Advanced Synthesis of Tianeptine Sodium Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for antidepressant agents, particularly for novel compounds like tianeptine sodium, which offers a unique mechanism of action distinct from traditional monoamine-based therapies. A recent technological breakthrough documented in patent CN115872951A introduces a highly efficient synthesis method for a critical methoxy-substituted intermediate, specifically 3-chloro-6, 11-dihydro-6-methyl-11-methoxy-dibenzo[c,f][1,2]thiazepine-5,5-dioxide. This innovation addresses long-standing challenges in the production of tianeptine precursors by shifting the paradigm from difficult impurity removal to constructive molecular transformation. By leveraging a base-catalyzed substitution in an alcohol-water medium, this method achieves exceptional purity levels exceeding 99.6% while maintaining mild operational parameters. For global procurement teams and R&D directors, this represents a significant opportunity to optimize the supply chain for high-value psychiatric medications, ensuring both cost efficiency and consistent quality in the final active pharmaceutical ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of tianeptine sodium intermediates has been plagued by intricate purification bottlenecks associated with the precursor 3, 11-dichloro-6, 11-dihydro-6-methyl-dibenzo[c,f][1,2]thiazepine-5,5-dioxide. In traditional workflows, the refining treatment steps required after the initial reaction are notoriously labor-intensive and prone to yield loss, often necessitating cooling to below 5°C followed by multiple washing cycles with dimethyl formamide and cold methanol. The core issue lies in the structural similarity between the desired dichloro product and the methoxy-substituted byproduct, which makes efficient separation via standard crystallization or chromatography extremely difficult and economically draining. Furthermore, strict fine operations are mandatory during soaking and washing; if the temperature rises slightly above 0°C or soaking times are extended, the unwanted methoxy byproduct forms inadvertently, contaminating the batch. Consequently, manufacturers have often been forced to accept lower purity starting materials or incur substantial costs attempting to remove these structurally analogous impurities, creating a fragile link in the supply chain for reliable pharmaceutical intermediates supplier networks.
![Chemical structure of 3,11-dichloro-6,11-dihydro-6-methyl-dibenzo[c,f][1,2]thiazepine-5,5-dioxide starting material](/insights/img/tianeptine-intermediate-synthesis-pharma-supplier-20260306190943-01.png)
The Novel Approach
In stark contrast to the cumbersome legacy processes, the novel approach detailed in the patent ingeniously bypasses the purification dilemma by converting the problematic starting material directly into the target high-purity intermediate. Instead of viewing the methoxy-substituted compound as a troublesome byproduct to be eliminated, this method utilizes it as the primary objective, reacting the dichloro precursor in an alcohol-water solution through base catalysis. This strategic pivot eliminates the need for special reaction equipment and harsh refining conditions, allowing for a streamlined operation that is inherently safer and more environmentally friendly. The process capitalizes on the stability of the methoxy intermediate, which can be stored conveniently and used as a high-quality starting material for subsequent steps when needed. This transformation not only simplifies the operational workflow but also drastically enhances the overall yield and reproducibility of the synthesis, providing a robust foundation for cost reduction in API manufacturing and ensuring a steady flow of materials for downstream drug production.
Mechanistic Insights into Base-Catalyzed Nucleophilic Substitution
The chemical elegance of this synthesis lies in its utilization of a base-catalyzed nucleophilic substitution mechanism within a biphasic-friendly alcohol-water solvent system. The reaction proceeds by treating the 3, 11-dichloro substrate with a base, preferably triethylamine or potassium carbonate, which facilitates the displacement of the chlorine atom at the 11-position with a methoxy group derived from the methanol solvent. The use of a 70-95% alcohol-water solution is critical, as it balances the solubility of the organic substrate with the polarity required to stabilize the transition state and the resulting ionic species. Operating at temperatures between 40-100°C, preferably 50-90°C, provides sufficient thermal energy to overcome the activation barrier for the substitution without degrading the sensitive dibenzothiazepine scaffold. This controlled environment ensures that the reaction kinetics favor the formation of the desired methoxy product while minimizing side reactions that could lead to complex impurity profiles, thereby securing the high-purity [精准的行业名词] required for regulatory compliance.
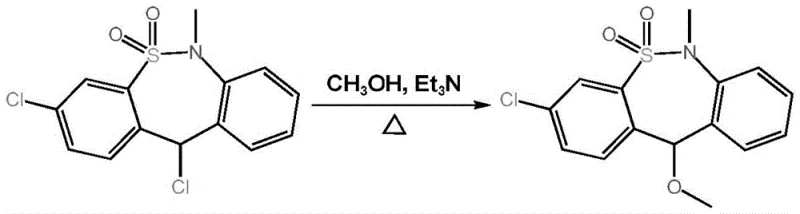
Impurity control in this mechanism is intrinsically managed through the stoichiometry of the base and the precise control of crystallization conditions post-reaction. By maintaining a molar ratio of substrate to base between 1:1.0 and 1:3.0, the system ensures complete consumption of the starting material while preventing excessive basicity that might trigger degradation pathways. Following the heating reaction, the mixture is cooled to a crystallization temperature of -10 to 10°C, preferably -5 to 5°C, which induces the precipitation of the product as a white flocculent solid. This crystallization step acts as a powerful purification tool, effectively excluding soluble impurities and unreacted reagents from the crystal lattice. The result is a product with a liquid phase purity of more than 99.6 percent, demonstrating that the mechanistic design inherently supports the production of high-purity OLED material or pharmaceutical grades without the need for additional chromatographic polishing, thus reinforcing the commercial viability of the route.
How to Synthesize 3-Chloro-6,11-dihydro-6-methyl-11-methoxy-dibenzo[c,f][1,2]thiazepine-5,5-dioxide Efficiently
Implementing this synthesis route requires careful attention to solvent composition and temperature profiling to maximize the benefits of the base-catalyzed transformation. The process begins with the preparation of the reaction mixture, where the dichloro precursor is combined with triethylamine in an 85% methanol aqueous solution, creating the optimal environment for the nucleophilic attack. Operators must monitor the reaction progress, typically via thin-layer chromatography, to ensure full conversion before initiating the cooling phase for crystallization. The detailed standardized synthesis steps outlined below provide a clear roadmap for scaling this technology from laboratory benchtop to pilot plant operations, ensuring consistency across batches. Adhering to these parameters allows manufacturers to replicate the high yields and purity reported in the patent examples, facilitating the commercial scale-up of complex polymer additives or pharmaceutical intermediates with confidence.
- Prepare the reaction mixture by dissolving 3,11-dichloro-6,11-dihydro-6-methyl-dibenzo[c,f][1,2]thiazepine-5,5-dioxide in a 70-95% alcohol-water solution with triethylamine.
- Heat the mixture to 50-90°C and maintain stirring for approximately 4 to 5 hours to ensure complete conversion monitored by TLC.
- Cool the reaction mass to -5 to 5°C to induce crystallization, then filter and dry the resulting white flocculent solid to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic advantages that extend beyond mere technical feasibility. The elimination of complex refining steps and the use of common, inexpensive solvents like methanol and water significantly lower the operational expenditure associated with producing this critical intermediate. By avoiding the need for specialized equipment and hazardous reagents, the process reduces capital investment requirements and minimizes safety risks, leading to substantial cost savings in the overall manufacturing budget. Furthermore, the high reproducibility and mild conditions ensure that production schedules are less likely to be disrupted by technical failures or purification bottlenecks, enhancing supply chain reliability for global partners. This robustness is essential for maintaining continuous supply lines in the volatile pharmaceutical market, where delays can have cascading effects on drug availability.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the workflow and the high yield of the final product. By converting the starting material directly into the desired intermediate with yields reaching up to 96.5%, the method minimizes raw material waste and reduces the volume of solvents required for purification. The absence of expensive transition metal catalysts or complex chromatographic media further drives down the variable costs per kilogram. Additionally, the ability to use lower purity starting materials effectively, as the process tolerates and converts impurities, allows for the sourcing of more cost-effective raw inputs. These factors combine to create a leaner manufacturing model that delivers significant value to the bottom line without compromising on the quality standards expected of a reliable agrochemical intermediate supplier.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the simplicity and robustness of the reaction conditions, which are less susceptible to minor fluctuations in operational parameters. The use of stable, non-hazardous reagents like triethylamine and methanol ensures that raw material sourcing is straightforward and less prone to regulatory restrictions or logistical delays. Moreover, the intermediate product itself is noted for its stable nature and convenient storage, allowing manufacturers to build strategic inventory buffers without concerns about rapid degradation. This stability reduces lead time for high-purity pharmaceutical intermediates, enabling faster response to market demand spikes and ensuring that downstream API production is never starved of critical inputs.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles and industrial safety standards. The reaction generates no waste gas, and the use of an alcohol-water system simplifies wastewater treatment compared to processes relying on chlorinated or aromatic solvents. The straightforward crystallization and filtration steps are easily adaptable to large-scale reactors, supporting the transition from 100 kgs to 100 MT annual commercial production without the need for process re-engineering. This scalability ensures that the technology can meet growing global demand for antidepressants while maintaining a low environmental footprint, a key consideration for companies aiming to meet stringent ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the specific data points and beneficial effects outlined in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the objective capabilities of the method as demonstrated in the experimental examples, offering clarity on purity, yield, and operational requirements. This transparency helps stakeholders make informed decisions about adopting this innovative approach for their specific production needs.
Q: What is the primary advantage of this new synthesis method over conventional refining?
A: The primary advantage is the elimination of complex purification steps for the dichloro starting material. Instead of struggling to remove the methoxy byproduct, this method intentionally converts the starting material directly into the desired high-purity methoxy intermediate, simplifying the workflow and improving overall yield.
Q: What purity levels can be achieved using this base-catalyzed protocol?
A: According to the experimental data in patent CN115872951A, this method consistently achieves liquid phase purity exceeding 99.6%, with specific examples demonstrating purity levels up to 99.72% without requiring extensive chromatographic purification.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It utilizes mild reaction conditions (40-100°C), common solvents like methanol and water, and avoids the need for special reaction equipment or hazardous reagents, making it safe and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloro-6,11-dihydro-6-methyl-11-methoxy-dibenzo[c,f][1,2]thiazepine-5,5-dioxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving medications like tianeptine sodium. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent supply regardless of market fluctuations. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-chloro-6, 11-dihydro-6-methyl-11-methoxy-dibenzo[c,f][1,2]thiazepine-5,5-dioxide meets the highest international standards. We are committed to leveraging advanced synthetic technologies, such as the base-catalyzed route described herein, to deliver superior value and performance to our global partners in the pharmaceutical industry.
We invite you to collaborate with us to explore how this optimized synthesis method can enhance your production efficiency and reduce overall costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how NINGBO INNO PHARMCHEM can become your trusted partner in achieving commercial success.
