Advanced Manufacturing of Tianeptine Sodium Intermediate via Fixed-Bed Oxidation Technology
Advanced Manufacturing of Tianeptine Sodium Intermediate via Fixed-Bed Oxidation Technology
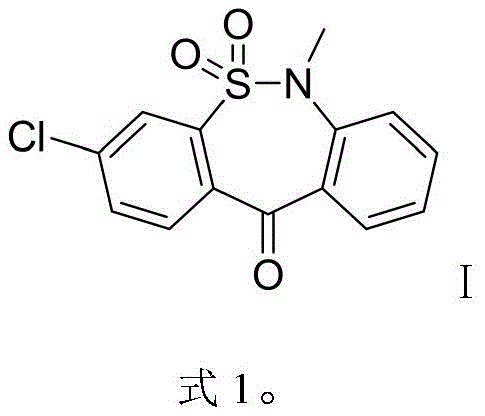
The pharmaceutical landscape for antidepressant therapies continues to evolve, with Tianeptine Sodium standing out as a unique atypical agent offering superior tolerability and efficacy profiles compared to traditional tricyclic antidepressants. Central to the secure supply of this vital medication is the efficient production of its key precursor, 3-chloro-6-methyl-dibenzo[c,f][1,2]thiazepin-11(6H)-one-5,5-dioxide (CAS 26638-53-9). A groundbreaking preparation method disclosed in patent CN110172045B introduces a paradigm shift in how this critical intermediate is manufactured, moving away from hazardous batch processes toward a greener, continuous-flow methodology. This technical insight report analyzes the strategic advantages of this novel route, highlighting its potential to redefine cost structures and supply chain reliability for global API producers seeking a reliable pharmaceutical intermediate supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this tricyclic sulfonamide intermediate has been plagued by significant environmental and safety hurdles that complicate large-scale manufacturing. Traditional routes, such as the one depicted in Formula 2, rely heavily on diazotization reactions which generate substantial volumes of nitrogenous wastewater, creating a severe burden on effluent treatment facilities. Furthermore, the critical cyclization step in these legacy processes typically employs aluminum chloride (AlCl3) as a Lewis acid catalyst; while effective, this reagent produces massive quantities of aluminum salt waste upon hydrolysis, necessitating complex and costly disposal protocols. Alternative pathways, illustrated in Formula 3, attempt to circumvent some issues but introduce new risks, such as the use of sodium hydride, a pyrophoric reagent that poses acute safety hazards in industrial settings, and polyphosphoric acid, which is highly corrosive and difficult to handle, leading to equipment degradation and further waste management challenges.
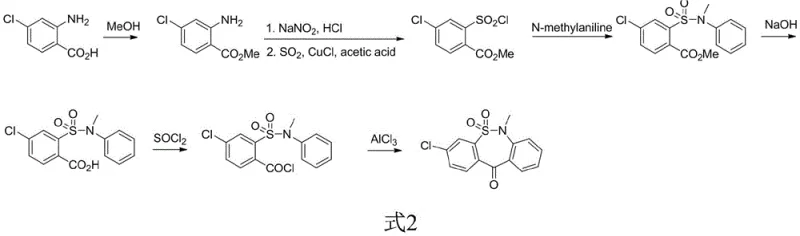
The Novel Approach
In stark contrast to these cumbersome legacy methods, the innovative process outlined in CN110172045B leverages p-chlorotoluene as a low-cost, readily available starting material to construct the molecular framework with remarkable efficiency. As shown in the reaction scheme below, the new route bypasses the need for diazotization entirely, instead utilizing a direct chlorosulfonation followed by amidation to establish the core sulfonamide linkage. The true breakthrough lies in the oxidation step, where a fixed-bed reactor loaded with a solid-supported catalyst facilitates the conversion of the methyl group to a carboxylic acid using air as the oxidant. This continuous flow approach not only enhances heat and mass transfer but also eliminates the stoichiometric waste associated with liquid-phase oxidants, culminating in a final cyclization driven by a reusable solid acid catalyst that can be easily separated and recycled.
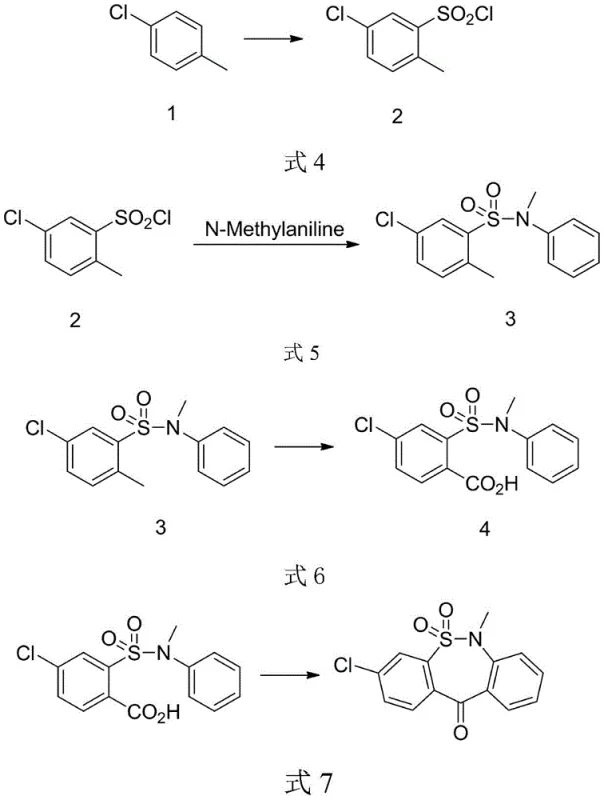
Mechanistic Insights into Fixed-Bed Catalytic Oxidation and Solid Acid Cyclization
The core technological advancement in this patent is the implementation of heterogeneous catalysis within a fixed-bed reactor system for the oxidation of the sulfonamide intermediate. In this mechanism, the substrate is vaporized or suspended and passed over a solid catalyst bed—typically comprising vanadium, molybdenum, or other transition metal oxides supported on alumina or titania—at elevated temperatures around 220°C. The use of air as the terminal oxidant is thermodynamically favorable and atom-economical, producing water as the only byproduct, which stands in sharp contrast to the heavy metal sludge generated by permanganate or chromate oxidations. The fixed-bed configuration ensures that the catalyst remains stationary while the reactants flow through, allowing for precise residence time control and preventing the catalyst from contaminating the product stream, thereby simplifying downstream purification and ensuring high purity specifications for the resulting carboxylic acid.
Following the oxidation, the intramolecular Friedel-Crafts acylation is achieved using a solid acid catalyst, such as a perfluorosulfonic acid resin, rather than traditional homogeneous Lewis acids. This mechanistic shift is crucial for impurity control; homogeneous acids often lead to over-reaction, polymerization, or difficult-to-remove metal complexes that require extensive washing and chelation steps. The solid acid provides strong Brønsted acidity sufficient to activate the carboxylic acid for cyclization but allows for a simple filtration workup where the catalyst is retained on the filter cake. This physical separation capability means the catalyst can be regenerated and reused in subsequent batches, maintaining consistent catalytic activity over time and ensuring that the final product is free from metallic impurities that could compromise the safety profile of the final API.
How to Synthesize 3-chloro-6-methyl-dibenzo[c,f][1,2]thiazepin-11(6H)-one-5,5-dioxide Efficiently
The execution of this synthesis requires careful attention to the continuous flow parameters and the selection of appropriate solid supports to maximize yield and throughput. The process begins with the chlorosulfonation of p-chlorotoluene, followed by amidation with N-methylaniline in the presence of an organic base like triethylamine or pyridine to scavenge HCl. The resulting sulfonamide is then subjected to the critical gas-phase or liquid-phase oxidation in the fixed-bed reactor, where temperature and flow rate must be optimized to prevent over-oxidation or decomposition. Finally, the carboxylic acid intermediate undergoes dehydration cyclization in a high-boiling solvent like dichlorobenzene or xylene using the solid acid resin. For detailed operational parameters, stoichiometry, and specific catalyst preparation methods, please refer to the standardized synthesis guide below.
- React p-chlorotoluene with chlorosulfonic acid to generate the sulfonyl chloride intermediate.
- Condense the sulfonyl chloride with N-methylaniline under alkaline conditions to form the sulfonamide.
- Oxidize the methyl group to a carboxylic acid using air in a fixed-bed reactor with a solid-supported catalyst.
- Perform intramolecular cyclization using a reusable solid acid catalyst to obtain the final tricyclic intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers a compelling value proposition centered on risk mitigation and long-term cost optimization. By transitioning from a multi-step batch process involving hazardous reagents to a streamlined continuous flow operation, manufacturers can significantly reduce the complexity of their supply chains. The reliance on commodity chemicals like p-chlorotoluene and air, rather than specialized and volatile reagents like sodium hydride or thionyl chloride in later stages, insulates the production process from raw material price volatility and availability shocks. Furthermore, the reduction in unit operations—from esterification, hydrolysis, and chlorination steps in older routes to a direct oxidation—shortens the overall production cycle time, enabling faster response to market demand fluctuations and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the elimination of expensive stoichiometric reagents and the drastic reduction in waste disposal costs. By replacing aluminum chloride and polyphosphoric acid with reusable solid catalysts, the process removes the need for costly neutralization and heavy metal removal steps, which traditionally account for a significant portion of manufacturing overhead. Additionally, the ability to recycle the solid acid catalyst multiple times without significant loss of activity translates to a lower cost of goods sold (COGS) per kilogram, providing a competitive pricing advantage in the global marketplace for antidepressant ingredients.
- Enhanced Supply Chain Reliability: The robustness of the fixed-bed oxidation process ensures a more stable and predictable output, which is critical for maintaining uninterrupted API supply. Unlike batch reactions that are susceptible to variability in mixing and heat transfer, continuous flow systems offer superior reproducibility, minimizing the risk of batch failures that can disrupt inventory levels. The use of air as an oxidant also removes the logistical burden of transporting and storing hazardous liquid oxidants, simplifying site operations and reducing the regulatory compliance burden associated with dangerous goods handling and storage.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this process represents a significant leap forward in green chemistry, aligning with increasingly stringent global environmental regulations. The near-elimination of heavy metal waste and acidic wastewater simplifies the permitting process for new manufacturing lines and reduces the liability associated with environmental remediation. The inherent safety of removing pyrophoric reagents like sodium hydride from the workflow further protects the workforce and facility, making the commercial scale-up of complex pharmaceutical intermediates safer and more sustainable for long-term operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and comparative analysis of prior art. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the strategic fit of this intermediate within their portfolio. These insights clarify how the specific mechanistic improvements translate into tangible operational benefits.
Q: How does the fixed-bed oxidation process improve environmental compliance compared to traditional methods?
A: The fixed-bed oxidation replaces hazardous stoichiometric oxidants and eliminates the heavy metal waste associated with aluminum chloride cyclization found in older routes. By utilizing air as the oxygen source and a reusable solid catalyst, the process drastically reduces wastewater generation and avoids the disposal issues of aluminum salts.
Q: What are the primary cost drivers reduced in this new synthesis pathway?
A: The primary cost reductions stem from the use of inexpensive bulk raw materials like p-chlorotoluene instead of complex amino-benzoic acids, and the elimination of expensive reagents like sodium hydride and polyphosphoric acid. Additionally, the ability to recycle the solid acid catalyst multiple times significantly lowers the operational expenditure per kilogram of product.
Q: Is this synthesis route scalable for industrial production of antidepressant intermediates?
A: Yes, the process is specifically designed for industrial scalability. The use of continuous flow technology in the fixed-bed reactor allows for precise control of reaction parameters and heat management, which are critical challenges in batch processing. This ensures consistent product quality and stable supply for large-scale API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tianeptine Sodium Intermediate Supplier
As the global demand for mental health treatments continues to rise, securing a stable supply of high-quality intermediates is paramount for pharmaceutical developers. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in process chemistry to bridge the gap between laboratory innovation and industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Tianeptine Sodium Intermediate adheres to the highest international standards required for API synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing footprint. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can drive value and security for your antidepressant drug development programs.
