Advanced Fluorination Technology for Commercial Scale Production of Key Agrochemical Intermediates
Introduction to Advanced Fluorination Technologies in Agrochemical Synthesis
The development of efficient synthetic routes for fluorinated pyridine derivatives remains a cornerstone of modern agrochemical manufacturing, particularly for the production of high-performance herbicides such as alkynes grass ester. Patent CN101648904B introduces a transformative synthesis method for 2,3-difluoro-5-chloropyridine, a critical building block in this sector. This technology addresses long-standing economic and operational challenges by replacing expensive fluorinating agents with cost-effective potassium fluoride, activated through a sophisticated phase transfer catalysis system. By optimizing reaction temperatures and catalyst selection, the process achieves high conversion rates while maintaining operational simplicity, positioning it as a superior alternative to legacy methods that rely on scarce resources or hazardous reagents.
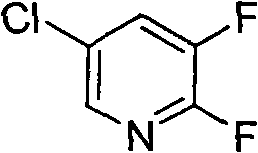
For procurement leaders and R&D directors seeking a reliable agrochemical intermediate supplier, understanding the nuances of this patent is essential. The shift from cesium-based chemistry to potassium-based systems represents not just a chemical modification, but a strategic supply chain optimization. The ability to utilize vacuum-dried potassium fluoride in common polar aprotic solvents like sulfolane or N-methylpyrrolidone (NMP) significantly lowers the barrier to entry for production. Furthermore, the detailed control over thermal profiles ensures that the reaction proceeds through a controlled mono-fluorinated intermediate before reaching the final difluoro product, minimizing impurity formation and simplifying downstream purification processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated pyridines has been plagued by significant economic and safety hurdles. Traditional approaches often relied on diazotization transformations, which necessitated the use of highly corrosive hydrogen fluoride and specialized autoclave reactors to manage low boiling points and high pressures. These methods, such as those described in US4831148, involve complex operational steps and pose severe safety risks due to the potential for explosive decomposition and the handling of toxic gases. Additionally, halogen displacement methods utilizing cesium fluoride, as seen in US4565568 and US4822887, suffered from prohibitively high raw material costs. Cesium salts are among the most expensive alkali metal reagents, and despite their use, reported yields rarely exceeded 30%, rendering these processes economically unviable for large-scale commercial applications.
The Novel Approach
The methodology outlined in CN101648904B fundamentally disrupts these limitations by leveraging the abundance of potassium fluoride. By introducing tetraphenylphosphonium bromide or tetrabutylphosphonium bromide as phase transfer catalysts, the process effectively solubilizes the fluoride anion in organic media, enhancing its nucleophilicity to levels comparable to expensive cesium salts. This innovation allows the reaction to proceed in standard organic solvents under inert gas protection, eliminating the need for specialized high-pressure equipment associated with HF chemistry. The result is a streamlined workflow that transforms 2,3,5-trichloropyridine into the target difluoro compound with transformation efficiencies reaching nearly 100%, offering a robust pathway for cost reduction in agrochemical intermediates manufacturing.
Mechanistic Insights into Phosphonium-Catalyzed Halogen Exchange
The core of this technological breakthrough lies in the precise mechanism of halogen exchange fluorination facilitated by quaternary phosphonium salts. Unlike simple thermal substitution, the presence of the phosphonium catalyst creates a lipophilic ion pair with the fluoride anion, allowing it to penetrate the organic phase and attack the electron-deficient pyridine ring. The reaction proceeds sequentially; initially, the chlorine atom at the 2-position is displaced due to its activation by the adjacent nitrogen atom and the electron-withdrawing effect of the ring. This generates the mono-fluorinated intermediate, 2-fluoro-3,5-dichloropyridine, which accumulates during the lower temperature phase of the reaction.
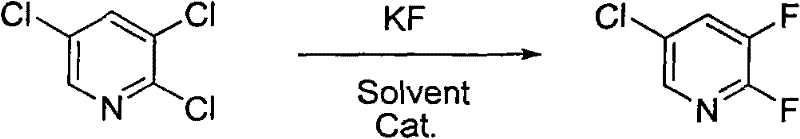
Subsequent elevation of the temperature provides the necessary activation energy to displace the second chlorine atom at the 3-position, completing the synthesis of 2,3-difluoro-5-chloropyridine. This stepwise thermal control is critical for impurity management; rushing the reaction to high temperatures immediately can lead to non-selective fluorination or degradation of the solvent system. By carefully managing the transition from 180-190°C to 200-210°C, the process ensures that the kinetic barriers for each substitution step are overcome sequentially. This mechanistic precision results in a cleaner crude product profile, reducing the burden on distillation columns and lowering the overall energy consumption per kilogram of high-purity agrochemical intermediate produced.
How to Synthesize 2,3-Difluoro-5-Chloropyridine Efficiently
Implementing this synthesis route requires strict adherence to the specified molar ratios and thermal gradients to ensure reproducibility and safety. The process begins with the rigorous drying of potassium fluoride to remove moisture, which can otherwise deactivate the catalyst or hydrolyze the sensitive chloropyridine substrates. Operators must maintain an inert atmosphere throughout the heating cycle to prevent oxidation of the phosphonium catalyst, which could lead to the formation of phosphine oxides and loss of catalytic activity. The following guide outlines the standardized operational parameters derived from the patent examples to assist technical teams in replicating this high-efficiency pathway.
- Prepare the reaction mixture by dissolving 2,3,5-trichloropyridine and vacuum-dried potassium fluoride in a polar aprotic solvent such as sulfolane or NMP, adding a phosphonium phase transfer catalyst.
- Heat the mixture under inert gas protection to an initial temperature range of 180-190°C and maintain for 5-10 hours to facilitate mono-fluorination.
- Increase the temperature to 200-210°C and maintain for an additional 10-15 hours to complete the di-fluorination, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the adoption of this synthesis method offers tangible strategic benefits beyond mere chemical yield. The primary advantage is the drastic reduction in raw material expenditure by substituting cesium fluoride with commodity-grade potassium fluoride. This switch decouples production costs from the volatile pricing of rare alkali metals, stabilizing the cost base for long-term contracts. Furthermore, the use of standard solvents like NMP and sulfolane, which are widely available in the global chemical market, mitigates the risk of supply disruptions that often plague specialty reagent markets. This reliability is crucial for maintaining continuous production schedules in the fast-paced agrochemical sector.
- Cost Reduction in Manufacturing: The elimination of expensive cesium salts directly impacts the bill of materials, allowing for significant margin improvement or competitive pricing strategies. Additionally, the high conversion rate of the starting material (up to 100%) minimizes waste disposal costs associated with unreacted chloropyridines. The simplified workup procedure, avoiding complex neutralization steps required for HF processes, further reduces utility consumption and labor hours, contributing to a leaner manufacturing overhead structure.
- Enhanced Supply Chain Reliability: Sourcing potassium fluoride is far less complex than securing high-purity cesium fluoride, which often involves limited suppliers and long lead times. By utilizing a ubiquitous reagent, manufacturers can diversify their supplier base and reduce inventory holding costs. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, ensuring consistent output even when sourcing from different batches of raw materials, thereby enhancing overall supply chain resilience.
- Scalability and Environmental Compliance: The absence of corrosive hydrogen fluoride and the avoidance of high-pressure autoclaves simplify the engineering requirements for plant scale-up. Standard glass-lined or stainless steel reactors are sufficient, reducing capital expenditure for new capacity. From an environmental perspective, the process generates less hazardous waste compared to diazotization routes, facilitating easier compliance with increasingly stringent environmental regulations regarding fluorine-containing effluents and heavy metal residues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational feasibility and economic rationale for adopting this specific synthetic route over traditional alternatives.
Q: Why is potassium fluoride preferred over cesium fluoride in this synthesis?
A: Potassium fluoride is significantly cheaper and more readily available than cesium fluoride. While traditional methods required expensive cesium salts to achieve reasonable yields, this patented process utilizes specific phosphonium phase transfer catalysts to activate the cheaper potassium fluoride, drastically reducing raw material costs without compromising conversion efficiency.
Q: What are the critical temperature controls for maximizing yield?
A: The process employs a two-stage heating protocol. The reaction is first held at 180-190°C to accumulate the mono-fluorinated intermediate (2-fluoro-3,5-dichloropyridine). Subsequently, the temperature is raised to 200-210°C to drive the second fluorination step. This staged approach prevents side reactions and ensures high transformation efficiency of the starting trichloropyridine.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for scalability. It avoids hazardous diazotization steps requiring autoclaves and corrosive hydrogen fluoride. The use of standard organic solvents like NMP or sulfolane, combined with stable phosphonium catalysts, allows for safe operation in standard stainless steel reactors, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Difluoro-5-Chloropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient fluorination chemistry plays in the global agrochemical supply chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering high-purity agrochemical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for fluorinated pyridine derivatives. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term growth objectives in the agricultural chemicals market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
