Advanced Manufacturing of High-Purity Cilnidipine: Overcoming Hydrolysis Impurities for Global Pharma Supply
Advanced Manufacturing of High-Purity Cilnidipine: Overcoming Hydrolysis Impurities for Global Pharma Supply
The global demand for potent calcium channel blockers continues to rise, driven by the increasing prevalence of hypertension and the need for medications with superior safety profiles. In this context, Patent CN111072552B introduces a transformative preparation method for Cilnidipine, a dual L/N-type calcium antagonist that offers distinct clinical advantages over traditional dihydropyridines. This technical disclosure addresses a persistent bottleneck in the pharmaceutical industry: the difficulty of removing specific hydrolysis impurities that arise during the synthesis of complex dihydropyridine esters. By integrating a critical intermediate purification step prior to the final cyclization, this methodology ensures that the resulting active pharmaceutical ingredient (API) meets stringent international quality standards. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic advantages of this route is essential for securing a stable, high-quality supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Cilnidipine often suffer from significant yield losses and purity issues stemming from the accumulation of acidic byproducts. In standard Hantzsch pyridine synthesis protocols, the initial condensation of methoxyethyl acetoacetate with m-nitrobenzaldehyde is typically catalyzed by strong acids like sulfuric acid. When this crude intermediate is carried forward directly to the cyclization step without adequate purification, residual acid remains in the reaction matrix. During the subsequent high-temperature reflux required for ring closure, this acidic environment catalyzes the hydrolysis of the sensitive ester moieties within the forming Cilnidipine molecule. This side reaction generates structural analogs that are chemically similar to the target product, making them exceptionally difficult to separate via standard recrystallization techniques. Consequently, manufacturers face reduced overall yields, often hovering around 60%, and struggle to achieve the >99% purity required for regulatory approval without resorting to costly and time-consuming chromatographic separations.
The Novel Approach
The innovative strategy outlined in Patent CN111072552B fundamentally alters this trajectory by inserting a dedicated purification stage for the 2-(3-nitrobenzylidene) methoxyethyl acetoacetate intermediate. Instead of proceeding with the crude condensate, the process dictates dissolving the intermediate in a water-insoluble organic solvent, such as dichloromethane or ethyl acetate, and washing it with an alkaline solution like sodium bicarbonate. This simple yet effective unit operation neutralizes and extracts the residual sulfuric acid and acetic acid generated in the first step. By eliminating the acidic catalyst before the thermal stress of the cyclization reaction, the pathway effectively shuts down the mechanism responsible for ester hydrolysis. The result is a dramatic improvement in the quality of the final crude product, simplifying the downstream refining process and enabling the consistent production of high-purity Cilnidipine with yields reaching up to 70%. 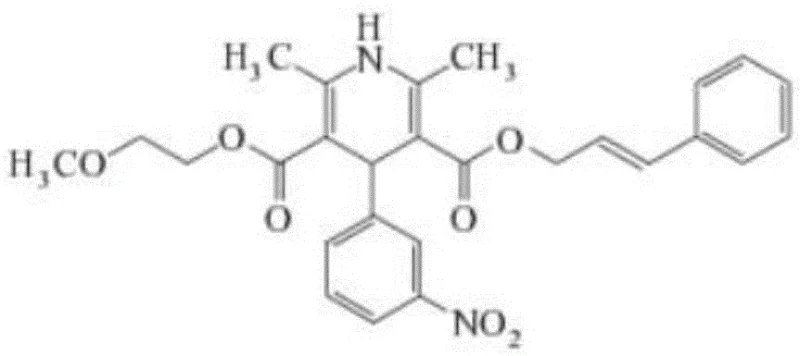
Mechanistic Insights into Acid-Catalyzed Ester Hydrolysis and Suppression
To fully appreciate the technical sophistication of this synthesis, one must examine the vulnerability of the dihydropyridine scaffold under acidic thermal conditions. The Cilnidipine molecule contains two distinct ester functionalities: a methoxyethyl ester and a cinnamyl ester. While the Hantzsch condensation requires thermal energy to drive the formation of the 1,4-dihydropyridine ring, this same energy, when coupled with proton donors (residual H2SO4 or HCl), activates the carbonyl oxygen of the ester groups towards nucleophilic attack by water. Even trace amounts of water generated during the condensation or present in the solvent can become aggressive nucleophiles in an acidic medium. This leads to the cleavage of the ester bond, converting the lipophilic prodrug-like structure into a more polar carboxylic acid derivative. These hydrolysis byproducts not only reduce the assay of the final API but also complicate the toxicological profile, necessitating rigorous control strategies that go beyond simple recrystallization.
The implementation of the alkaline wash acts as a chemical 'firewall' against this degradation pathway. By treating the organic phase containing the benzylidene intermediate with a mild base such as sodium bicarbonate, the process ensures that the pH of the reaction mixture entering the cyclization vessel is neutral. This suppression of acidity preserves the integrity of the ester bonds during the 22-hour reflux period in absolute ethanol. Furthermore, the use of absolute ethanol as the solvent for the cyclization step minimizes the availability of water, providing a dual-defense mechanism against hydrolysis. This mechanistic control allows the reaction to proceed to completion with minimal side reactions, resulting in a crude product that is already of high purity. For process chemists, this represents a shift from 'purification-heavy' workflows to 'prevention-first' synthesis design, significantly reducing the burden on analytical and separation teams.
How to Synthesize High-Purity Cilnidipine Efficiently
The execution of this synthesis requires precise control over stoichiometry and phase separation to maximize the benefits of the intermediate purification. The process begins with the condensation of methoxyethyl acetoacetate and m-nitrobenzaldehyde in acetic anhydride, followed immediately by the critical extraction sequence. Operators must ensure thorough mixing during the alkaline wash to guarantee complete acid removal, as pockets of unneutralized acid could still trigger localized hydrolysis. Following the isolation of the purified intermediate, the cyclization with 3-aminocrotonic acid cinnamyl ester is conducted under reflux, leveraging the high boiling point of ethanol to drive the equilibrium toward the dihydropyridine product. The detailed standardized operating procedures, including specific temperature ramps, stirring rates, and filtration parameters required to replicate these results on a multi-kilogram scale, are outlined in the technical guide below.
- Condense methoxyethyl acetoacetate with m-nitrobenzaldehyde using acetic anhydride and sulfuric acid catalysis to form the crude benzylidene intermediate.
- Purify the crude intermediate by dissolving in an organic solvent (e.g., dichloromethane) and washing with an alkali solution (e.g., sodium bicarbonate) to remove acid residues.
- React the purified intermediate with 3-aminocrotonic acid cinnamyl ester in absolute ethanol under reflux to form the dihydropyridine ring, followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this refined synthesis route offers substantial strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the downstream processing train. By preventing the formation of hydrolysis impurities at the source, manufacturers eliminate the need for complex, yield-eroding purification steps such as preparative HPLC or multiple iterative recrystallizations that are often required to salvage batches produced via conventional methods. This streamlining translates directly into reduced manufacturing cycle times and lower consumption of solvents and energy. Furthermore, the robustness of the process, which relies on commodity chemicals like ethanol and dichloromethane rather than exotic catalysts or inert gas protections, enhances supply chain resilience. Sourcing raw materials becomes more predictable, and the risk of batch failure due to sensitive reaction conditions is significantly mitigated, ensuring a steady flow of material for formulation.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of yield losses associated with impurity removal. In traditional routes, the presence of hydrolysis byproducts often forces manufacturers to sacrifice a significant portion of the batch to achieve specification compliance. By maintaining the structural integrity of the ester groups throughout the synthesis, this method maximizes the conversion of expensive starting materials, such as 3-aminocrotonic acid cinnamyl ester, into the final API. Additionally, the avoidance of transition metal catalysts or specialized reagents reduces the raw material cost base. The simplified workup procedure also lowers the operational expenditure related to waste disposal and solvent recovery, contributing to a leaner and more cost-competitive manufacturing model for high-purity Cilnidipine.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by processes that operate near the edge of their control limits. The conventional synthesis of Cilnidipine, with its sensitivity to acid residue and moisture, poses a risk of variable batch quality. The new method introduces a robust buffering step (the alkaline wash) that absorbs variations in the upstream condensation reaction, ensuring that the input to the cyclization step is consistently within specification. This consistency reduces the frequency of out-of-specification (OOS) investigations and batch rejections. Moreover, the use of standard industrial solvents and equipment means that production can be easily transferred between different manufacturing sites without requiring specialized infrastructure, thereby diversifying supply risk and ensuring reliable pharmaceutical intermediates supplier performance even during market fluctuations.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies heat transfer and mixing challenges, but this route is designed with scalability in mind. The exothermic nature of the initial condensation is managed by controlled addition at low temperatures, a parameter easily maintained in large reactors. The subsequent steps operate at atmospheric pressure and moderate temperatures, reducing the safety risks associated with high-pressure hydrogenation or cryogenic reactions. From an environmental perspective, the process avoids the generation of heavy metal waste streams, simplifying effluent treatment. The high selectivity of the reaction means less chemical waste is generated per kilogram of product, aligning with green chemistry principles and facilitating easier regulatory compliance in jurisdictions with strict environmental discharge limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity synthesis route. These insights are derived directly from the comparative data and experimental embodiments found in the patent literature, providing a transparent view of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer or assessing the quality parity of potential suppliers. The answers reflect the consensus on how intermediate purification impacts the final impurity profile and overall process economics.
Q: Why is the intermediate purification step critical in Cilnidipine synthesis?
A: Without removing acid residues from the first condensation step, the acidic environment during the subsequent high-temperature reflux causes hydrolysis of the ester bonds in the final Cilnidipine molecule. This creates difficult-to-remove byproducts that significantly lower API purity.
Q: What purity levels can be achieved with this novel method?
A: By implementing the alkaline washing step for the intermediate, the process consistently achieves HPLC purity levels exceeding 99.90%, compared to approximately 93.9% in conventional methods that skip this purification.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes common solvents like ethanol and dichloromethane and avoids complex requirements such as nitrogen protection or expensive transition metal catalysts, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cilnidipine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires more than just a patent; it demands engineering excellence and rigorous quality oversight. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced synthesis routes like the one described in CN111072552B are fully realized in bulk manufacturing. We understand that achieving high-purity Cilnidipine is not merely about hitting a number on a certificate of analysis but about ensuring batch-to-batch consistency that safeguards patient safety. Our facilities are equipped with rigorous QC labs and state-of-the-art analytical instrumentation capable of detecting trace impurities at ppm levels, guaranteeing that every shipment meets the stringent purity specifications required by global regulatory bodies.
We invite pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our technical procurement team is ready to provide specific COA data from recent pilot runs and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency. By collaborating with us, you gain access to a partner committed to continuous improvement and technical transparency, ensuring that your pipeline of cardiovascular medications is supported by the highest standards of chemical manufacturing and reliability.
