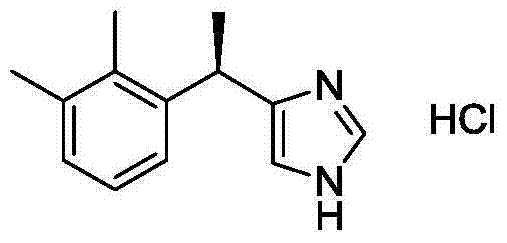
Optimized Synthetic Route for Dexmedetomidine Hydrochloride: Enhancing Purity and Commercial Scalability
Introduction to Advanced Dexmedetomidine Synthesis
The pharmaceutical industry constantly seeks more efficient pathways for producing critical sedative agents, and the synthesis of dexmedetomidine hydrochloride stands as a prime example of process optimization. Based on the technological breakthroughs detailed in patent CN103694175A, we observe a significant evolution in the manufacturing of this potent α2-adrenoceptor agonist. The disclosed method leverages a refined Friedel-Crafts alkylation strategy, utilizing specific Lewis acid catalysts to enhance the conversion of racemic medetomidine precursors. This approach not only addresses the historical challenges of low yields associated with earlier titanium-based catalytic systems but also integrates a robust chiral resolution step using (+)-di-p-toluoyl-tartaric acid. For R&D directors and procurement specialists, understanding this chemical architecture is vital, as it represents a shift towards higher efficiency and reduced waste generation in the production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
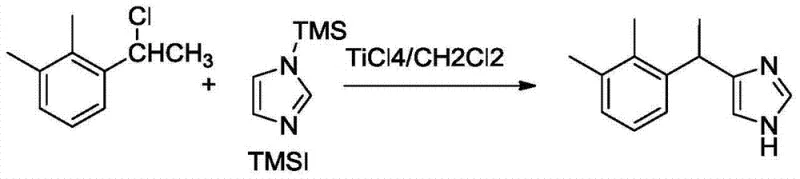
Historically, the synthesis of medetomidine, the racemic precursor to dexmedetomidine, relied heavily on titanium tetrachloride (TiCl4) mediated reactions in dichloromethane systems. While this traditional route, often involving 1-(1-chloroethyl)-2,3-dimethylbenzene and N-TMS-imidazole, was chemically feasible, it suffered from inherent inefficiencies that plagued large-scale manufacturing. The primary drawback was the suboptimal yield, which directly impacted the cost of goods sold and increased the environmental burden due to the need for extensive purification and waste treatment. Furthermore, the handling of TiCl4 requires stringent moisture control and specialized equipment, adding complexity to the supply chain and increasing the risk of batch-to-batch variability. These factors combined to create a bottleneck for manufacturers aiming to produce high-purity dexmedetomidine at a competitive price point, necessitating a search for more robust catalytic alternatives.
The Novel Approach
The innovative methodology presented in the patent data introduces a paradigm shift by screening various Lewis acids to identify a superior catalyst, ultimately selecting boron trifluoride diethyl etherate (BF3·OEt2). This novel approach maintains the core Friedel-Crafts alkylation logic but optimizes the electronic environment of the reaction to favor product formation significantly. By replacing the titanium catalyst with BF3·OEt2, the process achieves a marked improvement in yield while operating under similarly mild conditions, typically around 0°C in dichloromethane. This substitution simplifies the operational protocol and enhances the overall atom economy of the synthesis. Additionally, the integration of a specific resolution step using (+)-DDTA in an isopropanol-water mixture ensures that the final active pharmaceutical ingredient meets rigorous stereochemical specifications, thereby delivering a product that is both commercially viable and clinically effective.
Mechanistic Insights into BF3-Catalyzed Friedel-Crafts Alkylation
To fully appreciate the technical superiority of this route, one must delve into the mechanistic nuances of the Lewis acid-catalyzed alkylation. The reaction proceeds through the activation of the alkyl halide, specifically 1-(1-chloroethyl)-2,3-dimethylbenzene, by the Lewis acid catalyst. In the optimized system, BF3·OEt2 coordinates with the chlorine atom, facilitating the formation of a carbocation intermediate or a tight ion pair that is highly electrophilic. This activated species then attacks the nucleophilic C4 or C5 position of the trimethylsilyl (TMS) protected imidazole ring. The choice of solvent, dichloromethane, plays a critical role in stabilizing these ionic intermediates without participating in side reactions, which is a common pitfall in polar protic solvents. The subsequent hydrolysis step removes the TMS protecting group, revealing the free imidazole nitrogen and yielding the racemic medetomidine base. This mechanistic clarity allows process chemists to fine-tune reaction parameters such as temperature and addition rates to minimize impurities like regioisomers or polymerization byproducts.
Furthermore, the control of impurities extends beyond the alkylation step into the chiral resolution phase. The use of (+)-di-p-toluoyl-tartaric acid [(+)-DDTA] relies on the formation of diastereomeric salts between the resolving agent and the enantiomers of medetomidine. Due to the differences in solubility of these diastereomeric salts in the isopropanol-water solvent system, the desired (R)-enantiomer precipitates selectively while the (S)-enantiomer remains in the mother liquor. This thermodynamic control is essential for achieving the high optical purity required for clinical applications. The patent data indicates that optimizing the solvent ratio and temperature during crystallization is key to maximizing the recovery of the active isomer. Understanding these intermolecular interactions is crucial for scaling the process, as slight deviations in cooling rates or solvent composition can drastically affect the enantiomeric excess (ee) and overall yield of the final dexmedetomidine hydrochloride.
How to Synthesize Dexmedetomidine Hydrochloride Efficiently
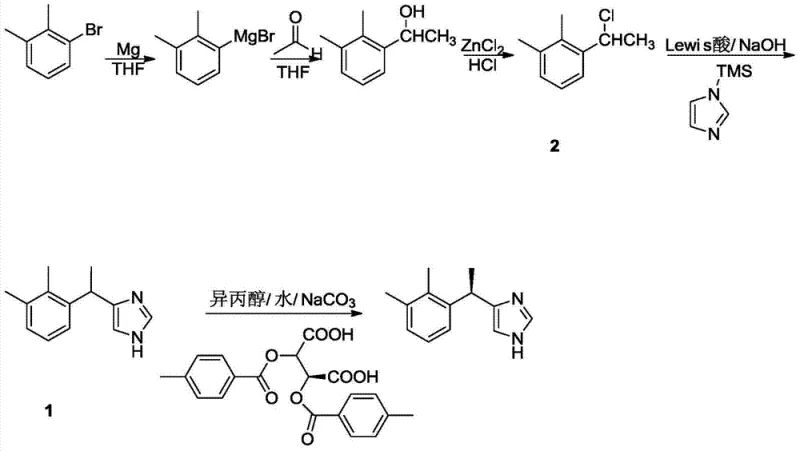
Implementing this synthesis requires a disciplined approach to unit operations, starting from the preparation of the key alkylating agent. The process begins with the formation of a Grignard reagent from 2,3-dimethylbromobenzene, followed by reaction with acetaldehyde to generate the secondary alcohol, which is subsequently converted to the chloride using zinc chloride and hydrochloric acid. This precursor is then subjected to the optimized Friedel-Crafts conditions described previously. The detailed标准化 synthesis steps见下方的指南 provide a roadmap for replicating these results in a pilot or production setting. Adhering to strict stoichiometric controls and temperature profiles during the alkylation and resolution stages is paramount to ensuring consistent quality. The following sections outline the critical operational parameters that define this efficient manufacturing pathway.
- Preparation of 1-(1-chloroethyl)-2,3-dimethylbenzene via Grignard reaction followed by chlorination with ZnCl2/HCl.
- Execution of Friedel-Crafts alkylation using TMS-imidazole and BF3·OEt2 catalyst in dichloromethane to form racemic medetomidine.
- Chiral resolution of racemic medetomidine using (+)-di-p-toluoyl-tartaric acid [(+)-DDTA] in isopropanol/water, followed by salification with HCl.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this optimized synthetic route offers tangible strategic benefits that extend beyond simple chemistry. The primary advantage lies in the substantial cost reduction in pharmaceutical intermediates manufacturing driven by improved process efficiency. By eliminating the reliance on less efficient titanium catalysts and adopting a higher-yielding boron-based system, manufacturers can significantly reduce the consumption of raw materials per kilogram of finished product. This improvement in atom economy translates directly into lower variable costs and reduced waste disposal expenses, which are increasingly significant in regulated markets. Moreover, the use of common, commercially available solvents like dichloromethane and isopropanol ensures that the supply chain remains resilient against fluctuations in specialty chemical availability, thereby enhancing overall supply security.
- Cost Reduction in Manufacturing: The adoption of BF3·OEt2 as a catalyst eliminates the need for expensive and difficult-to-handle titanium reagents, leading to a streamlined procurement process. The higher reaction yield means that less starting material is required to produce the same amount of active ingredient, effectively lowering the cost basis. Additionally, the simplified workup procedures reduce the demand for extensive purification resources, further driving down operational expenditures. These cumulative efficiencies allow for a more competitive pricing structure without compromising on the quality standards required for GMP production.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, such as 2,3-dimethylbromobenzene and imidazole derivatives, are commodity chemicals with robust global supply networks. This accessibility mitigates the risk of production delays caused by raw material shortages. Furthermore, the reaction conditions are mild and do not require exotic equipment or extreme temperatures, allowing for flexible manufacturing scheduling across multiple facilities. This flexibility is crucial for maintaining continuous supply to downstream formulation partners, ensuring that market demand for sedative medications is met without interruption.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton commercial production. The use of standard solvents facilitates easier recycling and recovery, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing site. The reduction in hazardous waste generation simplifies regulatory compliance and lowers the costs associated with environmental management. This sustainability profile is increasingly important for pharmaceutical companies aiming to meet corporate social responsibility goals while maintaining efficient production lines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of dexmedetomidine hydrochloride using this patented methodology. These insights are derived directly from the experimental data and process descriptions found in the source patent, providing a reliable foundation for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing portfolios. The answers below clarify the specific advantages of the Lewis acid selection and the resolution strategy employed.
Q: What is the primary advantage of using BF3·OEt2 over TiCl4 in this synthesis?
A: The use of BF3·OEt2 as a Lewis acid catalyst significantly improves the reaction yield compared to the traditional TiCl4 method, while maintaining milder reaction conditions that are easier to control on a large scale.
Q: How is the optical purity of dexmedetomidine ensured in this process?
A: Optical purity is achieved through a resolution step using (+)-di-p-toluoyl-tartaric acid [(+)-DDTA] in a specific isopropanol-water solvent system, which effectively separates the active dextrorotatory enantiomer from the racemic mixture.
Q: Is this synthetic route suitable for industrial scale-up?
A: Yes, the process utilizes common solvents like dichloromethane and THF, and avoids extremely harsh conditions, making it highly adaptable for commercial scale-up with robust quality control measures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexmedetomidine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to delivering high-purity dexmedetomidine hydrochloride that adheres to stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging the optimized Friedel-Crafts technology discussed herein, we can offer a product that balances cost-efficiency with the highest standards of pharmaceutical quality, making us an ideal partner for your long-term projects.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis can benefit your specific application. Request a Customized Cost-Saving Analysis today to understand the potential economic impact of switching to this superior manufacturing method. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless transition to a more efficient and reliable supply chain.
