Advanced Manufacturing of High-Purity Cilnidipine: Technical Breakthroughs and Commercial Scalability
The pharmaceutical industry continuously demands higher purity standards for cardiovascular active pharmaceutical ingredients (APIs), particularly for complex dihydropyridine calcium channel blockers like Cilnidipine. Patent CN111072552A, published in April 2020, introduces a transformative preparation method that addresses long-standing challenges in impurity control and yield optimization. This technical disclosure outlines a robust synthetic route that achieves an exceptional HPLC purity of 99.92% and a total yield of 70.0%, significantly outperforming conventional methodologies that often struggle with hydrolysis byproducts and viscous reaction masses. For R&D directors and procurement specialists, understanding the nuances of this process is vital for securing a reliable cilnidipine supplier capable of meeting stringent regulatory specifications. The core innovation lies not merely in the reaction conditions but in a strategic intermediate purification step that fundamentally alters the impurity profile of the final bulk drug.
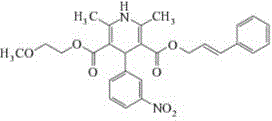
Cilnidipine, chemically known as 1,4-dihydro-2,6-dimethyl-4-(3-nitrophenyl)-3,5-pyridinedicarboxylic acid 2-methoxyethyl cinnamyl alcohol ester, serves as a potent antihypertensive agent. Its dual mechanism of action, inhibiting both L-type and N-type calcium channels, offers superior clinical benefits with reduced sympathetic nerve activity. However, the molecular complexity, featuring two distinct ester side chains and a sensitive dihydropyridine ring, makes its synthesis prone to degradation. The patent data provides a clear roadmap for overcoming these stability issues, positioning this method as a benchmark for cost reduction in antihypertensive API manufacturing. By shifting from traditional acid-catalyzed condensations that leave residual acidity to a buffered and refined approach, manufacturers can ensure batch-to-batch consistency essential for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cilnidipine has been plagued by operational inefficiencies and chemical instability inherent to standard Hantzsch pyridine synthesis variations. Traditional protocols often involve the direct condensation of methoxyethyl acetoacetate and m-nitrobenzaldehyde using concentrated sulfuric acid without adequate solvent management, leading to a reaction mass that becomes increasingly viscous as the reaction progresses. This physical change severely impedes effective stirring and heat transfer, resulting in incomplete conversion rates and a typical yield hovering around 58.6%. Furthermore, the crude intermediates generated in these processes retain significant amounts of acidic residues, such as sulfuric acid and acetic acid byproducts. When these unpurified intermediates are subjected to the subsequent high-temperature cyclization steps, the residual acids act as catalysts for the hydrolysis of the delicate ester linkages within the Cilnidipine structure. This side reaction generates hydrolysis byproducts that are structurally similar to the target molecule, making them notoriously difficult to remove via standard recrystallization, ultimately capping the purity at approximately 93.93% and necessitating expensive and wasteful reprocessing.
The Novel Approach
The methodology disclosed in CN111072552A represents a paradigm shift by introducing a dedicated intermediate refining stage that decouples the condensation chemistry from the cyclization environment. Instead of proceeding directly to the final ring closure, the process isolates the 2-(3-nitrobenzylidene)acetoacetoxyethyl ester intermediate and subjects it to a rigorous washing protocol using organic solvents like dichloromethane and alkaline solutions such as sodium bicarbonate. This critical intervention effectively neutralizes and removes the acidic residues before they can compromise the final product. Additionally, the use of acetic anhydride as a reaction solvent in the initial step improves the fluidity of the reaction mixture, ensuring homogeneous mixing and complete reaction at moderate temperatures of 25-30°C. By eliminating the source of hydrolysis early in the workflow, the novel approach allows the final cyclization to proceed in absolute ethanol under controlled reflux conditions, yielding a product with 99.92% purity. This strategic modification not only simplifies the downstream purification but also drastically enhances the overall material throughput, making it highly suitable for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Acid-Catalyzed Hydrolysis Prevention
To fully appreciate the technical superiority of this route, one must analyze the degradation mechanisms that typically afflict dihydropyridine synthesis. The formation of Cilnidipine involves a multi-component condensation that is thermodynamically favorable but kinetically sensitive to pH levels. In conventional routes, the presence of strong mineral acids (H2SO4) and carboxylic acids (AcOH) in the reaction matrix creates a highly acidic microenvironment. During the subsequent heating phase required for the Hantzsch cyclization (typically 60-78°C), protons from these residual acids attack the carbonyl oxygen of the ester groups attached to the dihydropyridine ring. This protonation activates the ester towards nucleophilic attack by trace water molecules present in the solvent or generated during condensation, leading to cleavage of the methoxyethyl or cinnamyl ester chains. The resulting carboxylic acid derivatives possess polarity and solubility profiles very similar to the parent drug, rendering standard chromatographic or crystalline separation inefficient. The patent's innovation effectively breaks this degradation cycle by ensuring the intermediate entering the cyclization vessel is chemically neutral.
Furthermore, the choice of solvents plays a pivotal role in the mechanistic control of impurity formation. The transition to using absolute ethanol for the final cyclization step, combined with the pre-removal of water-generating acidic components, creates an anhydrous environment that kinetically suppresses hydrolysis. The patent specifies a reflux temperature range of 60-78°C, with an optimal window of 60-65°C, which provides sufficient thermal energy to drive the cyclization forward without accelerating thermal degradation pathways. The addition of activated carbon during the post-reaction treatment further adsorbs colored impurities and trace organic byproducts, polishing the crude material before the final crystallization. This multi-layered approach to impurity control—combining chemical neutralization, solvent optimization, and physical adsorption—demonstrates a sophisticated understanding of process chemistry that ensures the structural integrity of the sensitive dihydropyridine core is maintained throughout the synthesis.
How to Synthesize Cilnidipine Efficiently
Implementing this synthesis route requires precise adherence to the four-step protocol outlined in the patent data to replicate the high-yield and high-purity results. The process begins with the low-temperature condensation of precursors, followed by the crucial aqueous workup of the intermediate, and concludes with the thermal cyclization and recrystallization. Operators must pay close attention to the temperature gradients, particularly maintaining the 0-5°C range during the initial acid addition to prevent exothermic runaway, and strictly controlling the reflux time of 22 hours to ensure complete conversion without over-exposure to heat. The following guide summarizes the standardized operational parameters derived from the patent examples, serving as a foundational reference for process engineers aiming to establish this workflow in a GMP environment. Detailed standard operating procedures (SOPs) should be developed based on these core technical pillars.
- Condense methoxyethyl acetoacetate with m-nitrobenzaldehyde using acetic anhydride and sulfuric acid catalyst at 0-5°C, followed by reaction at 25-30°C.
- Critically refine the intermediate 2-(3-nitrobenzylidene)acetoacetoxyethyl ester by dissolving in organic solvent and washing with alkali to remove acid residues.
- React the refined intermediate with 3-aminocrotonic acid cinnamyl ester in absolute ethanol under reflux (60-78°C) for 22 hours, followed by ethanol recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the technical refinements in this patent translate directly into tangible business value, primarily through enhanced process reliability and reduced waste generation. The elimination of difficult-to-remove hydrolysis byproducts means that the final purification step is significantly more efficient, reducing the consumption of recrystallization solvents and minimizing the loss of valuable API mass during filtering. This efficiency gain is critical for cost reduction in pharmaceutical intermediate manufacturing, as it lowers the cost of goods sold (COGS) without compromising on quality metrics. Moreover, the avoidance of specialized equipment requirements, such as nitrogen protection systems needed for aluminum alkoxide-catalyzed methods mentioned in the background art, allows for production in standard stainless steel reactors, thereby lowering capital expenditure barriers and increasing facility flexibility.
- Cost Reduction in Manufacturing: The implementation of the intermediate refining step, while adding a unit operation, paradoxically lowers overall production costs by preventing the formation of intractable impurities. In traditional processes, the presence of hydrolysis byproducts often necessitates multiple recrystallizations or preparative chromatography to meet pharmacopeial standards, which consumes vast amounts of solvent and time. By ensuring the intermediate is acid-free, the new method achieves a purity of 99.92% in a single final crystallization, drastically simplifying the downstream processing train. This streamlining reduces utility consumption, labor hours, and solvent disposal costs, creating a leaner manufacturing profile that is economically superior to legacy methods yielding only 58.6%.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes significantly to supply chain stability by reducing the risk of batch failures. Conventional methods are sensitive to minor variations in stirring efficiency due to viscosity changes, which can lead to inconsistent reaction outcomes and unpredictable delivery schedules. The new method's use of acetic anhydride as a solvent maintains a manageable viscosity throughout the reaction, ensuring consistent heat transfer and mixing even at larger scales. This predictability allows for more accurate production planning and shorter lead times for high-purity pharmaceutical intermediates. Additionally, the reliance on commodity chemicals like ethanol, dichloromethane, and sodium bicarbonate ensures that raw material sourcing remains stable and unaffected by the supply volatility often associated with specialized catalysts or exotic reagents.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this process offers distinct advantages by minimizing the generation of hazardous waste. The ability to achieve high yields (70.0%) means less starting material is required per kilogram of finished product, inherently reducing the E-factor (mass of waste per mass of product). The washing steps utilize aqueous bicarbonate solutions which are easier to treat than heavy metal catalyst residues found in alternative routes. Furthermore, the process operates at atmospheric pressure and moderate temperatures, reducing energy intensity. These factors facilitate a smoother technology transfer from pilot plant to commercial scale, enabling manufacturers to rapidly ramp up production volumes to meet market demand for cardiovascular medications while adhering to increasingly strict environmental regulations regarding solvent emissions and waste discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Cilnidipine using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis provided in patent CN111072552A, offering clarity on why this route is preferred for modern API manufacturing. Understanding these details helps stakeholders make informed decisions regarding vendor qualification and process adoption. The answers reflect the consensus on best practices for handling dihydropyridine derivatives to ensure maximum stability and efficacy.
Q: Why is the intermediate refining step critical in Cilnidipine synthesis?
A: The intermediate refining step removes residual sulfuric acid and acetic acid. Without this removal, these acids catalyze the hydrolysis of the sensitive ester bonds in the final Cilnidipine molecule during the high-temperature cyclization step, leading to difficult-to-remove hydrolysis byproducts and lower purity (dropping from 99.92% to ~93.9%).
Q: What represents the primary yield improvement in this novel method compared to traditional routes?
A: Traditional methods often suffer from viscous reaction masses and incomplete reactions, yielding approximately 58.6%. The patented method utilizing acetic anhydride as a solvent and optimizing the temperature profile achieves a total yield of approximately 70.0%, representing a significant increase in material efficiency.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It avoids the need for inert gas protection (nitrogen) required by some aluminum alkoxide-catalyzed methods and utilizes common solvents like ethanol and dichloromethane, simplifying equipment requirements and reducing operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cilnidipine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high-purity standards demonstrated in the lab are faithfully reproduced in the plant. Our facilities are equipped with state-of-the-art reactor systems capable of handling the specific temperature and solvent requirements of the Cilnidipine synthesis, including precise low-temperature control for the condensation step and efficient solvent recovery systems for the ethanol recrystallization. We maintain stringent purity specifications through our rigorous QC labs, utilizing HPLC methods identical to those described in the patent to verify that every batch meets the 99.9% purity threshold required for global regulatory submissions.
We invite pharmaceutical companies seeking to optimize their supply chain for antihypertensive APIs to engage with our technical team for a Customized Cost-Saving Analysis. Our experts can evaluate your current sourcing strategy and demonstrate how adopting this refined synthesis route can enhance your margin structure while securing a stable supply of critical intermediates. We encourage you to contact our technical procurement team to request specific COA data from our recent pilot batches and to discuss route feasibility assessments tailored to your specific volume requirements. By partnering with us, you gain access to a supply chain that is not only compliant and reliable but also technically advanced enough to support the next generation of cardiovascular therapies.
