Advanced Low-Pressure Hydrogenation for Pentazocine Intermediate Manufacturing and Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of potent analgesics, particularly opioid derivatives like pentazocine. A pivotal advancement in this domain is detailed in patent CN112679363A, which discloses a highly efficient method for preparing a critical pentazocine intermediate, specifically 3-methyl-3-pentenylamine (Compound XVII). This innovation addresses longstanding bottlenecks in the synthetic route, transitioning from hazardous high-pressure conditions to a milder, industrially viable process. The core of this technology lies in the catalytic hydrogenation of 3-methyl-3-pentenenitrile (Compound XV) using Raney nickel in the presence of an ammonia-alcohol solution. By optimizing the reaction environment, the patent demonstrates that it is possible to achieve high conversion rates while significantly mitigating operational risks. This technical breakthrough not only enhances the safety profile of the manufacturing process but also opens new avenues for cost-effective production at a commercial scale. For global supply chains, the ability to produce high-purity pharmaceutical intermediates under such controlled and mild conditions represents a substantial leap forward in process chemistry reliability.
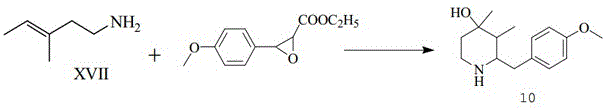
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Compound XVII has been plagued by severe operational constraints that hinder large-scale industrial adoption. Traditional routes, such as those referenced in older literature like Chem. Pharm. Bull. Vol 24 (6) 1246-1253 (1976), typically require hydrogenation pressures ranging from 5 to 6 MPa. Such high-pressure environments necessitate the use of specialized, expensive autoclaves and impose rigorous safety protocols that increase both capital expenditure and operational complexity. Furthermore, these conventional methods often suffer from prolonged reaction times, frequently extending up to 40 hours, which severely limits throughput and equipment utilization rates. Another critical drawback is the high catalyst loading required, with mass ratios of catalyst to raw material often exceeding 1.2:1. This excessive use of Raney nickel not only drives up raw material costs but also complicates downstream waste management and catalyst recovery processes. Additionally, the purification of the starting material, Compound XV, is notoriously difficult, often leading to the use of impure mixtures that further compromise yield and selectivity, with reported yields for the mixture of products XVII and XVIII hovering around a suboptimal 55%.
The Novel Approach
In stark contrast, the methodology outlined in patent CN112679363A introduces a paradigm shift by leveraging an ammonia-alcohol solution during the hydrogenation step. This innovative approach allows the reduction reaction to proceed effectively at a drastically reduced hydrogen pressure of merely 0.6 to 0.7 MPa. The introduction of ammonia gas dissolved in alcohols such as ethanol, methanol, or isopropanol creates a unique reaction medium that facilitates the reduction at much lower temperatures, specifically between 35°C and 45°C. This mild condition profile eliminates the need for extreme pressure vessels, thereby enhancing the intrinsic safety of the operation. Moreover, the reaction time is compressed significantly to a window of just 6 to 9 hours, representing a massive improvement in process efficiency. The optimized conditions also permit a reduction in catalyst usage, with the Raney nickel to raw material ratio lowered to a range of 0.6-0.8:1. This reduction in catalyst loading, combined with the ability to process crude mixtures of Compound XV and its isomer Compound XVI directly without prior purification, streamlines the entire workflow. The result is a process that is not only safer and faster but also inherently more economical and suitable for continuous industrial production.

Mechanistic Insights into Raney Nickel Catalyzed Reduction with Ammonia
The efficacy of this novel process is rooted in the specific interaction between the Raney nickel catalyst, the nitrile substrate, and the ammonia-alcohol solvent system. In standard nitrile hydrogenation, the formation of secondary and tertiary amines via condensation reactions between the intermediate imine and the primary amine product is a common side reaction that reduces yield and purity. The presence of ammonia in the reaction mixture serves a critical mechanistic role by shifting the equilibrium and suppressing these condensation pathways. By maintaining a high concentration of ammonia (optimized between 12% and 14% by mass in the alcohol solvent), the system ensures that the intermediate imine species preferentially reacts with ammonia to form the desired primary amine (Compound XVII) rather than coupling with other amine molecules. This suppression of by-product formation is crucial for maintaining high selectivity. Furthermore, the alcohol solvent plays a dual role: it acts as a carrier for the ammonia gas, ensuring uniform distribution throughout the reaction matrix, and it aids in the solvation of the organic substrates. The specific choice of alcohol (ethanol, methanol, or isopropanol) influences the solubility of ammonia and the stability of the catalyst, with ethanol often providing an optimal balance for industrial applications.
From an impurity control perspective, the mechanism also accounts for the presence of the isomeric starting material, Compound XVI (3-methyl-2-pentenenitrile). Under the optimized Raney nickel catalysis, both the Z/E isomers of the starting nitrile (Compound XV) and the positional isomer (Compound XVI) are reduced to their corresponding amines, Compound XVII and Compound XVIII. While this results in a mixed product, the downstream synthesis of pentazocine is tolerant to this mixture. The key mechanistic advantage here is that the mild conditions prevent the degradation or polymerization of these sensitive unsaturated nitriles, which can occur under harsher thermal or acidic conditions. The catalyst surface activity is modulated by the ammonia, preventing over-reduction of the double bond while ensuring complete reduction of the nitrile group. This selectivity is vital for preserving the structural integrity of the carbon backbone required for the subsequent cyclization steps in the pentazocine synthesis pathway. The ability to handle the crude mixture directly without isolating pure Compound XV simplifies the impurity profile management, as the downstream crystallization of the final intermediate (Compound 10) effectively separates the desired product from the isomeric by-products.
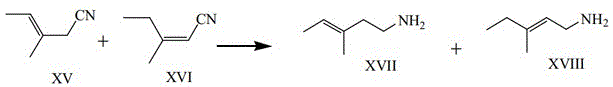
How to Synthesize 3-Methyl-3-pentenylamine Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of unit operations that are well-suited for standard chemical manufacturing facilities. The process begins with the preparation of the ammonia-alcohol solution, where ammonia gas is sparged into the chosen alcohol solvent under cooling to maintain temperatures below 10°C, ensuring high solubility and safety. This solution is then transferred to a hydrogenation reactor pre-charged with the Raney nickel catalyst. The crude nitrile mixture, obtained from the condensation of cyanoacetic acid and butanone, is added directly to this slurry. Following the displacement of air with nitrogen to prevent oxidation hazards, hydrogen gas is introduced to reach the target operating pressure. The reaction is exothermic, but the mild operating temperature range of 35-45°C allows for easy thermal management using standard jacketed reactors. Upon completion, typically indicated by the cessation of hydrogen uptake or confirmed via GC analysis after 6-9 hours, the catalyst is filtered off, and the solvent and excess ammonia are removed under reduced pressure. The resulting crude amine mixture is stable and ready for the next synthetic transformation. For a detailed, step-by-step breakdown of the standardized operating procedures and quality control checkpoints, please refer to the technical guide below.
- Prepare the catalyst system by adding Raney nickel to a reactor and introducing ammonia gas into an alcohol solvent (ethanol, methanol, or isopropanol) at temperatures below 10°C to achieve an ammonia mass content of 5-30%.
- Charge the reactor with the ammonia-alcohol solution and the reaction raw material containing Compound XV (3-methyl-3-pentenenitrile) and its isomer Compound XVI.
- Introduce hydrogen gas to maintain a pressure of 0.6-0.7 MPa, heat the system to 35-45°C, and stir for 6-9 hours to complete the reduction to Compound XVII.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this low-pressure hydrogenation technology translates into tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the infrastructure requirements. By eliminating the need for high-pressure (5-6 MPa) reactors, manufacturers can utilize more common, lower-cost equipment, which significantly lowers the barrier to entry for production and reduces capital depreciation costs. This flexibility allows for a more diversified supplier base, as more contract manufacturing organizations (CMOs) possess the capability to run low-pressure hydrogenations compared to specialized high-pressure facilities. Furthermore, the reduction in reaction time from roughly 40 hours to under 9 hours dramatically increases asset turnover. This means that the same reactor volume can produce significantly more batches per month, effectively expanding capacity without the need for new construction. This enhanced throughput is critical for meeting fluctuating market demands for opioid analgesics and ensures a more resilient supply chain capable of absorbing demand shocks.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, driven primarily by the optimization of input materials and energy consumption. The reduction in catalyst loading from a ratio of >1.2 to approximately 0.6-0.8 represents a direct saving in one of the more expensive reagents in the process. Additionally, the ability to bypass the purification of the starting nitrile (Compound XV) eliminates an entire unit operation, saving on solvents, energy for distillation, and labor. The lower operating pressure and temperature also result in significantly lower energy costs for compression and heating compared to the energy-intensive conventional methods. These cumulative efficiencies lead to a substantially lower cost of goods sold (COGS), providing a competitive pricing advantage in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Safety is a cornerstone of supply chain continuity. High-pressure hydrogenation carries inherent risks of leaks and catastrophic failure, which can lead to unplanned plant shutdowns and regulatory scrutiny. By operating at near-atmospheric pressures (0.6-0.7 MPa), the risk profile is minimized, leading to more predictable production schedules and fewer interruptions due to safety incidents. Moreover, the use of common solvents like ethanol and standard catalysts like Raney nickel ensures that raw material sourcing is robust and less susceptible to geopolitical or logistical disruptions. The shorter cycle time also means that inventory levels can be kept leaner, reducing working capital tied up in work-in-progress stock and allowing for a more agile response to customer orders.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, particularly with exothermic reactions and high-pressure systems. The mild conditions of this novel method make it inherently easier to scale from pilot plants to multi-ton commercial production without significant re-engineering. The reduced catalyst usage also has a positive environmental impact, generating less hazardous solid waste that requires disposal. The process aligns well with green chemistry principles by improving atom economy (through higher selectivity) and reducing energy intensity. For companies facing increasing pressure to reduce their carbon footprint and adhere to strict environmental regulations, this technology offers a pathway to sustainable manufacturing that satisfies both regulatory bodies and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits and chemical nuances for potential partners and technical stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your existing supply chain or R&D pipeline.
Q: How does the new ammonia-alcohol method improve safety compared to conventional high-pressure hydrogenation?
A: The novel method operates at a significantly lower hydrogen pressure of 0.6-0.7 MPa compared to the conventional 5-6 MPa, drastically reducing the risk associated with high-pressure vessel operations and improving overall plant safety.
Q: What is the impact of ammonia concentration on the reaction efficiency and catalyst usage?
A: Maintaining an ammonia concentration between 5% and 30% (optimally 12-14%) prevents the formation of excessive by-products and avoids catalyst poisoning, allowing for a reduced loading of Raney nickel (0.6-0.8:1 ratio) while maintaining high conversion rates.
Q: Can the crude product mixture be used directly in subsequent pentazocine synthesis steps?
A: Yes, the process yields a mixture of Compound XVII and Compound XVIII which can be directly utilized in the subsequent cyclization reaction to form the key pentazocine intermediate (Compound 10) without the need for complex purification, streamlining the workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pentazocine Intermediate Supplier
The technological advancements described in patent CN112679363A underscore the evolving landscape of opioid intermediate synthesis, where safety and efficiency are paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such innovations to the global market. Our state-of-the-art facilities are equipped to handle the specific requirements of low-pressure hydrogenation and sensitive amine chemistry, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process excellence, we guarantee the consistent quality and reliability that top-tier pharmaceutical companies demand. We understand that the transition to a new supplier requires confidence in both technical capability and regulatory compliance, and we are dedicated to providing that assurance through transparent communication and validated processes.
We invite you to explore how our optimized manufacturing capabilities can enhance your supply chain resilience and cost structure. Our technical team is prepared to discuss the specifics of this pentazocine intermediate route and how it aligns with your project goals. Please contact our Customized Cost-Saving Analysis team today to request specific COA data and route feasibility assessments. Let us partner with you to accelerate your development timelines and secure a stable, high-quality supply of this critical pharmaceutical building block.
