Advanced Synthesis of Axially Chiral Bissulfoxide Ligands for Asymmetric Catalysis
Advanced Synthesis of Axially Chiral Bissulfoxide Ligands for Asymmetric Catalysis
The landscape of asymmetric synthesis is continually evolving, driven by the demand for highly efficient and selective chiral catalysts. Patent CN102030690A introduces a groundbreaking methodology for the design and synthesis of a novel class of axially chiral bissulfoxide ligands. These specialized organosulfur compounds represent a significant advancement over traditional chiral auxiliaries, offering superior thermal stability and tunable stereoelectronic properties. The core innovation lies in a streamlined two-step synthetic route that begins with readily available optically pure sulfinate esters and aryl halides. This approach not only simplifies the construction of the chiral axis but also facilitates the rapid generation of diverse ligand libraries. For research and development teams focused on high-value pharmaceutical intermediates, this technology provides a robust platform for inducing high enantioselectivity in challenging transformations, particularly in rhodium-catalyzed conjugate additions.
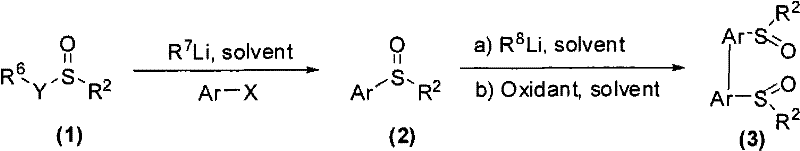
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral ligands has been plagued by complex multi-step sequences that often suffer from poor atom economy and low overall yields. Traditional methods frequently rely on resolution of racemic mixtures, which inherently limits the maximum theoretical yield to fifty percent and generates substantial chemical waste. Furthermore, many existing chiral sulfoxide ligands lack the necessary thermal stability for rigorous industrial processing, leading to potential racemization under elevated reaction temperatures. The structural rigidity required to maintain axial chirality often necessitates bulky substituents that are difficult to introduce selectively without harsh conditions. Consequently, procurement managers and supply chain heads have faced challenges in sourcing these critical reagents at a scale that supports commercial manufacturing, as the legacy processes are neither cost-effective nor easily scalable for large-volume production runs.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this field by employing a direct oxidative coupling strategy that bypasses the need for resolution. By starting with optically pure sulfinate esters, the chirality is introduced early in the sequence via nucleophilic substitution, preserving the optical integrity throughout the synthesis. The subsequent oxidative dimerization, mediated by inexpensive transition metal salts like ferric chloride, efficiently constructs the biaryl axis with high fidelity. This route is exceptionally modular, allowing chemists to easily vary the electronic and steric environment of the ligand by simply changing the starting aryl halide or the sulfinate group. Such flexibility is crucial for fine-tuning catalyst performance across a broad spectrum of substrates, making this approach a superior choice for developing reliable pharmaceutical intermediate supplier capabilities where customization is key.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Coupling
The mechanistic pathway for generating these axially chiral bissulfoxide ligands involves a sophisticated interplay of organolithium chemistry and single-electron transfer oxidation. Initially, the aryl halide undergoes lithium-halogen exchange at cryogenic temperatures to generate a reactive aryllithium species. This nucleophile attacks the sulfur center of the chiral sulfinate ester, displacing the alkoxide and forming the monomeric chiral sulfoxide intermediate with inversion or retention of configuration depending on the specific conditions. The critical step follows, where a strong base such as lithium diisopropylamide (LDA) directs ortho-lithiation adjacent to the sulfoxide group. This directed metalation is essential for positioning the reactive site for the subsequent coupling event.
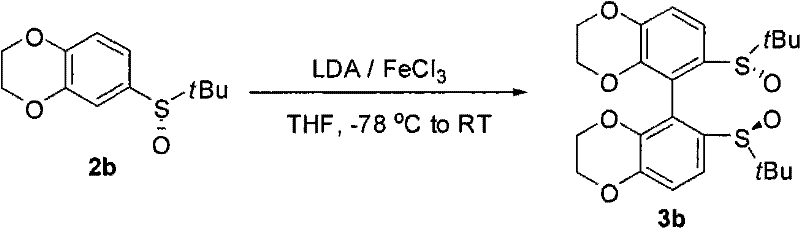
Upon addition of the oxidant, typically a ferric salt like FeCl3 or a cupric salt, the organolithium species undergoes a single-electron transfer process to generate radical intermediates. These radicals rapidly couple to form the new carbon-carbon bond that defines the axial chirality of the final bis-sulfoxide structure. The cone-shaped geometry of the sulfoxide moiety plays a pivotal role in this process, providing significant steric differentiation that favors the formation of one atropisomer over the other. This inherent stereocontrol eliminates the need for downstream purification to remove unwanted enantiomers, thereby streamlining the manufacturing process. For R&D directors, understanding this mechanism highlights the robustness of the impurity profile, as the reaction conditions are mild enough to prevent decomposition of the sensitive sulfoxide functionality while being vigorous enough to ensure complete conversion.
How to Synthesize Axially Chiral Bissulfoxide Ligands Efficiently
The practical execution of this synthesis requires careful control of temperature and stoichiometry to maximize yield and enantiomeric excess. The process begins with the preparation of the chiral sulfoxide monomer, followed by the oxidative coupling step to generate the final ligand. Detailed protocols emphasize the use of anhydrous conditions and inert atmospheres to protect the reactive organolithium intermediates from moisture and oxygen. The following guide outlines the standardized operational procedure derived from the patent examples, ensuring reproducibility and safety in a laboratory or pilot plant setting.
- React aryl halides with organolithium reagents at low temperature, then add optically pure sulfinate esters to form chiral sulfoxide intermediates.
- Treat the chiral sulfoxide intermediate with a strong base like LDA to induce ortho-lithiation.
- Perform oxidative coupling using ferric chloride or copper salts to form the final axially chiral bissulfoxide ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages that directly address the pain points of cost and supply chain reliability in fine chemical manufacturing. The reliance on commodity chemicals such as aryl bromides, n-butyllithium, and ferric chloride significantly reduces the raw material cost compared to routes requiring exotic chiral pool resources or precious metal catalysts for the ligand synthesis itself. The elimination of resolution steps effectively doubles the theoretical yield relative to racemic pathways, which translates to substantial cost savings in terms of raw material consumption and waste disposal. Furthermore, the operational simplicity of the reaction, which proceeds in common solvents like tetrahydrofuran, facilitates easy technology transfer from laboratory to commercial scale without the need for specialized high-pressure or high-temperature equipment.
- Cost Reduction in Manufacturing: The process utilizes inexpensive oxidants like ferric chloride instead of costly noble metals for the coupling step, drastically lowering the bill of materials. By avoiding chromatographic resolution and achieving high yields through direct asymmetric synthesis, the overall production cost is significantly optimized. This efficiency allows for competitive pricing strategies when supplying high-purity OLED material or pharmaceutical intermediates to downstream clients.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted aryl halides and sulfinate esters, are widely available from global chemical suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted production schedules. This reliability reduces lead time for high-purity pharmaceutical intermediates, enabling faster time-to-market for new drug candidates.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, having been demonstrated effectively from milligram to multi-gram scales in the patent examples. The use of standard aqueous workups and the absence of toxic heavy metal residues in the final ligand structure simplify waste treatment and regulatory compliance. This aligns with modern green chemistry principles, making the commercial scale-up of complex polymer additives or agrochemical intermediates more sustainable and environmentally friendly.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, application, and customization of these novel ligands. The answers are derived directly from the experimental data and theoretical framework provided in the patent documentation, offering clarity for technical decision-makers evaluating this technology for integration into their existing catalytic processes.
Q: What is the optical stability of these axially chiral bissulfoxide ligands?
A: The ligands exhibit remarkable optical stability, with thermodynamic calculations indicating that racemization requires temperatures around 200°C, ensuring robustness during storage and reaction conditions.
Q: Can the steric and electronic properties of the ligands be tuned?
A: Yes, the synthesis allows for easy derivatization by varying the aryl halide and sulfinate ester starting materials, enabling precise tuning of steric and electronic effects for specific catalytic applications.
Q: What types of asymmetric reactions are these ligands suitable for?
A: These ligands are highly effective when coordinated with rhodium precursors for the asymmetric addition of aryl boronic acids to alpha,beta-unsaturated ketones, esters, and imines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Bissulfoxide Ligands Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of these axially chiral bissulfoxide ligands in advancing asymmetric synthesis. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from discovery to manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of ligand meets the exacting standards required for sensitive catalytic applications. We are committed to delivering high-purity pharmaceutical intermediates that empower your R&D teams to achieve breakthrough results.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific synthetic challenges. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and project timelines. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and innovation in your supply chain.
