Advanced Synthesis of Dexmedetomidine Hydrochloride for Commercial Scale API Manufacturing
Introduction to Next-Generation Dexmedetomidine Synthesis
The pharmaceutical landscape for sedative agents continues to evolve, driven by the demand for safer, more efficient manufacturing processes for critical care medications. Patent CN111548308A introduces a transformative synthesis process for dexmedetomidine hydrochloride, a potent alpha-2 adrenoceptor agonist widely used for intensive care sedation and procedural anesthesia. This novel methodology addresses longstanding challenges in chiral resolution and impurity control, offering a robust pathway for reliable API intermediate supplier networks seeking to optimize their production capabilities. By leveraging L-tartaric acid as a highly effective resolving agent and streamlining the salification steps, the invention achieves a finished product purity of greater than 99.8 percent with isomer content strictly controlled below 0.2 percent. This level of quality assurance is paramount for meeting the stringent regulatory requirements of global pharmacopoeias, ensuring that the final active pharmaceutical ingredient is safe for human administration without extensive downstream purification.
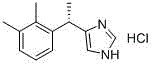
The strategic significance of this patent lies in its ability to bypass the complex and hazardous steps associated with legacy manufacturing routes. Traditional methods often rely on expensive protecting groups or high-risk catalytic hydrogenation, which introduce significant operational overhead and safety liabilities. In contrast, the disclosed process utilizes readily available starting materials such as 1-(1-chloroethyl)-2,3-dimethylbenzene and trimethylsilylimidazole, facilitating a more direct and cost-effective assembly of the molecular scaffold. For procurement managers and supply chain directors, this represents a pivotal shift towards more resilient sourcing strategies, reducing dependency on specialized reagents that are prone to market volatility. The integration of a one-step free salt formation method further simplifies the workflow, eliminating intermediate quality control bottlenecks and accelerating the time-to-market for generic formulations of this essential medicine.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing dexmedetomidine hydrochloride have been plagued by inefficiencies that hinder commercial scalability and economic viability. One prominent prior art route, detailed in patent US4443466, relies on a synthetic strategy that begins with 1-(2,3-dimethylphenyl)-1-(1-trityl-1H-imidazole-5-yl) ethanol. This pathway necessitates the use of trityl protecting groups, which are not only costly but also require additional synthetic steps for installation and subsequent removal, thereby lowering the overall atom economy of the process.
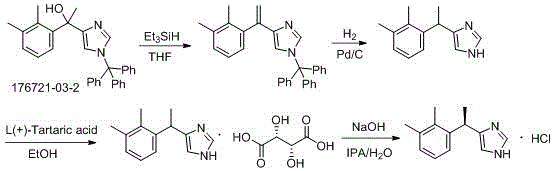
Furthermore, this conventional method involves a catalytic hydrogenation step to reduce the intermediate alcohol to the desired ethyl linkage. Hydrogenation reactions typically require high-pressure equipment and specialized catalysts, introducing significant safety risks related to explosive hazards and catalyst poisoning. The operational complexity of managing high-pressure reactors increases capital expenditure (CAPEX) and maintenance costs, making this route less attractive for manufacturers aiming to minimize production overhead. Additionally, the presence of the bulky trityl group can lead to steric hindrance issues during reaction steps, potentially resulting in lower yields and difficult purification profiles that compromise the final product's quality.
The Novel Approach
In stark contrast, the novel approach disclosed in CN111548308A eliminates the need for protecting groups and high-pressure hydrogenation entirely. The new synthesis initiates with a direct alkylation reaction between 1-(1-chloroethyl)-2,3-dimethylbenzene and trimethylsilylimidazole, mediated by titanium tetrachloride. This Friedel-Crafts-type alkylation is operationally simple, proceeding under mild conditions that do not require specialized high-pressure infrastructure. The elimination of the trityl group removes two entire synthetic steps (protection and deprotection), drastically shortening the production timeline and reducing the consumption of solvents and reagents.
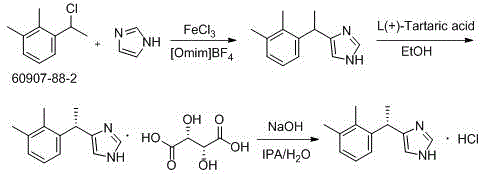
Another comparative route, referenced in patent CN106588778, utilizes ferric chloride (FeCl3) as a catalyst for the coupling of imidazole and the chloroethyl benzene derivative. While this method avoids hydrogenation, it suffers from significant post-treatment challenges. The removal of iron residues from the reaction mixture is notoriously difficult and often requires extensive washing and chelation steps, generating large volumes of acidic wastewater that complicate environmental compliance. Moreover, the lack of protection on the imidazole nitrogen in this route can lead to non-selective alkylation and polymerization side reactions, creating a complex impurity profile that is difficult to purge. The new process overcomes these issues by employing a silylated imidazole species which enhances nucleophilicity and selectivity, ensuring a cleaner reaction profile that simplifies downstream processing.
Mechanistic Insights into Chiral Resolution and Impurity Control
The cornerstone of this advanced synthesis is the chiral resolution step, which dictates the optical purity of the final dexmedetomidine hydrochloride. The process employs L-tartaric acid as the resolving agent, capitalizing on the formation of diastereomeric salts between the racemic medetomidine base and the chiral acid. The solubility differences between the (S)-enantiomer tartrate salt and the (R)-enantiomer tartrate salt in specific solvent systems allow for the selective crystallization of the desired dextrorotatory isomer. The patent specifies a critical molar equivalent ratio of medetomidine to L-tartaric acid ranging from 1:0.4 to 0.6. Maintaining this sub-stoichiometric ratio is essential; using an excess of the resolving agent could lead to the co-crystallization of the unwanted enantiomer or the formation of double salts that reduce optical purity. By carefully controlling this ratio, the process maximizes the yield of the desired isomer while minimizing the loss of material to the mother liquor.
Impurity control is further enhanced through a rigorous recrystallization protocol. The crude dexmedetomidine tartrate obtained from the initial resolution is subjected to repeated crystallization cycles, specifically two to three times, using a 90 percent ethanol and water mixture. This iterative purification leverages the thermodynamic stability of the crystal lattice to exclude structurally similar impurities and the opposite enantiomer. The choice of ethanol/water as the solvent system is particularly effective due to its polarity profile, which balances the solubility of the tartrate salt at elevated temperatures (75 degrees Celsius) against its low solubility at reduced temperatures (5 to 15 degrees Celsius). This thermal gradient drives the precipitation of high-purity crystals, effectively scrubbing away trace organic impurities and residual solvents. The result is a solid intermediate with exceptional stereochemical integrity, which serves as a robust foundation for the subsequent salification step, ensuring the final API meets the strict isomer limit of not more than 0.2 percent.
How to Synthesize Dexmedetomidine Hydrochloride Efficiently
The implementation of this synthesis route requires precise adherence to the four-stage protocol outlined in the patent to ensure reproducibility and quality. The process begins with the formation of the racemic medetomidine backbone, followed by the critical resolution, dissociation, and final salt formation. Each stage has been optimized to balance reaction kinetics with purification efficiency, allowing for a seamless transition from laboratory scale to industrial manufacturing. Operators must pay close attention to temperature controls during the crystallization phases and pH adjustments during the extraction steps to maintain the integrity of the chiral center. The following guide summarizes the standardized operational procedure derived from the patent examples, providing a clear roadmap for technical teams aiming to adopt this superior methodology.
- Synthesize medetomidine via alkylation of 1-(1-chloroethyl)-2,3-dimethylbenzene with trimethylsilylimidazole using titanium tetrachloride.
- Perform chiral resolution by reacting medetomidine with L-tartaric acid in ethanol, followed by repeated recrystallization to isolate dexmedetomidine tartrate.
- Dissociate the tartrate salt using sodium hydroxide and extract the free base with organic solvents like dichloromethane.
- Convert the free base to the final hydrochloride salt using ethanolic HCl and crystallize using methyl tert-butyl ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis process offers substantial strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for trityl-protected intermediates and specialized hydrogenation catalysts, manufacturers can source starting materials from a broader base of commodity chemical suppliers. This diversification reduces the risk of supply disruptions caused by the monopoly of single-source vendors for exotic reagents. Furthermore, the removal of high-pressure hydrogenation steps significantly lowers the barrier to entry for contract manufacturing organizations (CMOs) that may lack specialized high-pressure reactor infrastructure, thereby expanding the pool of potential manufacturing partners and increasing competition which drives down costs.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the reduction in unit operations and reagent costs. The elimination of the trityl protection and deprotection sequence removes two distinct reaction steps, along with the associated solvent usage, labor hours, and waste disposal costs. Additionally, the avoidance of noble metal catalysts required for hydrogenation removes a significant line item from the bill of materials. From a qualitative perspective, the simplified workup procedures, which rely on standard crystallization rather than complex chromatography or extensive aqueous washes to remove metal residues, translate directly into higher throughput and lower utility consumption. These efficiencies compound to deliver a significantly reduced cost of goods sold (COGS), enabling more competitive pricing for the final API in the generic marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, stable intermediates that are less prone to degradation during storage and transport. The key intermediate, dexmedetomidine tartrate, is a stable solid that can be stockpiled effectively, acting as a buffer against demand fluctuations. Unlike liquid intermediates or those requiring cold chain logistics, this solid salt simplifies warehousing requirements. Moreover, the process's tolerance for variation in reaction conditions, evidenced by the broad temperature ranges acceptable for crystallization, ensures consistent output even when minor fluctuations in utility supplies occur. This reliability is crucial for maintaining continuous production schedules and meeting just-in-time delivery commitments to downstream formulation partners without the risk of batch failures.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial tonnage is straightforward due to the absence of exothermic hazards associated with hydrogenation and the use of common organic solvents like ethanol and dichloromethane. The waste profile is markedly cleaner; without iron catalysts or heavy metal residues, the wastewater treatment load is significantly lighter, facilitating easier compliance with increasingly stringent environmental regulations. The ability to recycle solvents such as ethanol and methyl tert-butyl ether further enhances the sustainability profile of the manufacturing process. This alignment with green chemistry principles not only reduces environmental liability but also appeals to pharmaceutical customers who are increasingly prioritizing suppliers with strong environmental, social, and governance (ESG) credentials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing route.
Q: How does the new process improve chiral purity compared to traditional methods?
A: The process utilizes L-tartaric acid as a resolving agent with optimized molar ratios (1:0.4 to 0.6) and multiple recrystallization steps, achieving isomer levels below 0.2% and overall purity exceeding 99.8%.
Q: What are the safety advantages of this synthesis route over hydrogenation methods?
A: Unlike prior art routes requiring high-pressure hydrogenation which pose explosion risks, this method relies on ambient pressure alkylation and crystallization, significantly enhancing operational safety for commercial plants.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method avoids expensive protecting groups and complex chromatographic purifications, utilizing simple crystallization and extraction techniques that are easily scalable from pilot to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexmedetomidine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthesis route requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our facilities are equipped with state-of-the-art crystallization and filtration units capable of handling the specific solvent systems required for the L-tartaric acid resolution. We maintain stringent purity specifications across all our production lines, supported by rigorous QC labs that utilize advanced HPLC and chiral chromatography to verify that every batch of dexmedetomidine hydrochloride meets the >99.8 percent purity benchmark before release.
We invite pharmaceutical companies and generic manufacturers to collaborate with us to leverage this advanced technology for their supply chains. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this process can optimize your margins. We encourage you to contact our technical procurement team to request specific COA data from our pilot batches and comprehensive route feasibility assessments. By partnering with us, you secure a supply of high-purity dexmedetomidine hydrochloride that is not only cost-effective but also produced with the highest standards of quality and regulatory compliance.
