Revolutionizing Cefazedone Production: A High-Efficiency Catalytic Route for Commercial Scale-Up
Revolutionizing Cefazedone Production: A High-Efficiency Catalytic Route for Commercial Scale-Up
The pharmaceutical landscape for cephalosporin antibiotics is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable manufacturing processes. A significant breakthrough in this domain is detailed in patent CN110563750B, which discloses a novel synthesis method for Cefazedone, a potent semi-synthetic cephalosporin with broad-spectrum antibacterial activity. This patent introduces a streamlined two-step catalytic route that fundamentally alters the production paradigm, moving away from harsh, multi-step traditional methods towards a more elegant, high-yield process. By leveraging a specific boron trifluoride-dimethyl carbonate complex and a unique boronic acid-promoted coupling strategy, this technology addresses critical pain points in API manufacturing, including impurity control, reaction safety, and overall process economics. For R&D directors and supply chain leaders, understanding this technological shift is paramount for securing a competitive edge in the global antibiotic market.
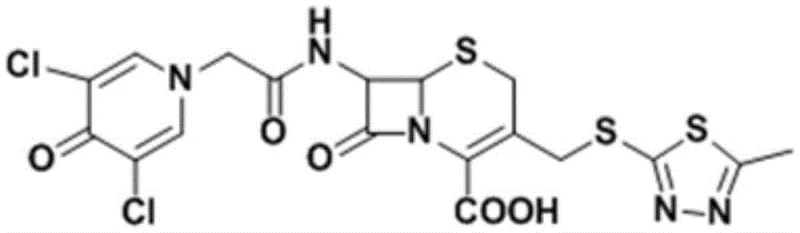
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cefazedone has been plagued by significant technical hurdles that impact both cost and quality. As highlighted in the background of the patent, prior art methods such as those disclosed in DE2345402 rely heavily on the use of Dicyclohexylcarbodiimide (DCC) as a coupling agent. While effective in laboratory settings, DCC coupling generates dicyclohexylurea as a stoichiometric byproduct, which is notoriously difficult to remove completely from the reaction mixture, often requiring complex purification steps that erode overall yield. Furthermore, these traditional routes frequently demand strictly anhydrous conditions and the use of aggressive reagents like trifluoroacetic acid (TFA) for deprotection steps. The necessity for TFA introduces severe corrosion risks to reactor equipment and creates substantial challenges in waste treatment and environmental compliance. Additionally, older methods like US5945414 involve reacting glutaryl 7-ACA at elevated temperatures (90°C) in aqueous solutions, which can lead to thermal degradation of the sensitive beta-lactam ring, resulting in lower purity and increased formation of polymeric impurities that are difficult to separate.
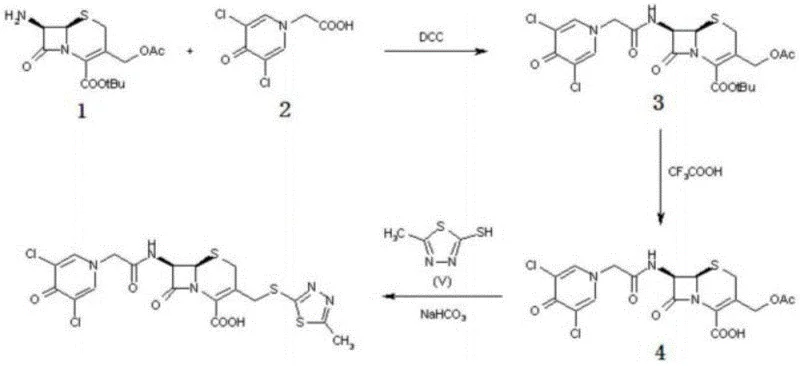
The Novel Approach
In stark contrast to these cumbersome legacy processes, the method described in CN110563750B offers a remarkably simplified and robust alternative. The core innovation lies in the strategic replacement of harsh coupling conditions with a mild, catalytic system. Instead of relying on stoichiometric amounts of expensive and waste-generating coupling agents, the new process utilizes a boron trifluoride-dimethyl carbonate complex to facilitate the initial acylation of 7-ACA. This is followed by a second coupling step promoted by 2-iodophenylboronic acid, which enables the efficient formation of the amide bond between the cephalosporin nucleus and the pyridone side chain. This approach not only eliminates the generation of difficult-to-remove urea byproducts but also allows the reactions to proceed at significantly lower temperatures (0-15°C), thereby preserving the integrity of the thermally sensitive beta-lactam structure. The result is a process that is not only chemically superior in terms of selectivity but also operationally simpler, reducing the burden on downstream purification units.
Mechanistic Insights into Boron-Catalyzed Cephalosporin Functionalization
The success of this synthetic route hinges on the precise mechanistic role of the boron-based catalytic systems employed in both key steps. In the first stage, the boron trifluoride-dimethyl carbonate (BF3-DMC) complex acts as a potent Lewis acid catalyst. It likely coordinates with the carbonyl oxygen of the acylating agent or the hydroxyl group of the substrate, increasing the electrophilicity of the reaction center and facilitating nucleophilic attack by the amino group of 7-ACA under mild conditions. This activation allows the reaction to proceed rapidly at 0-15°C, a temperature range that effectively suppresses competing side reactions such as the hydrolysis of the beta-lactam ring or the rearrangement of the double bond in the dihydrothiazine ring. The use of dimethyl carbonate as the solvent further enhances this effect, providing a polar yet non-nucleophilic environment that stabilizes the transition state while being environmentally benign compared to chlorinated solvents often used in older protocols.
In the second critical step, the introduction of 2-iodophenylboronic acid serves as a sophisticated promoter for the condensation reaction between Compound III and Compound IV. Boronic acids are known to facilitate amide bond formation through the formation of reactive mixed anhydrides or by activating the carboxylic acid moiety via coordination. This specific promoter ensures high regioselectivity and minimizes racemization at the chiral centers of the cephalosporin nucleus, which is crucial for maintaining biological activity. The mechanistic efficiency of this step is evidenced by the high molar yields (up to 93%) and exceptional purity profiles reported in the examples, where the maximum single impurity content is kept below 0.1%. This level of control over the reaction pathway demonstrates a deep understanding of physical organic chemistry, translating directly into a manufacturing process that requires fewer recrystallizations and delivers a product ready for final formulation with minimal additional processing.
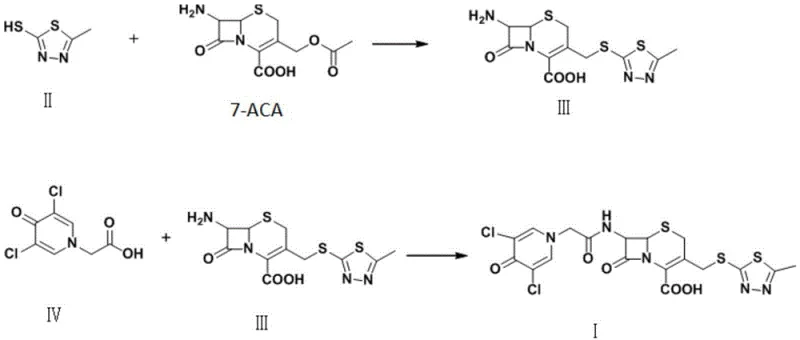
How to Synthesize Cefazedone Efficiently
The implementation of this advanced synthesis route requires careful attention to reaction parameters, particularly temperature control and stoichiometry, to maximize the benefits of the catalytic system. The process begins with the preparation of the key intermediate, Compound III, by dissolving the catalytic system in dimethyl carbonate and adding the thiadiazole thiol derivative. Maintaining the temperature strictly between 0°C and 15°C during the addition of 7-ACA is critical to prevent exothermic runaway and ensure high stereochemical fidelity. Following the isolation of Compound III via pH-controlled crystallization, the final coupling with the pyridone acetic acid derivative is performed in toluene or dichloromethane under reflux conditions. The detailed standardized operating procedures, including specific molar ratios and workup protocols necessary to replicate the high yields described in the patent, are outlined in the technical guide below.
- Prepare Compound III by reacting 5-methyl-1,3,4-thiadiazole-2-thiol with 7-ACA using a boron trifluoride-dimethyl carbonate complex catalyst at 0-15°C.
- Isolate Compound III via pH adjustment and crystallization to achieve >99% purity.
- Couple Compound III with 3,5-dichloro-4-pyridone-1-acetic acid (Compound IV) using 2-iodophenylboronic acid in toluene under reflux to obtain Cefazedone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method represents a strategic opportunity to optimize the cost structure and reliability of the Cefazedone supply chain. The elimination of expensive coupling reagents like DCC and the avoidance of corrosive acids like TFA directly translate into significant raw material cost savings. Furthermore, the simplified workup procedure, which relies on straightforward crystallization rather than complex chromatographic separations or extensive washing to remove urea byproducts, drastically reduces solvent consumption and waste disposal costs. This leaner manufacturing footprint not only lowers the variable cost per kilogram but also shortens the batch cycle time, allowing for increased throughput within existing facility constraints. The robustness of the process against minor variations in reaction conditions also enhances supply continuity by reducing the risk of batch failures.
- Cost Reduction in Manufacturing: The transition to a catalytic protocol fundamentally alters the economic model of production by removing the need for stoichiometric amounts of high-cost activators. By replacing DCC with a catalytic amount of boron complexes, the process eliminates the generation of solid urea waste, which simplifies filtration and reduces the load on wastewater treatment facilities. Additionally, the ability to use common, recyclable solvents like toluene and dimethyl carbonate instead of specialized anhydrous grades further drives down operational expenditures. These cumulative efficiencies result in a substantially lower cost of goods sold (COGS), providing a competitive pricing advantage in the generic antibiotic market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The operational simplicity of this route significantly de-risks the supply chain. Traditional methods requiring strict anhydrous conditions are prone to failure if moisture ingress occurs, leading to batch rejection and supply delays. In contrast, the new method's tolerance for milder conditions and its reliance on stable, commercially available catalysts ensure consistent batch-to-batch performance. The high purity achieved directly from the reactor (>99%) minimizes the need for reprocessing, ensuring that delivery schedules are met reliably. This predictability is invaluable for pharmaceutical companies managing tight inventory levels and Just-In-Time manufacturing schedules for essential medicines.
- Scalability and Environmental Compliance: From a sustainability perspective, this process aligns perfectly with modern green chemistry principles. The reduction in hazardous waste generation, specifically the absence of dicyclohexylurea and the minimized use of halogenated solvents in the first step, simplifies regulatory compliance and environmental permitting. The process is inherently scalable, as demonstrated by the successful execution of the reaction on multi-gram scales in the patent examples without loss of efficiency. This scalability ensures that suppliers can rapidly ramp up production volumes to meet surges in demand, such as during flu seasons or public health emergencies, making it a resilient choice for long-term sourcing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in patent CN110563750B, offering clarity on how this method compares to established industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or for procurement specialists assessing supplier capabilities.
Q: What are the key advantages of the BF3-DMC catalytic system over traditional DCC coupling?
A: The BF3-DMC system operates under mild low-temperature conditions (0-15°C), avoiding the harsh anhydrous requirements and difficult urea byproduct removal associated with Dicyclohexylcarbodiimide (DCC) methods, resulting in significantly higher purity.
Q: How does the new method improve impurity profiles for regulatory compliance?
A: By utilizing 2-iodophenylboronic acid promotion and controlled low-temperature crystallization, the process limits maximum single impurities to below 0.1%, ensuring the final API meets stringent pharmacopoeia standards.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the process eliminates sensitive anhydrous steps and uses common solvents like toluene and dimethyl carbonate, making it highly scalable, safer to operate, and cost-effective for multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefazedone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing processes like the one described in CN110563750B requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO and supplier in the fine chemical sector, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific catalytic requirements of this synthesis, ensuring that every batch of Cefazedone meets stringent purity specifications. With our rigorous QC labs and commitment to continuous process improvement, we guarantee a supply of high-quality antibiotic intermediates that support your regulatory filings and commercial launch timelines.
We invite you to leverage our technical proficiency to optimize your supply chain for Cefazedone and related cephalosporins. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this new catalytic route can improve your margins. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us collaborate to bring this next-generation synthesis technology to your production line, ensuring a reliable and cost-effective supply of this critical antibiotic.
