Advanced One-Pot Synthesis of 6,7-Dimethoxy-3,4-Dihydroisoquinoline Hydrochloride for Pharmaceutical Manufacturing
Introduction to Patent CN110845410B
The pharmaceutical industry constantly seeks more efficient and safer pathways for synthesizing critical intermediates, particularly for neurological treatments like Huntington's disease. Patent CN110845410B introduces a groundbreaking one-pot method for preparing 6,7-dimethoxy-3,4-dihydroisoquinoline hydrochloride, a key precursor for Tetrabenazine. This innovation addresses the longstanding challenges of corrosion, safety, and purification associated with traditional Bischler-Napieralski reactions. By utilizing phosphotungstic acid as a novel catalyst, the process achieves exceptional purity levels exceeding 99.0% with minimal impurities, all while simplifying the operational workflow into a single vessel sequence.

This technical advancement represents a significant leap forward for reliable pharmaceutical intermediate supplier networks, offering a route that drastically reduces equipment corrosion risks and waste generation. The ability to obtain cGMP-compliant material through simple cooling and filtration, rather than complex chromatographic separations, underscores the industrial viability of this approach for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
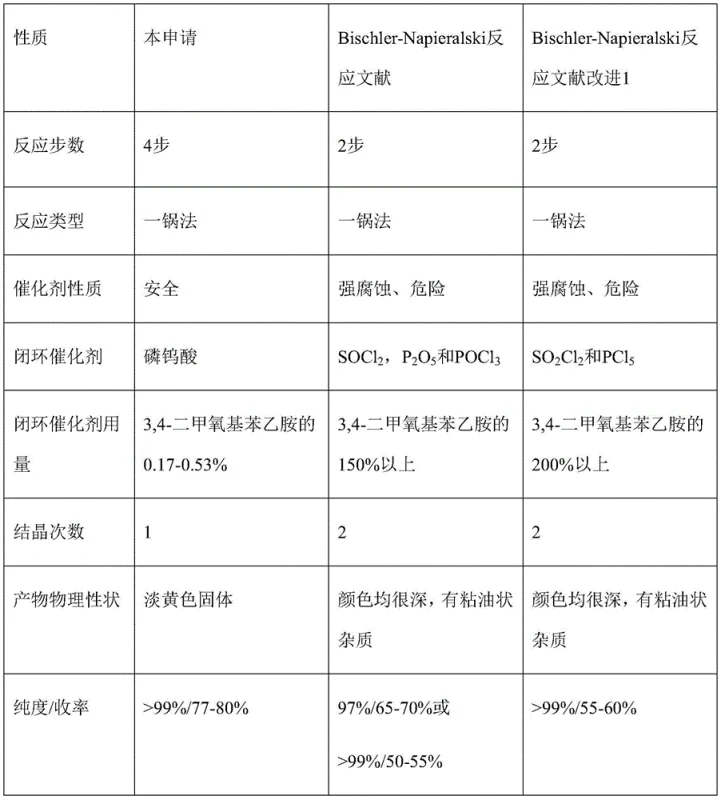
Historically, the synthesis of dihydroisoquinoline derivatives has relied on harsh conditions that pose significant operational hazards and quality control issues. Traditional routes often employ aggressive chlorinating agents such as phosphorus oxychloride (POCl3), thionyl chloride (SOCl2), or phosphorus pentoxide (P2O5) to effect ring closure. These reagents are not only highly corrosive to standard reactor linings but also generate violent exotherms during quenching, elevating the safety risk profile of the manufacturing site. Furthermore, literature data indicates that reactions using these catalysts frequently result in reaction masses that are very dark in color and contain stubborn oily impurities.

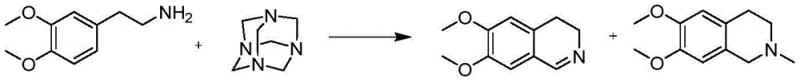
To achieve the requisite purity of >99% for pharmaceutical use, these conventional methods typically necessitate two or more recrystallization steps, which severely impacts the overall yield, often dropping it to between 50% and 60%. Additionally, alternative routes using urotropine have been plagued by the formation of N-methyl impurities at levels as high as 10%, creating a difficult purification burden that increases cost reduction in pharmaceutical intermediate manufacturing efforts.
The Novel Approach
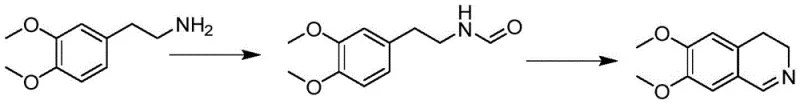
In stark contrast, the methodology disclosed in CN110845410B utilizes a mild yet effective catalytic system centered around phosphotungstic acid. This approach integrates formylation, activation with oxalyl chloride, and cyclization into a seamless one-pot operation. The use of oxalyl chloride allows for the generation of the reactive imidoyl chloride intermediate under controlled temperatures (10-20°C), avoiding the thermal runaway risks associated with traditional reagents. The subsequent addition of phosphotungstic acid promotes the cyclization with remarkable selectivity, yielding a pale yellow solid product directly from the reaction mixture.
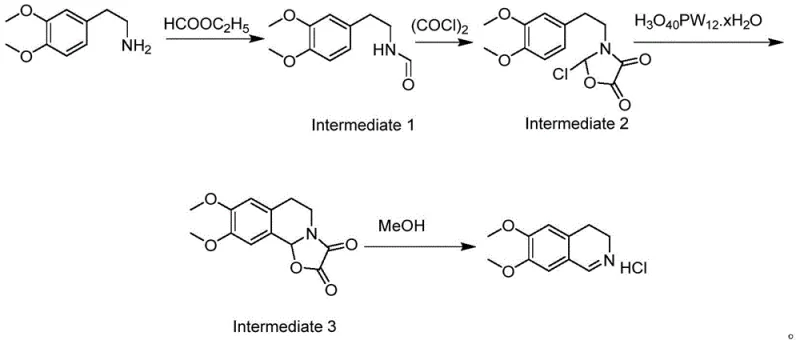
This streamlined process eliminates the need for multiple isolation steps and hazardous reagent handling. The final product is obtained simply by cooling the reaction mass, filtering, and drying, which not only improves the yield to over 75% but also ensures a consistent physical appearance free from the dark tarry byproducts common in older literature methods. This represents a robust solution for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Phosphotungstic Acid Catalyzed Cyclization
The core of this innovation lies in the replacement of traditional Lewis acids with a heteropoly acid catalyst. In the proposed mechanism, 3,4-dimethoxyphenethylamine is first formylated to protect the amine and activate the side chain. Upon treatment with oxalyl chloride, the formamide oxygen is activated, likely forming an imidoyl chloride species. In conventional chemistry, strong Lewis acids like FeCl3 or POCl3 are required to facilitate the subsequent electrophilic aromatic substitution to close the ring. However, these often lead to over-reaction or decomposition.
Phosphotungstic acid acts as a solid superacid that provides the necessary proton activity to drive the cyclization without the aggressive halogenation side reactions seen with SOCl2 or PCl5. The bulky structure of the phosphotungstate anion may also play a role in stabilizing the transition state, preventing the formation of polymeric tars. This specific catalytic environment ensures that the electron-rich dimethoxy-substituted benzene ring undergoes cyclization efficiently while minimizing oxidative degradation, resulting in the observed high purity and light color of the final hydrochloride salt.
How to Synthesize 6,7-Dimethoxy-3,4-Dihydroisoquinoline Efficiently
The synthesis protocol outlined in the patent describes a precise sequence of additions and temperature controls designed to maximize safety and yield. The process begins with the reflux of the starting amine with ethyl formate, followed by the careful addition of oxalyl chloride in acetonitrile. The critical step involves the introduction of the phosphotungstic acid catalyst at low loading (0.17-0.53% by weight), followed by a methanol quench to remove oxalic acid byproducts and form the hydrochloride salt. For the complete standardized operating procedure and specific reaction parameters, please refer to the detailed guide below.
- React 3,4-dimethoxyphenethylamine with ethyl formate under reflux to form the formylated intermediate.
- Add oxalyl chloride solution at 10-20°C to activate the intermediate for cyclization.
- Introduce catalytic amounts of phosphotungstic acid to promote ring closure safely.
- Quench with methanol, cool to crystallize, and filter to obtain the high-purity hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this one-pot methodology offers substantial strategic benefits beyond mere chemical efficiency. The elimination of highly corrosive reagents like sulfuryl chloride and phosphorus oxychloride significantly reduces the maintenance costs associated with reactor vessels and piping, extending the lifespan of capital equipment. Furthermore, the simplified workup procedure, which avoids multiple recrystallizations and column chromatography, translates directly into reduced labor hours and faster batch cycle times, enhancing overall production throughput.
- Cost Reduction in Manufacturing: The process utilizes inexpensive and readily available raw materials such as ethyl formate and oxalyl chloride, avoiding the need for costly noble metal catalysts like ruthenium or expensive solvents like trifluoroacetic acid. By achieving high purity (>99%) in a single crystallization step, the method drastically reduces solvent consumption and waste disposal costs associated with mother liquor recycling, leading to significant overall cost optimization.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than specialized or regulated reagents ensures a stable supply chain with minimal risk of raw material shortages. The robustness of the reaction conditions, specifically the tolerance for slight variations in temperature and the use of stable solid catalysts, minimizes the risk of batch failures, thereby guaranteeing consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental perspective, the replacement of hazardous chlorinating agents with phosphotungstic acid markedly lowers the toxicity profile of the waste stream. The process generates less acidic wastewater and avoids the release of sulfur dioxide or phosphorus-containing gases, facilitating easier compliance with increasingly stringent environmental regulations and reducing the burden on effluent treatment plants.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, highlighting the practical advantages for industrial adoption.
Q: Why is phosphotungstic acid preferred over POCl3 or SOCl2 for this cyclization?
A: Traditional catalysts like POCl3 and SOCl2 are highly corrosive and dangerous, often producing dark-colored products with oily impurities that require multiple recrystallizations. Phosphotungstic acid is a safe, solid acid catalyst that yields a pale yellow solid with >99% purity in a single crystallization step.
Q: What is the expected purity and yield of the one-pot method?
A: The patented one-pot method consistently achieves a chemical purity of greater than 99.0% with single impurities controlled below 0.15%. The overall yield is significantly improved to over 75%, compared to 50-60% for traditional high-purity routes.
Q: Does this process avoid the N-methyl impurity found in other routes?
A: Yes. Unlike routes using urotropine which can generate up to 10% N-methyl impurity, this formylation-oxalyl chloride pathway ensures high selectivity for the target dihydroisoquinoline structure without methylated byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,7-Dimethoxy-3,4-Dihydroisoquinoline Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of high-quality intermediates in the development of life-saving neurological medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to full-scale manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 6,7-dimethoxy-3,4-dihydroisoquinoline hydrochloride meets the highest cGMP standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your next project milestone.
