Advanced Manufacturing of Pentazocine Intermediates: A Safe and Scalable Synthetic Route
Advanced Manufacturing of Pentazocine Intermediates: A Safe and Scalable Synthetic Route
The pharmaceutical landscape for opioid analgesics continues to evolve, demanding higher standards of safety and efficiency in the production of active pharmaceutical ingredients (APIs) and their precursors. Patent CN109438351B introduces a groundbreaking preparation method for a critical pentazocine intermediate, specifically addressing the longstanding challenges of low yield and hazardous processing conditions inherent in legacy synthetic routes. Pentazocine, a benzomorphan derivative acting as a kappa-opioid receptor agonist, requires precise stereochemical control and high purity to ensure therapeutic efficacy and patient safety. This new technology offers a robust solution by replacing dangerous high-pressure hydrogenation and explosive reagents with a streamlined, ten-step sequence that operates under mild conditions. By leveraging accessible starting materials and optimizing reaction parameters, this method not only enhances the safety profile of the manufacturing process but also significantly improves the economic viability of producing high-quality pharmaceutical intermediates for the global market.
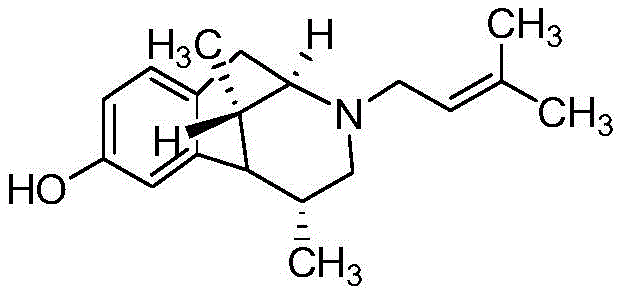
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of pentazocine and its intermediates has been plagued by significant safety hazards and operational inefficiencies that hinder scalable production. Traditional routes often necessitate the use of highly reactive and dangerous reagents, such as metallic sodium and n-butyllithium, which pose severe fire and explosion risks in a plant environment. Furthermore, earlier methodologies frequently relied on high-pressure hydrogenation steps, typically requiring pressures of 10-15 kg, to reduce key intermediates, demanding specialized and expensive reactor infrastructure. Some asymmetric synthesis approaches utilized chiral catalysts that were not only difficult to prepare but also resulted in mixtures of isomers, complicating downstream purification. Additionally, certain pathways involved explosive starting materials like 2,4-dinitroaniline, creating unacceptable risks for large-scale operations. These factors collectively contributed to low overall yields, often hovering around 25-30% for critical intermediates, and generated substantial chemical waste, driving up both production costs and environmental compliance burdens for manufacturers.
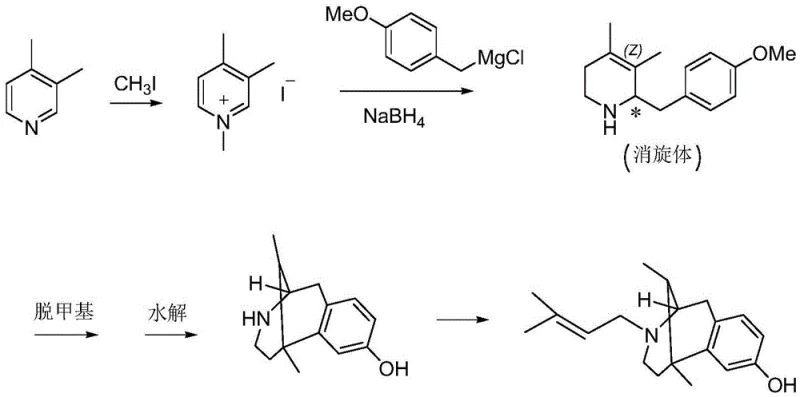
The Novel Approach
In stark contrast to these cumbersome legacy processes, the method disclosed in patent CN109438351B presents a sophisticated yet practical alternative that prioritizes safety and yield without compromising on chemical complexity. This innovative route circumvents the need for high-pressure equipment and explosive reagents entirely, utilizing a series of controlled organic transformations that are inherently safer and easier to manage. The process achieves a remarkable purity of over 97% for intermediate Compound 9 and ultimately delivers the final intermediate, Compound 11, with a purity exceeding 98%. By employing common reagents such as sodium borohydride for reduction and avoiding exotic chiral catalysts, the method drastically simplifies the supply chain requirements. The ten-step sequence is designed for high atom economy and operational simplicity, allowing for straightforward workup procedures like distillation and crystallization. This shift from hazardous, low-yield chemistry to a mild, high-efficiency protocol represents a paradigm shift in the manufacturing of opioid analgesic intermediates, offering a clear path toward cost reduction and enhanced supply chain reliability.
Mechanistic Insights into the Ten-Step Synthetic Pathway
The core of this technological advancement lies in its meticulously orchestrated ten-step reaction sequence, which transforms simple starting materials into the complex pentazocine scaffold with high fidelity. The synthesis initiates with a methylation reaction where Compound 1 is treated with methyl iodide and potassium carbonate in an acetone medium at 50°C, establishing the necessary carbon framework. This is followed by a critical ring-opening step using concentrated hydrochloric acid, which prepares the molecule for subsequent functionalization. A key feature of this route is the selective reduction using sodium borohydride at controlled low temperatures of 0-5°C, which effectively reduces the ketone functionality while preserving other sensitive groups, a significant improvement over harsher reducing agents. The process then employs a protection strategy using trimethylchlorosilane (TMSCl) to mask the hydroxyl group, ensuring chemoselectivity during the subsequent nucleophilic substitution with potassium phthalimide. This substitution introduces the nitrogen atom required for the final heterocyclic ring, setting the stage for the construction of the benzazocine core.
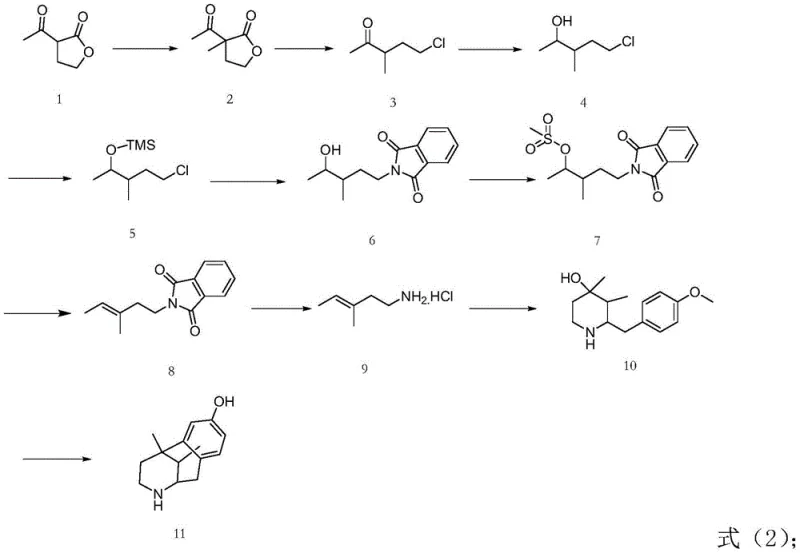
Following the introduction of the nitrogen moiety, the synthesis proceeds through an esterification and elimination sequence to install the crucial prenyl side chain. The hydroxyl group is activated via mesylation using methylsulfonyl chloride, followed by an elimination reaction mediated by potassium carbonate and lithium bromide at 105°C to generate the alkene functionality found in Compound 8. The phthalimide protecting group is then removed via hydrazinolysis in ethanol under reflux, liberating the primary amine as Compound 9 with exceptional purity. The cyclization step involves reacting this amine with a specific epoxide derivative (Compound 9a) under slightly acidic conditions to close the piperidine ring, forming Compound 10. The final transformation is a condensation-demethylation reaction using 48% hydrobromic acid at elevated temperatures (140°C), which simultaneously removes the methyl ether protecting group and finalizes the structure of the target intermediate, Compound 11. Each step is optimized for yield and purity, minimizing the formation of byproducts and ensuring a robust impurity profile suitable for pharmaceutical applications.
How to Synthesize Pentazocine Intermediate Efficiently
Implementing this synthesis requires strict adherence to the specified reaction conditions and stoichiometry to maximize yield and safety. The process begins with the methylation of the starting ketone, followed by acid-mediated ring opening and careful reduction. Detailed standard operating procedures for each of the ten steps, including specific solvent volumes, temperature ramping rates, and quenching protocols, are essential for reproducibility. For instance, the reduction step must be maintained between 0-5°C to prevent side reactions, while the final demethylation requires sustained heating at 140°C for 20 hours to ensure complete conversion. The purification of intermediates, particularly the crystallization of Compound 10 and the final extraction of Compound 11, plays a vital role in achieving the reported 98% purity. Operators should utilize the detailed experimental examples provided in the patent as a baseline for scaling, paying close attention to the molar ratios of reagents such as the 1:1.3 ratio of starting material to methyl iodide.
- Perform methylation of Compound 1 using methyl iodide and potassium carbonate in acetone at 50°C.
- Execute ring opening with concentrated hydrochloric acid followed by sodium borohydride reduction at 0-5°C.
- Protect the hydroxyl group with TMSCl, substitute with phthalimide, mesylate, and eliminate to form the alkene chain.
- Conduct hydrazinolysis to free the amine, cyclize with epoxide Compound 9a, and finalize with HBr demethylation at 140°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling strategic advantages that extend beyond mere technical feasibility. The elimination of high-pressure hydrogenation and explosive reagents fundamentally alters the risk profile of the manufacturing facility, potentially lowering insurance premiums and reducing the need for specialized containment infrastructure. By relying on commodity chemicals like acetone, ethanol, and sodium borohydride, the process insulates the supply chain from the volatility associated with sourcing exotic catalysts or hazardous specialty gases. The high yields reported at each step, particularly the near-quantitative conversion in the hydrazinolysis and mesylation steps, translate directly into reduced raw material consumption and lower waste disposal costs. Furthermore, the simplified purification protocols, which favor crystallization and standard extraction over complex chromatography, enable faster batch turnover times. This efficiency allows manufacturers to respond more agilely to market demand fluctuations, ensuring a steady flow of high-purity intermediates to downstream API producers without the bottlenecks typical of older, more cumbersome synthetic methods.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive and dangerous reagents with cost-effective alternatives. By avoiding the use of precious metal catalysts and high-pressure equipment, capital expenditure (CAPEX) and operational expenditure (OPEX) are significantly reduced. The high atom economy and minimal waste generation further contribute to a leaner cost structure, making the final intermediate more price-competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the route depends on widely available bulk chemicals rather than niche reagents with long lead times. The robustness of the reaction conditions means that production is less susceptible to interruptions caused by equipment failure or stringent safety shutdowns. This reliability ensures consistent delivery schedules for pharmaceutical clients who depend on uninterrupted supplies of critical pain management intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to kilogram scales in the patent examples. The absence of heavy metals and explosive byproducts simplifies wastewater treatment and aligns with increasingly strict environmental regulations. This 'green chemistry' aspect not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis. Understanding these details is crucial for R&D teams evaluating the technology for technology transfer and for procurement officers assessing supplier capabilities. The answers are derived directly from the experimental data and technical specifications outlined in patent CN109438351B, providing a factual basis for decision-making.
Q: How does this new process improve safety compared to traditional pentazocine synthesis?
A: Traditional methods often rely on hazardous reagents like metallic sodium, n-butyllithium, or explosive substances such as 2,4-dinitroaniline, and require high-pressure hydrogenation (10-15 kg). This patented route eliminates these dangers by using mild reagents like sodium borohydride and atmospheric pressure conditions, significantly reducing operational risk.
Q: What purity levels can be achieved with this synthetic method?
A: The process is designed for high purity, achieving over 97% purity for key intermediate Compound 9 and reaching 98.28% purity for the final intermediate Compound 11. This is a substantial improvement over conventional methods which often struggle to exceed 25-30% purity for similar intermediates without extensive purification.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the route is specifically optimized for scalability. It avoids complex chiral catalysts that are difficult to prepare and removes the need for high-pressure equipment. The use of cheap, readily available raw materials and simple unit operations like distillation and crystallization makes it highly viable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pentazocine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of complex pharmaceutical intermediates like the pentazocine precursor described in CN109438351B. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory-scale methodology into a robust commercial reality. Our facilities are equipped to handle diverse synthetic pathways, ranging from early-stage process development to full-scale production, with a proven track record of scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of intermediate meets the exacting standards required for API synthesis, guaranteeing consistency and safety for your final drug product.
We invite you to collaborate with us to leverage this advanced synthetic route for your pentazocine supply chain. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this safer, high-yield process can optimize your overall manufacturing budget. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical solution available.
