reduces the long-term liability associated with hazardous waste disposal, making the process sustainable for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These answers are derived from the specific experimental data and optimization studies presented in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this method for their own manufacturing pipelines.
Q: Why is the Fmoc protection strategy superior to traditional benzyl protection for this intermediate?
A: Traditional routes utilize benzyl protection which requires dangerous high-pressure hydrogenation (Pd/C) for removal. The Fmoc strategy described in CN114573500A allows for mild deprotection using morpholine at atmospheric pressure, significantly enhancing operational safety and eliminating the need for expensive high-pressure reactors.
Q: What are the critical solvent choices for maximizing yield in this synthesis?
A: The patent identifies 2-methyltetrahydrofuran as the optimal solvent for the initial Fmoc protection step, ensuring complete reaction where methanol or ethyl acetate fail. Similarly, tetrahydrofuran is critical for the reductive amination and final deprotection steps to minimize impurity formation and ensure reaction completion.
Q: How does this method impact the impurity profile compared to prior art?
A: By avoiding transition metal catalysts like Pd/C, this method eliminates the risk of heavy metal contamination in the final product. Furthermore, the specific optimization of reagent equivalents, such as using 1.2 equivalents of sodium triacetoxyborohydride, ensures complete conversion while minimizing side reactions, leading to a cleaner crude product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lefenacin Intermediate Supplier
The technological advancements detailed in CN114573500A represent a significant leap forward in the synthesis of respiratory disease therapeutics, offering a pathway that is safer, cleaner, and more economically viable than previous methods. NINGBO INNO PHARMCHEM stands at the forefront of translating such innovative laboratory processes into commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this Fmoc-based route are fully realized in a GMP-compliant setting. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required, while our rigorous QC labs guarantee that every batch meets the stringent purity specifications demanded by global regulatory bodies.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis technology for their lefenacin projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this safer route. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project needs. Together, we can accelerate the delivery of high-quality COPD treatments to patients worldwide while optimizing your manufacturing economics.
