Advanced Synthesis Of Axially Chiral Bissulfoxide Ligands For High-Performance Asymmetric Catalysis
Advanced Synthesis Of Axially Chiral Bissulfoxide Ligands For High-Performance Asymmetric Catalysis
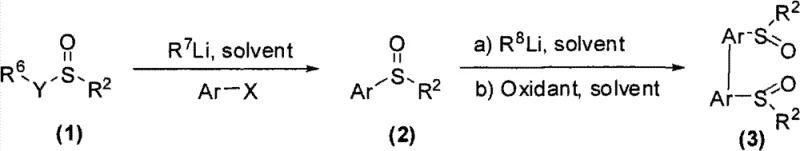
The landscape of asymmetric synthesis is continually evolving, driven by the demand for highly efficient and selective chiral catalysts. A significant breakthrough in this domain is detailed in patent CN102030690B, which discloses a robust methodology for the design and synthesis of a novel class of axially chiral bissulfoxide ligands. These ligands are pivotal for inducing high enantioselectivity in various organic transformations, particularly in the asymmetric addition of aryl boronic acids to α,β-unsaturated ketones and imines. The invention addresses the critical need for versatile chiral tools that can be easily derivatized to tune steric and electronic properties, thereby expanding the substrate scope for pharmaceutical and fine chemical manufacturing. By starting from optically pure sulfinate esters, this technology offers a streamlined pathway to complex chiral architectures that were previously difficult to access with high fidelity.
For procurement managers and supply chain directors, the implications of this technology extend beyond mere chemical novelty. The ability to reliably source high-purity pharmaceutical intermediates based on this scaffold ensures a stable supply chain for downstream drug synthesis. The method described eliminates the reliance on scarce natural chiral pools for the final ligand structure, instead building chirality from commercially available precursors. This shift not only enhances supply security but also opens avenues for cost reduction in pharmaceutical intermediate manufacturing by simplifying the synthetic sequence. As the industry moves towards more sustainable and efficient processes, adopting such innovative ligand systems becomes a strategic imperative for maintaining competitive advantage in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral ligands has been fraught with challenges related to complexity and low efficiency. Traditional routes often involve multi-step sequences requiring harsh conditions or expensive chiral auxiliaries that are difficult to recover. Many existing methods suffer from poor atom economy and generate significant waste, which poses environmental and disposal cost burdens. Furthermore, conventional approaches frequently lack modularity; modifying the electronic or steric environment of the ligand to optimize a specific reaction often necessitates a complete redesign of the synthetic route. This rigidity limits the ability of R&D teams to rapidly screen and identify the optimal catalyst for new substrates, leading to prolonged development timelines and increased resource consumption in the discovery phase.
The Novel Approach
The methodology presented in the patent data revolutionizes this landscape by introducing a concise, two-step strategy that maximizes efficiency and flexibility. By utilizing optically pure sulfinate esters as the chiral source, the process bypasses the need for resolution steps, directly transferring chirality to the final product with high fidelity. The first step involves the generation of a chiral sulfoxide intermediate via nucleophilic substitution, while the second step employs an oxidative coupling mechanism to forge the biaryl axis. This approach allows for the rapid diversification of the ligand library; researchers can simply vary the aryl halide or the sulfinate ester to access a wide array of structures. Such modularity is crucial for fine-tuning catalytic performance, enabling the precise regulation of enantioselectivity across different reaction types without compromising yield or operational simplicity.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Coupling
The core of this synthetic innovation lies in the oxidative coupling step, which constructs the sterically hindered biaryl axis essential for axial chirality. The mechanism begins with the deprotonation of the chiral sulfoxide intermediate using a strong base such as lithium diisopropylamide (LDA) at low temperatures, typically around -78°C. This generates a reactive carbanion species stabilized by the adjacent sulfoxide group. Upon introduction of an oxidant, specifically ferric chloride (FeCl3) or copper chloride, a single-electron transfer process initiates the radical coupling of two sulfoxide units. The choice of oxidant is critical, as it influences the rate of coupling and the suppression of side reactions. The resulting dimerization creates the axially chiral framework, locking the conformation due to the significant steric bulk of the substituents, which prevents rotation around the newly formed carbon-carbon bond.
Controlling impurities in this process is paramount for achieving high catalytic performance in downstream applications. The use of low-temperature conditions during the lithiation and coupling phases minimizes thermal degradation and racemization risks. Additionally, the specific stoichiometry of reagents, such as maintaining a molar ratio of sulfoxide to organolithium reagent between 1:1 and 1:2, ensures complete conversion while limiting over-reaction. The workup procedure, involving quenching with dilute acid and extraction, effectively removes metal salts and inorganic byproducts. This rigorous control over reaction parameters results in ligands with exceptional optical purity, which is a prerequisite for achieving high enantiomeric excess (ee) values in subsequent asymmetric catalysis, as demonstrated by the successful application in rhodium-catalyzed additions.
How to Synthesize Axially Chiral Bissulfoxide Ligands Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable ligands with high reproducibility. The process is designed to be operationally simple, utilizing standard laboratory equipment and widely available reagents. Key to success is the strict control of temperature and atmosphere, particularly during the handling of sensitive organolithium species. The following guide summarizes the critical stages involved in transforming simple precursors into sophisticated chiral ligands, ensuring that technical teams can implement this chemistry with confidence and precision.
- Perform lithiation of aryl halides at low temperature (-78°C) followed by nucleophilic substitution with optically pure sulfinate esters to form chiral sulfoxide intermediates.
- Treat the chiral sulfoxide intermediate with a strong base like LDA to generate an anion, followed by oxidative coupling using ferric chloride or copper chloride.
- Purify the resulting axially chiral bissulfoxide ligand via silica gel column chromatography to achieve high optical purity suitable for catalytic applications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits that align with the strategic goals of cost efficiency and supply reliability. The reliance on readily available starting materials, such as aryl halides and sulfinate esters, mitigates the risk of raw material shortages that often plague specialized chemical supply chains. Furthermore, the high yields observed in the exemplified reactions, such as the 88% yield for intermediate 2a and 75% for the final ligand 3a, translate directly into improved process economics. By reducing the number of synthetic steps and avoiding complex purification protocols, manufacturers can significantly lower production costs and minimize waste generation, contributing to a more sustainable and profitable operation.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolution steps and the use of cost-effective oxidants like ferric chloride drastically reduce the overall cost of goods sold. The high atom economy of the coupling reaction ensures that raw materials are utilized efficiently, minimizing waste disposal costs. Additionally, the simplified workflow reduces labor and energy consumption associated with prolonged reaction times and multiple isolation steps, leading to comprehensive financial savings.
- Enhanced Supply Chain Reliability: Sourcing stability is enhanced by the use of commodity chemicals as precursors. Unlike proprietary chiral pools that may have limited suppliers, aryl halides and sulfinate esters are produced by multiple vendors globally. This diversity in the supply base reduces dependency on single sources and buffers against market volatility. The robustness of the synthesis also means that production schedules are less likely to be disrupted by technical failures, ensuring consistent delivery of high-quality intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in batch reactors with standard cooling and stirring capabilities. The use of common solvents like tetrahydrofuran facilitates solvent recovery and recycling, aligning with green chemistry principles. Moreover, the avoidance of toxic heavy metals in the ligand synthesis itself (though used in catalysis later) simplifies regulatory compliance and environmental reporting, making it an attractive option for large-scale industrial adoption.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and application of this technology. These insights are derived directly from the experimental data and theoretical framework provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating these ligands into their existing catalytic processes.
Q: What represents the primary advantage of this ligand synthesis method?
A: The primary advantage lies in the operational simplicity and high yield. Starting from readily available optically pure sulfinate esters, the method achieves diverse structural modifications through a concise two-step sequence, allowing for precise tuning of steric and electronic effects without complex resolution steps.
Q: How stable are these axially chiral bissulfoxide ligands under reaction conditions?
A: These ligands exhibit remarkable thermal and optical stability. Thermodynamic calculations indicate that racemization typically requires temperatures around 200°C, ensuring that the chiral integrity is maintained during standard catalytic processes such as asymmetric additions at moderate temperatures.
Q: Can this synthetic route be scaled for industrial production?
A: Yes, the route is highly scalable. It utilizes common reagents like n-BuLi and FeCl3 in standard solvents like THF. The high yields reported in examples (e.g., 75-88%) and the straightforward workup procedures involving standard extraction and chromatography support robust commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Bissulfoxide Ligands Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced chiral ligands in modern drug discovery and process development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of axially chiral bissulfoxide ligands meets the highest standards of optical and chemical purity required for sensitive asymmetric syntheses.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your development timeline while optimizing your manufacturing costs.
