Revolutionizing Dexmedetomidine Production: A Safer, Scalable Synthetic Route for Global Pharma Supply Chains
The pharmaceutical landscape for sedatives and anesthetics has long relied on the efficient production of alpha-2 adrenoreceptor agonists, with dexmedetomidine hydrochloride standing out as a critical active pharmaceutical ingredient (API) for intensive care and surgical anesthesia. As detailed in the recent patent CN111217756B, a groundbreaking preparation method has been disclosed that fundamentally alters the synthetic strategy for this high-value compound. This novel approach addresses the longstanding safety and scalability issues inherent in traditional manufacturing processes by replacing hazardous high-pressure hydrogenation and complex protection-deprotection sequences with a streamlined, robust chemical pathway. For global procurement leaders and R&D directors, this patent represents a pivotal shift towards safer, more cost-effective production capabilities that align with modern green chemistry principles and stringent industrial safety regulations. The methodology leverages accessible starting materials and avoids the use of exotic catalysts, thereby enhancing the reliability of the supply chain for this essential medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dexmedetomidine hydrochloride has been plagued by significant technical hurdles that compromise both economic viability and operational safety. Traditional routes, such as those involving Friedel-Crafts alkylation followed by chiral resolution, often suffer from low overall yields and difficult purification profiles due to the formation of regioisomers. Furthermore, alternative pathways reported in literature frequently rely on the use of trityl protecting groups, which necessitate additional synthetic steps for installation and removal, drastically increasing the consumption of reagents and solvents while generating substantial chemical waste. Perhaps most critically, several established methods depend on catalytic hydrogenation using palladium on carbon under high pressure, a process that introduces severe safety risks in a manufacturing environment, including the potential for runaway reactions and the handling of pyrophoric catalysts. These conventional strategies also often employ super-strong bases like alkyl lithium, which require cryogenic conditions and inert atmospheres, adding layers of complexity and cost that are unsustainable for large-scale commercial production.
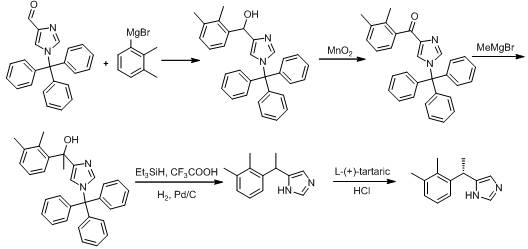
The Novel Approach
In stark contrast to these cumbersome legacy methods, the process outlined in patent CN111217756B introduces a remarkably direct and safe synthetic trajectory that bypasses the need for hazardous reagents and complex protecting group chemistry. This innovative route initiates with the formation of a Grignard reagent from 2,3-dimethyl-chloroethylbenzene, which is subsequently reacted with formaldehyde to construct the carbon skeleton efficiently. The subsequent oxidation to the aldehyde and conversion to an imine sets the stage for a highly selective Van Leusen reaction, which constructs the imidazole ring with precision without the need for transition metal catalysts. By eliminating the reliance on high-pressure hydrogenation and alkyl lithium deprotonation, this method significantly lowers the barrier to entry for safe manufacturing, making it an ideal candidate for facilities prioritizing process safety management (PSM). The simplicity of the reaction conditions, often proceeding at moderate temperatures and atmospheric pressure, ensures that the process is not only safer but also easier to control and scale, offering a distinct competitive advantage in the production of high-purity API intermediates.
Mechanistic Insights into Van Leusen Imidazole Synthesis
The core of this technological breakthrough lies in the strategic application of the Van Leusen reaction for the construction of the imidazole heterocycle, a transformation that offers superior regioselectivity compared to traditional cyclization methods. Mechanistically, this process involves the deprotonation of p-toluenesulfonylmethyl isonitrile (TosMIC) by a mild base such as potassium carbonate to generate a nucleophilic carbanion. This species then attacks the electrophilic carbon of the pre-formed imine intermediate, derived from the 2-(2,3-dimethylphenyl)propanal, initiating a cascade that leads to ring closure and the expulsion of the sulfonyl group. This elegant mechanism avoids the harsh conditions typically associated with imidazole synthesis, such as high-temperature condensation reactions that often lead to polymerization or tar formation. Furthermore, the use of an imine substrate rather than a ketone or aldehyde directly in the cyclization step allows for better control over the reaction kinetics, minimizing the formation of side products and ensuring a cleaner reaction profile that simplifies downstream processing.
Impurity control is another critical aspect where this mechanistic design excels, particularly in the management of chiral purity and related substances. The oxidative step converting the alcohol to the aldehyde is carefully managed using potassium dichromate under controlled boiling conditions, which prevents over-oxidation to the corresponding carboxylic acid, a common impurity that can be difficult to remove. Additionally, the final chiral resolution step utilizing L-(+)-tartaric acid is optimized through iterative recrystallization, effectively enriching the desired dextrorotatory enantiomer while rejecting the levo-isomer. This rigorous control over stereochemistry is paramount for meeting the stringent pharmacopoeial standards required for dexmedetomidine hydrochloride, ensuring that the final product possesses the necessary biological activity and safety profile for clinical use without the burden of toxic heavy metal residues often found in catalytic routes.
How to Synthesize Dexmedetomidine Hydrochloride Efficiently
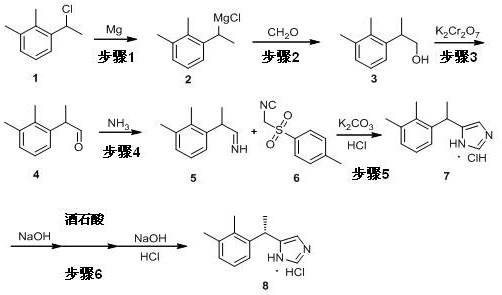
Implementing this synthesis requires precise adherence to the reaction parameters defined in the patent to maximize yield and safety. The process begins with the careful preparation of the Grignard reagent in anhydrous conditions, followed by the controlled introduction of formaldehyde gas to build the hydroxy-ethyl side chain. Subsequent oxidation and imine formation steps must be monitored closely to ensure complete conversion before proceeding to the critical Van Leusen cyclization. For a comprehensive understanding of the specific molar ratios, temperature profiles, and workup procedures required to replicate this high-efficiency pathway, please refer to the standardized technical guide below.
- Preparation of Grignard Reagent and Alcohol Intermediate: React 2,3-dimethyl-chloroethylbenzene with magnesium in dry THF, followed by the addition of formaldehyde gas to yield 2-(2,3-dimethylphenyl)propanol.
- Oxidation and Imine Formation: Oxidize the alcohol intermediate to the corresponding aldehyde using potassium dichromate, then react with ammonia methanol solution to form the imine derivative.
- Cyclization and Resolution: Perform the Van Leusen reaction with p-toluenesulfonylmethyl isonitrile to form medetomidine, followed by chiral resolution with tartaric acid to obtain the final dexmedetomidine hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. By fundamentally redesigning the process to eliminate high-risk unit operations, manufacturers can significantly reduce the overhead costs associated with safety compliance, insurance, and specialized equipment maintenance. The removal of precious metal catalysts like palladium not only lowers the direct material cost but also eliminates the need for expensive and time-consuming metal scavenging steps, which are often a bottleneck in API production. Furthermore, the reliance on commodity chemicals such as formaldehyde, ammonia, and potassium carbonate ensures a robust and resilient supply chain that is less susceptible to the volatility of the specialty reagent market. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of trityl protecting groups and palladium catalysts results in a drastic simplification of the bill of materials, directly lowering the cost of goods sold (COGS). Without the need for expensive noble metals or complex protecting group chemistry, the process achieves substantial cost savings through reduced reagent consumption and simplified waste treatment protocols. The avoidance of high-pressure hydrogenation equipment further reduces capital expenditure (CAPEX) requirements, allowing for production in standard glass-lined or stainless steel reactors that are widely available in the fine chemical industry.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this route is significantly more straightforward, as the key reagents are bulk commodities with stable global availability. Unlike routes dependent on specialized organometallic reagents or custom-synthesized intermediates, this method utilizes widely traded chemicals, mitigating the risk of supply disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even when sourcing from diverse suppliers, which is a key factor in building a resilient supply chain.
- Scalability and Environmental Compliance: The process is inherently designed for scale, operating under mild conditions that facilitate easy heat transfer and mixing in large-scale reactors. The absence of hazardous high-pressure steps and pyrophoric reagents simplifies the regulatory approval process for new manufacturing sites, accelerating time-to-market. Additionally, the reduced generation of heavy metal waste and organic solvents aligns with increasingly strict environmental regulations, positioning manufacturers as responsible partners in the sustainable production of essential medicines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the feasibility and advantages of this approach for industrial application. Understanding these details is essential for technical teams evaluating the potential for technology transfer or process optimization.
Q: Why is the new synthetic route for dexmedetomidine considered safer than prior art?
A: The new route eliminates high-risk reactions such as catalytic hydrogenation under high pressure and the use of super-strong bases like alkyl lithium, significantly reducing industrial safety hazards and explosion risks.
Q: How does this method improve cost efficiency for large-scale manufacturing?
A: By avoiding expensive protecting groups like trityl and precious metal catalysts like palladium, the process reduces raw material costs and simplifies downstream purification, leading to substantial operational savings.
Q: What is the role of tartaric acid in the final step of this synthesis?
A: Tartaric acid is used for chiral resolution to separate the active dextrorotatory isomer (dexmedetomidine) from the racemic medetomidine mixture, ensuring high optical purity required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexmedetomidine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this safer, more efficient synthetic route for dexmedetomidine hydrochloride and are fully equipped to bring this technology to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant with cGMP standards. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of API intermediate meets the highest quality benchmarks, providing our partners with the confidence they need to secure their supply chains. Our commitment to technical excellence allows us to navigate the complexities of chiral resolution and impurity control with precision, delivering a product that is ready for formulation.
We invite forward-thinking pharmaceutical companies to collaborate with us to optimize their supply chains using this advanced manufacturing technology. By leveraging our expertise, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most reliable and cost-effective production strategy available in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
