Revolutionizing 2,3-Difluoro-5-chloropyridine Production: A Technical Deep Dive into Scalable Synthesis
The landscape of fluorinated heterocycle synthesis is undergoing a significant transformation, driven by the urgent demand for high-purity intermediates in the agrochemical and pharmaceutical sectors. Patent CN115894349A introduces a groundbreaking synthetic methodology for 2,3-difluoro-5-chloropyridine, a critical building block for herbicides like clodinafop-propargyl. Unlike traditional approaches that rely on harsh halogen exchange reactions requiring extreme thermal energy, this novel protocol leverages a sophisticated two-step sequence involving diazotization-borylation followed by copper-mediated fluorination. This technical breakthrough not only addresses the longstanding issue of raw material scarcity but also fundamentally alters the impurity profile of the final product. For R&D directors and process chemists, this represents a paradigm shift towards milder, more selective chemistry that aligns perfectly with modern green manufacturing principles while ensuring robust supply chain continuity for high-value fluorinated pyridines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2,3-difluoro-5-chloropyridine has been plagued by significant technical and economic bottlenecks that hinder efficient commercial scale-up. One prevalent route, disclosed in earlier literature such as US4713109A1, relies on the nucleophilic aromatic substitution of 2,4-dichloro-3-fluoropyridine using cesium fluoride. While this method can achieve yields up to 87%, it suffers from a critical supply chain vulnerability: the starting material, 2,4-dichloro-3-fluoropyridine, is notoriously difficult to source commercially and commands a premium price due to its own complex synthesis. Furthermore, alternative routes utilizing 2,3,5-trichloropyridine as a substrate often necessitate the use of expensive ionic liquids or phase transfer catalysts under extremely harsh conditions.
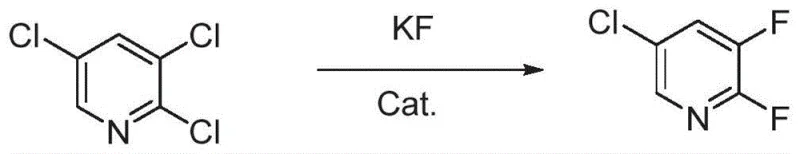
These conventional halogen exchange processes typically require reaction temperatures soaring between 180°C and 210°C, maintained for extended periods of 10 to 15 hours, which results in exorbitant energy consumption and safety risks. More critically, the high thermal energy input promotes non-selective fluorination, leading to a complex mixture of isomeric byproducts such as 2,5-difluoro-3-chloropyridine and other poly-halogenated species. Separating these structurally similar impurities often demands multi-stage distillation or chromatography, drastically reducing the overall process yield and increasing the cost of goods sold. The accumulation of these impurities poses a severe risk for downstream applications, particularly in pharmaceutical synthesis where strict impurity thresholds must be met.
The Novel Approach
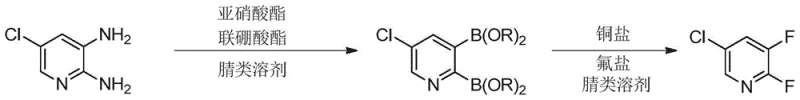
In stark contrast to the brute-force thermal methods of the past, the methodology described in CN115894349A offers an elegant solution by reimagining the bond construction strategy entirely. This innovative route begins with 2,3-diamino-5-chloropyridine, a commodity chemical that is readily available and cost-effective, thereby eliminating the raw material bottleneck associated with fluoro-chloro precursors. The process proceeds through a mild borylation step where the amino groups are converted into boronate esters using nitrites and diboronates in a nitrile solvent. This is followed by a copper-catalyzed fluorination that installs the fluorine atoms with high precision. The entire sequence operates at significantly lower temperatures, ranging from 40°C to 80°C, which represents a massive reduction in thermal load compared to the 200°C required by legacy methods.
The strategic advantage of this approach lies in its exceptional selectivity and operational simplicity. By avoiding the high-energy conditions that drive random halogen scrambling, the new method produces a much cleaner reaction profile with minimal formation of isomeric byproducts. This inherent purity means that the downstream purification burden is drastically reduced, often requiring only standard extraction and concentration rather than complex fractional distillation. For procurement managers, this translates to a more reliable supply of high-purity 2,3-difluoro-5-chloropyridine with consistent quality, while for plant managers, the lower operating temperatures and shorter reaction times facilitate safer and more energy-efficient commercial scale-up of complex agrochemical intermediates.
Mechanistic Insights into Copper-Catalyzed Fluorination
To fully appreciate the sophistication of this synthetic route, one must delve into the intricate mechanistic pathway that enables such high selectivity under mild conditions. The reaction initiates with the diazotization of 2,3-diamino-5-chloropyridine by the nitrite ester, generating a reactive diazonium salt intermediate in situ. This electrophilic species immediately undergoes a single-electron transfer borylation reaction with the diboronate reagent, effectively replacing the amino groups with boronate esters to form 2,3-diboronate-5-chloropyridine. This borylation step is crucial as it activates the pyridine ring for the subsequent metal-mediated transformation, setting the stage for the precise installation of fluorine atoms without disturbing the chlorine substituent at the 5-position.
The second phase involves a complex catalytic cycle mediated by copper salts. Upon addition of the fluoride salt and copper catalyst, a transmetallation event occurs where the boronate groups transfer the pyridyl moiety to the copper center, forming a pyridyl-copper-fluoride species. Mechanistic studies suggest that this divalent copper intermediate undergoes oxidation in the presence of additional copper salts to form a high-valent trivalent copper complex. It is from this trivalent state that the critical reductive elimination step takes place, forging the strong carbon-fluorine bonds and releasing the desired 2,3-difluoro-5-chloropyridine product. This mechanism explains the remarkable regioselectivity observed, as the copper center directs the fluorination specifically to the positions previously occupied by the boronates, effectively preventing the formation of the troublesome isomers seen in direct nucleophilic substitution.
How to Synthesize 2,3-Difluoro-5-chloropyridine Efficiently
Implementing this advanced synthesis protocol requires careful attention to reagent stoichiometry and solvent selection to maximize yield and purity. The process is designed to be telescoped, meaning the intermediate 2,3-diboronate-5-chloropyridine does not need to be isolated, which streamlines the workflow and reduces material loss. Detailed standardized operating procedures regarding specific molar ratios, heating rates, and workup protocols are essential for reproducibility. For technical teams looking to adopt this methodology, the following guide outlines the critical operational parameters derived from the patent examples to ensure successful execution.
- Heat a mixture of 2,3-diamino-5-chloropyridine, nitrite, and diboronate in a nitrile solvent to perform the borylation reaction.
- Add copper salt and fluoride salt to the reaction system without isolating the intermediate.
- Heat the second mixed system to carry out the fluorination reaction, then purify to obtain 2,3-difluoro-5-chloropyridine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers profound advantages that extend far beyond simple chemical yield improvements. For procurement managers tasked with optimizing the cost structure of agrochemical manufacturing, the shift away from scarce, expensive fluoro-chloro starting materials to abundant diamino precursors represents a fundamental stabilization of the supply base. The reliance on commodity chemicals like 2,3-diamino-5-chloropyridine mitigates the risk of price volatility and supply disruptions that often plague specialized fluorinated building blocks. Furthermore, the elimination of expensive ionic liquids and phase transfer catalysts, which were necessary in previous high-temperature methods, directly contributes to substantial cost savings in raw material expenditure.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the drastic reduction in energy consumption and processing time. By lowering the reaction temperature from over 200°C to a mild range of 40°C to 80°C, manufacturing facilities can achieve significant utility savings. The shorter reaction times, often completing within 12 to 24 hours compared to multi-day cycles in older methods, increase reactor throughput and asset utilization. Additionally, the simplified purification process, which avoids complex multi-stage distillations required to remove isomeric impurities, reduces both the capital equipment requirements and the operational labor costs associated with product isolation.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the use of robust, shelf-stable reagents such as alkyl nitrites and pinacol borates, which are widely available from multiple global suppliers. This diversification of the supply base reduces dependency on single-source vendors for exotic catalysts or hard-to-find substrates. The high selectivity of the reaction ensures consistent product quality batch after batch, minimizing the risk of production delays caused by out-of-specification material that requires re-processing. This reliability is critical for maintaining continuous production schedules for downstream herbicide formulations.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly smaller, aligning with increasingly stringent global regulations on chemical manufacturing. The use of common nitrile solvents like acetonitrile or propionitrile simplifies solvent recovery and recycling systems. Moreover, the reduction in byproduct formation means less hazardous waste generation, lowering the costs associated with waste treatment and disposal. The mild operating conditions also enhance process safety, reducing the risk of thermal runaways and making the technology highly suitable for safe commercial scale-up from pilot plants to multi-ton annual production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on how this method compares to established industry standards. Understanding these nuances is vital for decision-makers evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of the new synthesis route over traditional halogen exchange methods?
A: The new route utilizes readily available 2,3-diamino-5-chloropyridine instead of scarce fluoro-chloro precursors, operates at significantly lower temperatures (40-80°C vs 200°C), and minimizes difficult-to-separate isomeric byproducts.
Q: How does this method impact the purity profile of the final agrochemical intermediate?
A: By avoiding the harsh conditions of direct fluorination which often generate multiple chloro-fluoro isomers, this borylation-fluorination sequence ensures high regioselectivity, resulting in a product with superior purity suitable for sensitive pharmaceutical applications.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly highlights the ease of mass preparation due to the use of common nitrile solvents, stable reagents, and a simplified workup procedure that avoids complex multi-stage purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Difluoro-5-chloropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN115894349A are fully realized in practical, large-scale operations. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry, including corrosion-resistant reactors and rigorous QC labs capable of detecting trace impurities to meet stringent purity specifications demanded by the global agrochemical market.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your bottom line. We encourage potential partners to contact our technical procurement team to request specific COA data from our pilot batches and comprehensive route feasibility assessments. Let us help you secure a sustainable, high-quality supply of 2,3-difluoro-5-chloropyridine that drives your business forward.
