Optimizing Lefenacin Intermediate Production: A Safer, Scalable Route for API Manufacturing
Optimizing Lefenacin Intermediate Production: A Safer, Scalable Route for API Manufacturing
The pharmaceutical landscape for Chronic Obstructive Pulmonary Disease (COPD) treatments continues to evolve, demanding robust and scalable supply chains for critical active pharmaceutical ingredients. A pivotal development in this domain is detailed in patent CN112830890A, which discloses a novel preparation method for Lefenacin (also known as Rifenacin) and its key intermediates. This technical breakthrough addresses long-standing inefficiencies in the synthesis of this long-acting muscarinic antagonist, offering a pathway that significantly enhances process safety and product quality. For global procurement and R&D teams, understanding the nuances of this synthetic route is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting rigorous regulatory standards. The innovation lies not merely in the chemical transformation but in the strategic elimination of hazardous unit operations that have historically plagued the manufacturing of this complex molecule.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Lefenacin has relied on routes that introduce substantial operational risks and environmental burdens, primarily due to the reliance on catalytic hydrogenation. As illustrated in prior art such as CN1930125A, the conventional pathway necessitates the use of palladium on carbon catalysts under high-pressure hydrogen conditions to remove protecting groups. This dependency creates a bottleneck for commercial scale-up of complex pharmaceutical intermediates, as high-pressure hydrogenation requires specialized, expensive equipment and stringent safety protocols to mitigate explosion hazards. Furthermore, the use of heavy metal catalysts introduces the risk of residual palladium contamination in the final API, necessitating costly and time-consuming purification steps to meet ICH guidelines for elemental impurities. The cumulative effect of these factors is a process that is not only energy-intensive but also prone to batch-to-batch variability, complicating the validation process for regulatory filings.
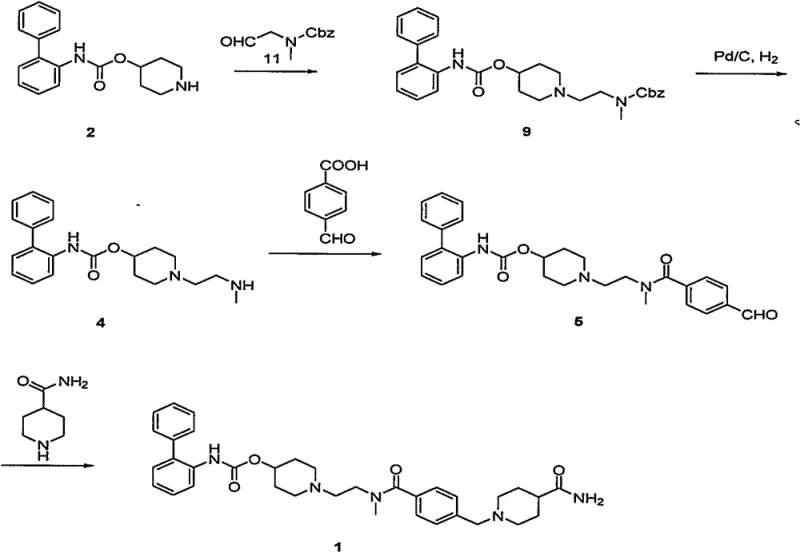
The Novel Approach
In stark contrast, the methodology presented in CN112830890A circumvents these critical vulnerabilities by employing a reductive amination strategy that operates under mild, ambient conditions. This novel approach replaces the hazardous hydrogenation step with a chemoselective reduction using borohydride reagents, which are safer to handle and do not introduce heavy metal contaminants into the reaction matrix. By shifting the paradigm from catalytic hydrogenation to chemical reduction, the process drastically simplifies the reactor requirements, allowing for standard glass-lined or stainless steel vessels to be used without the need for high-pressure ratings. This transition not only enhances the inherent safety of the manufacturing facility but also streamlines the downstream processing, as the removal of inorganic boron salts is significantly more straightforward than scavenging trace palladium from a complex organic mixture. The result is a streamlined workflow that aligns perfectly with the needs of a reliable pharmaceutical intermediate supplier seeking to optimize throughput and safety.
Mechanistic Insights into Reductive Amination and Deprotection
The core of this technological advancement rests on the precise execution of reductive amination between piperidine-4-yl [1,1-biphenyl]-2-carbamate and methyl (2-oxyethyl) tert-butyl carbamate. This transformation is critical for constructing the central amine linkage while maintaining the integrity of the sensitive carbamate protecting groups elsewhere in the molecule. The reaction mechanism involves the initial formation of an iminium ion intermediate, which is subsequently reduced in situ by agents such as sodium triacetoxyborohydride or sodium cyanoborohydride. The choice of reducing agent is paramount, as it must be sufficiently potent to drive the reduction to completion while remaining selective enough to avoid reducing other functional groups, such as the ester or amide moieties present in the scaffold. This selectivity ensures that the reaction yield remains high, often exceeding 80%, while minimizing the formation of side products that could complicate the impurity profile of the final drug substance.
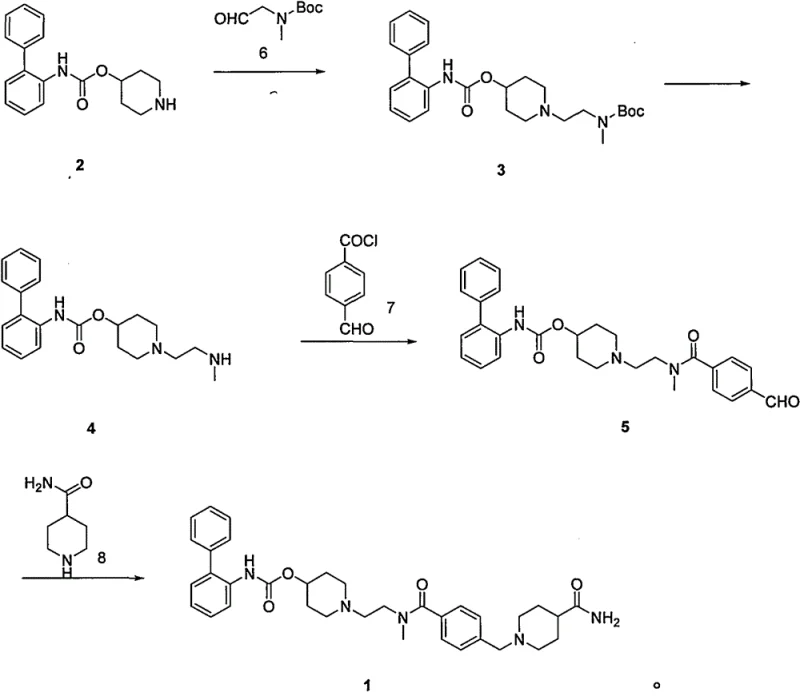
Following the construction of the intermediate scaffold, the process employs a controlled acid-mediated deprotection strategy to reveal the reactive amine necessary for the final coupling steps. This step is meticulously optimized to occur at low temperatures, typically between 0 to 80°C, using acids like trifluoroacetic acid or hydrochloric acid gas. The mildness of these conditions is crucial for preventing the racemization or degradation of the chiral centers and sensitive biphenyl structures within the molecule. By avoiding harsh thermal conditions or strong nucleophiles during deprotection, the process maintains a high degree of stereochemical integrity and chemical purity. This attention to mechanistic detail ensures that the resulting high-purity Lefenacin intermediate meets the stringent specifications required for subsequent API synthesis, reducing the burden on quality control laboratories to screen for complex degradation products.
How to Synthesize Lefenacin Intermediate Efficiently
The implementation of this synthesis route requires a disciplined approach to reaction monitoring and workup procedures to fully realize its efficiency benefits. The process begins with the careful stoichiometric balancing of the amine and aldehyde precursors, followed by the controlled addition of the reducing agent to manage exotherms. Detailed standard operating procedures are essential to ensure that the reaction kinetics are managed effectively, preventing the accumulation of unstable intermediates. While the specific laboratory protocols are extensive, the general workflow emphasizes simplicity and robustness, making it highly transferable from pilot scale to full commercial production. For a comprehensive breakdown of the specific reagent quantities, solvent choices, and isolation techniques, please refer to the standardized guide below.
- Perform reductive amination of piperidine-4-yl [1,1-biphenyl]-2-carbamate with methyl (2-oxyethyl) tert-butyl carbamate using a borohydride reducing agent.
- Execute acid-mediated deprotection of the tert-butyl carbamate group to reveal the secondary amine functionality.
- Conduct acylation with p-formyl benzoyl chloride followed by final reductive amination with isopiperidinecarboxamide to yield Lefenacin.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this novel synthetic route offers profound advantages that extend beyond mere chemical elegance. For procurement managers focused on cost reduction in API manufacturing, the elimination of noble metal catalysts represents a direct saving on raw material costs, as palladium and other precious metals are subject to significant market volatility. Moreover, the removal of the hydrogenation step reduces the capital expenditure required for facility upgrades, as existing infrastructure can often be utilized without modification. This flexibility allows suppliers to respond more agilely to market demand fluctuations, ensuring a more stable supply of high-purity OLED material or pharmaceutical intermediates without the lead times associated with specialized equipment procurement. The overall effect is a more resilient supply chain that is less susceptible to disruptions caused by equipment maintenance or safety incidents.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of expensive catalytic systems with commodity chemical reagents. By avoiding the use of palladium on carbon, manufacturers eliminate the need for costly metal scavenging resins and the associated validation testing for residual metals. This simplification of the purification train reduces solvent consumption and waste disposal costs, contributing to substantial cost savings over the lifecycle of the product. Additionally, the higher yields achieved through this selective chemistry mean that less raw material is wasted, further enhancing the overall cost-efficiency of the production campaign.
- Enhanced Supply Chain Reliability: The operational simplicity of the new route directly translates to improved reliability in meeting delivery schedules. Without the need for high-pressure hydrogenation, the risk of unplanned downtime due to equipment failure or safety shutdowns is significantly minimized. The use of commercially available reagents ensures that raw material sourcing is not a bottleneck, allowing for consistent production runs. This stability is critical for reducing lead time for high-purity pharmaceutical intermediates, enabling downstream API manufacturers to plan their production schedules with greater confidence and accuracy.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial quantities is facilitated by the mild reaction conditions and the absence of hazardous gases. The reduced environmental footprint, characterized by lower energy consumption and the elimination of heavy metal waste streams, aligns with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden on the manufacturer and minimizes the risk of production halts due to environmental permitting issues. Consequently, the process supports sustainable manufacturing practices while maintaining the high throughput required for commercial success.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis method, we have compiled a set of answers based on the patent data and our internal process knowledge. These responses are designed to clarify the feasibility of the route and its alignment with industry standards for quality and safety. Understanding these details is vital for stakeholders evaluating the potential for technology transfer or long-term supply agreements. The following section addresses specific concerns regarding safety, purity, and scalability that are often raised during the vendor qualification process.
Q: How does the new synthesis route improve safety compared to prior art?
A: The novel route eliminates the use of palladium on carbon and high-pressure hydrogenation, removing significant explosion risks and heavy metal contamination concerns associated with conventional methods.
Q: What is the expected purity profile of the intermediate?
A: The optimized process conditions allow for HPLC purity exceeding 99%, meeting stringent pharmaceutical grade specifications without complex purification steps.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the mild reaction conditions, ambient temperature operations, and simplified workup procedures make this route highly adaptable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lefenacin Intermediate Supplier
The technical potential of the synthesis route described in CN112830890A is best realized through partnership with an experienced CDMO capable of navigating the complexities of fine chemical manufacturing. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to product is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Lefenacin intermediate meets the exacting standards required for global pharmaceutical markets. We understand that the consistency of your supply chain is paramount, and our robust quality management systems are designed to provide the transparency and reliability you need.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this safer, more efficient process. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics. Let us collaborate to enhance your supply chain resilience and drive innovation in your drug development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
