Scalable Chiral Resolution of Tetrabenazine for High-Purity API Intermediate Production
The pharmaceutical landscape for treating neurodegenerative disorders such as Huntington's disease has increasingly demanded high-purity chiral intermediates to ensure efficacy and safety profiles. Patent CN111285867A introduces a transformative approach to the preparation of benzoquinazine derivatives, specifically focusing on the chiral resolution of Tetrabenazine (TBZ). This intellectual property outlines a robust methodology that bypasses the limitations of traditional asymmetric synthesis or expensive chromatographic separations by utilizing diastereomeric salt formation with specific tartaric acid derivatives. For R&D directors and procurement specialists in the fine chemical sector, this technology represents a pivotal shift towards more cost-effective and scalable manufacturing of critical API intermediates. The core innovation lies in the strategic selection of L- or D-di-p-methylbenzoyltartaric acid as the resolving agent, which enables the isolation of (3R,11bR)-Tetrabenazine or (3S,11bS)-Tetrabenazine with exceptional optical purity. By leveraging this patented resolution strategy, manufacturers can achieve optical purity levels exceeding 99% ee, a benchmark essential for regulatory compliance in modern drug development. This report analyzes the technical merits and commercial viability of this process, highlighting its potential to redefine supply chain standards for rare disease therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active Tetrabenazine has been plagued by significant technical and economic hurdles that hinder efficient large-scale manufacturing. Prior art methods, such as those described in WO2008058261, often rely on complex asymmetric synthesis routes involving toxic reagents like methyl iodide, which pose severe environmental and safety risks in an industrial setting. Furthermore, these conventional synthetic pathways frequently suffer from extremely low reaction yields, reported as low as 16%, necessitating the recovery of excessive starting materials and driving up production costs substantially. Alternative approaches utilizing chiral chromatographic columns, while effective for analytical purposes, are notoriously difficult to scale due to the high cost of stationary phases and the low throughput associated with column loading capacities. Other resolution attempts using camphorsulfonic acid have demonstrated instability upon scale-up, with patent data indicating a drastic drop in yield to merely 6% when moving from gram to kilogram scales. These inefficiencies create a bottleneck in the supply chain, leading to inconsistent batch quality and prolonged lead times for downstream drug formulation. Consequently, the industry has urgently required a resolution method that eliminates toxic reagents, avoids complex purification equipment, and maintains high efficiency regardless of batch size.
The Novel Approach
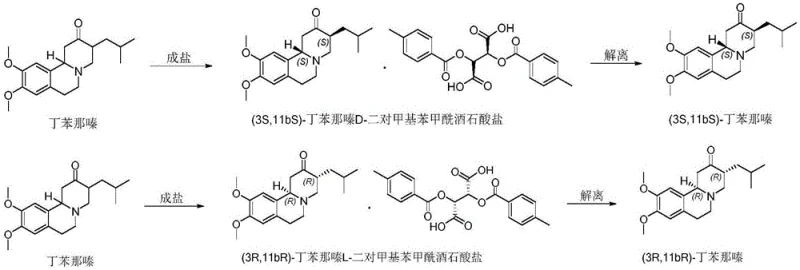
The methodology disclosed in CN111285867A offers a streamlined alternative that addresses these historical pain points through a simplified salification and crystallization protocol. Instead of relying on hazardous alkylating agents or expensive chromatography, this novel approach utilizes the differential solubility of diastereomeric salts formed between racemic Tetrabenazine and di-p-methylbenzoyltartaric acid. The process involves a straightforward reaction in ethyl acetate, where the target enantiomer selectively precipitates out of the solution upon cooling, leaving impurities and the undesired enantiomer in the mother liquor. This crystallization-driven separation is inherently scalable, as evidenced by successful pilot runs ranging from 100 grams to 5 kilograms without loss of performance. The use of ethyl acetate as the primary solvent further enhances the green chemistry profile of the process, replacing more problematic solvents often found in legacy methods. By integrating a recrystallization step, the optical purity can be upgraded from an initial 80-88% to over 99% ee, ensuring the final product meets stringent pharmaceutical specifications. This shift from synthesis-dependent chirality to resolution-dependent chirality significantly reduces the complexity of the manufacturing workflow, making it an ideal candidate for reliable API intermediate supplier networks seeking to optimize their production capabilities.
Mechanistic Insights into Diastereomeric Salt Resolution
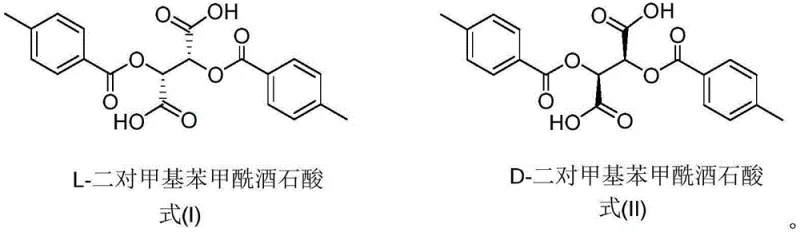
The success of this resolution strategy hinges on the precise stereochemical interaction between the basic nitrogen of the Tetrabenazine scaffold and the acidic protons of the chiral resolving agent. When racemic Tetrabenazine is introduced to L-di-p-methylbenzoyltartaric acid, two distinct diastereomeric salts are formed: one comprising the (3R,11bR)-enantiomer and the other comprising the (3S,11bS)-enantiomer. Due to the three-dimensional arrangement of the bulky p-toluoyl groups on the tartaric acid backbone, these two salts exhibit significantly different lattice energies and solubility profiles in ethyl acetate. The (3R,11bR)-salt, for instance, possesses a crystal lattice structure that is less soluble in the chosen solvent system at lower temperatures, prompting it to nucleate and grow preferentially during the cooling phase. This thermodynamic preference is the driving force behind the enrichment of the desired enantiomer in the solid phase, while the unwanted isomer remains dissolved in the liquid phase. The structural integrity of the resolving agent is crucial, as the para-methyl substituents on the benzoyl rings provide the necessary steric bulk to differentiate between the enantiomers effectively. Without this specific substitution pattern, as seen with unsubstituted dibenzoyltartaric acid in screening experiments, the solubility differences are insufficient to induce precipitation, resulting in failure to resolve the mixture.
Impurity control within this mechanism is achieved through the iterative nature of the crystallization process, which acts as a powerful purification engine. During the initial salification, the crude precipitate may contain minor amounts of the counter-enantiomer or unreacted starting material, typically yielding an ee value around 80-88%. However, the subsequent suspension and recrystallization step in fresh ethyl acetate leverages the eutectic composition of the system to further exclude impurities from the growing crystal lattice. As the crystals reform under controlled heating and cooling cycles, the thermodynamic stability of the pure diastereomeric salt drives the exclusion of mismatched stereoisomers, pushing the optical purity to exceed 99% ee. This self-correcting mechanism ensures that even if the initial reaction conditions vary slightly, the final product quality remains consistent and high. Furthermore, the dissociation step using mild alkaline solutions like sodium carbonate or potassium hydroxide ensures that the chiral integrity of the Tetrabenazine molecule is preserved during the liberation of the free base. This gentle workup prevents racemization, a common risk in chiral processing, thereby guaranteeing that the high optical purity achieved during crystallization is retained in the final isolated API intermediate.
How to Synthesize (3R,11bR)-Tetrabenazine Efficiently
The synthesis of high-purity (3R,11bR)-Tetrabenazine via this resolution pathway is designed for operational simplicity and robustness in a GMP environment. The process begins with the precise stoichiometric mixing of racemic Tetrabenazine and L-di-p-methylbenzoyltartaric acid in ethyl acetate, followed by a controlled thermal cycle to induce selective crystallization. Detailed standard operating procedures regarding temperature gradients, stirring rates, and filtration protocols are critical to maximizing both yield and optical purity. For a comprehensive understanding of the specific parameters required to replicate this high-efficiency outcome, please refer to the standardized synthesis guide below.
- Salification and Crystallization: React racemic Tetrabenazine with L- or D-di-p-methylbenzoyltartaric acid in ethyl acetate under reflux, followed by cooling to precipitate the diastereomeric salt.
- Purification: Suspend the crude salt in ethyl acetate, heat to reflux, and cool to recrystallize, achieving optical purity (ee value) greater than 99%.
- Dissociation: Treat the purified salt with an alkaline aqueous solution (e.g., sodium carbonate or hydroxide), extract with an organic solvent like dichloromethane, and dry to obtain the free base enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this chiral resolution technology offers substantial strategic benefits for procurement managers and supply chain directors aiming to optimize their sourcing strategies for pharmaceutical intermediates. The elimination of complex asymmetric synthesis steps and toxic reagents translates directly into a simplified supply chain with fewer raw material dependencies and reduced regulatory burden. By shifting to a crystallization-based purification method, manufacturers can significantly reduce the capital expenditure associated with specialized chromatography equipment and the ongoing operational costs of column packing and solvent consumption. This process intensification allows for higher throughput in existing reactor infrastructure, effectively increasing production capacity without the need for new facility construction. Moreover, the use of common, non-hazardous solvents like ethyl acetate simplifies waste management and lowers the environmental compliance costs associated with volatile organic compound disposal. These factors collectively contribute to a more resilient and cost-efficient manufacturing model that can better withstand market fluctuations and raw material price volatility.
- Cost Reduction in Manufacturing: The transition from low-yield asymmetric synthesis or expensive chromatographic separation to this high-yield resolution process fundamentally alters the cost structure of Tetrabenazine production. By avoiding the use of costly chiral catalysts and eliminating the need for preparative HPLC columns, the direct material costs are drastically lowered while simultaneously improving the overall mass balance of the process. The ability to recover and recycle the resolving acid from the mother liquor further enhances the economic viability, creating a closed-loop system that minimizes waste generation. Additionally, the high optical purity achieved reduces the need for downstream reprocessing or additional purification steps, streamlining the entire production timeline. This efficiency gain allows suppliers to offer more competitive pricing models while maintaining healthy margins, providing a clear financial advantage for long-term procurement contracts.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the robustness and scalability of this resolution method, which has been proven effective from laboratory bench scales up to multi-kilogram pilot batches. The reliance on commercially available and stable chiral acids ensures that raw material sourcing is not subject to the bottlenecks often associated with custom-synthesized chiral auxiliaries. The process tolerance to minor variations in reaction conditions means that batch-to-batch consistency is high, reducing the risk of production delays caused by failed quality control tests. Furthermore, the simplified operational workflow requires less specialized labor training, allowing for more flexible staffing and reduced dependency on highly niche technical expertise. This operational stability ensures that delivery schedules can be met reliably, safeguarding the downstream drug formulation timelines against upstream manufacturing disruptions.
- Scalability and Environmental Compliance: The environmental footprint of this manufacturing process is markedly smaller than traditional methods, aligning with the increasing global demand for sustainable chemical production practices. The replacement of toxic reagents like methyl iodide with benign carboxylic acids removes a major hazardous waste stream, simplifying effluent treatment and reducing the liability associated with hazardous material handling. The high atom economy of the resolution step, combined with the potential for solvent recycling, supports a greener manufacturing profile that appeals to environmentally conscious stakeholders. Scalability is inherent in the design, as crystallization processes generally translate linearly from pilot to commercial scale without the nonlinear challenges often seen in catalytic reactions. This ease of scale-up ensures that supply can be rapidly expanded to meet market demand surges without compromising on quality or environmental standards, securing a long-term license to operate in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral resolution technology for Tetrabenazine derivatives. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production lines or for qualifying new suppliers.
Q: Why is di-p-methylbenzoyltartaric acid preferred over other chiral acids for Tetrabenazine resolution?
A: Screening experiments demonstrated that while acids like D-camphoric acid or D-mandelic acid yielded lower optical purity and recovery, di-p-methylbenzoyltartaric acid provided superior crystallization properties and achieved ee values exceeding 99% after recrystallization.
Q: What is the critical solvent for maximizing optical purity in this resolution process?
A: Ethyl acetate was identified as the optimal solvent. Compared to acetone or alcohol mixtures, ethyl acetate facilitated the formation of the diastereomeric salt with the highest initial ee value and allowed for effective purification through recrystallization without significant yield loss.
Q: Is this chiral resolution method suitable for large-scale industrial production?
A: Yes, the patent data confirms successful scale-up from gram scales to multi-kilogram batches (e.g., 3kg to 5kg inputs). The process maintains stable yields around 40-46% and consistent optical purity above 99% ee, indicating robust reproducibility for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrabenazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-quality chiral intermediates that meet the rigorous demands of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of Tetrabenazine exceeds the 99% ee threshold required for therapeutic applications. Our infrastructure is designed to support the complex crystallization and purification steps outlined in this patent, guaranteeing a consistent supply of premium-grade API intermediates for your drug development programs.
We invite you to collaborate with us to leverage this advanced resolution technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that details how implementing this process can optimize your budget without compromising quality. Please contact us today to request specific COA data and route feasibility assessments tailored to your volume requirements. By partnering with NINGBO INNO PHARMCHEM, you secure a supply chain partner dedicated to technical excellence, regulatory compliance, and long-term value creation in the synthesis of complex benzoquinazine derivatives.
