Revolutionizing Tembotrione Production: A Safe, High-Yield Base-Catalyzed C-Acylation Strategy
Revolutionizing Tembotrione Production: A Safe, High-Yield Base-Catalyzed C-Acylation Strategy
The global demand for high-efficiency herbicides continues to drive innovation in agrochemical manufacturing, particularly for HPPD inhibitors like Tembotrione. As a leading player in the fine chemical sector, we have closely analyzed the technological breakthroughs presented in patent CN114560795A, which details a transformative method for preparing Tembotrione. This patent introduces a novel C-acylation reaction under alkaline conditions that fundamentally alters the synthetic landscape by bypassing the traditional, hazardous rearrangement steps. By leveraging a direct coupling strategy between a functionalized benzoate derivative and 1,3-cyclohexanedione, this process offers a pathway to high-purity intermediates with exceptional safety profiles. For R&D directors and procurement specialists, understanding this shift is critical for future-proofing supply chains against increasingly stringent environmental regulations.
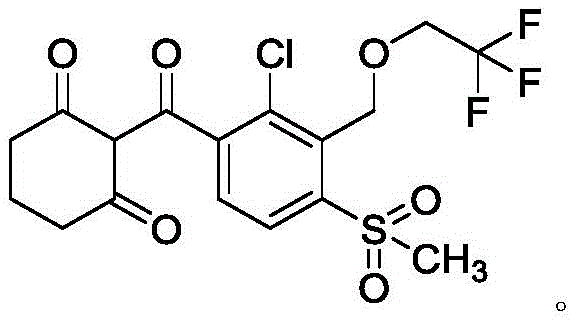
The significance of this patent lies in its ability to deliver a robust, scalable solution for agrochemical intermediate production. Unlike previous iterations that struggled with low overall yields and toxic waste streams, this methodology prioritizes atom economy and operational safety. The core innovation involves the use of strong bases with a conjugate acid pKa value exceeding 12, such as potassium tert-butoxide, to facilitate the nucleophilic attack necessary for bond formation. This technical advancement not only simplifies the workflow but also aligns perfectly with the industry's move towards greener chemistry, ensuring that manufacturers can meet both productivity targets and sustainability goals without compromise.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tembotrione has been plagued by reliance on highly toxic reagents, specifically acetone cyanohydrin or hydrogen cyanide, which are essential for the rearrangement of enol ester intermediates. As illustrated in prior art routes like those described in CN 104292137A, the process typically involves multiple steps where an intermediate is first alkylated, hydrolyzed, and then subjected to a dangerous cyanide-catalyzed rearrangement to form the final trione structure. These conventional pathways suffer from significant drawbacks, including a total yield often hovering around merely 28.4 percent, which represents a massive loss of raw materials and increased cost per kilogram. Furthermore, the handling of virulent reagents necessitates expensive containment infrastructure, rigorous safety protocols, and complex waste treatment systems, creating a substantial burden on both operational expenditure and environmental compliance.
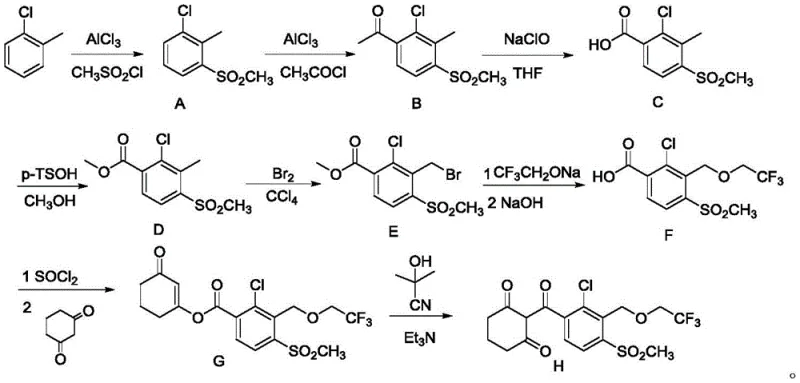
The Novel Approach
In stark contrast, the novel approach disclosed in the patent eliminates the need for any cyanide-based rearrangement entirely. Instead, it employs a direct C-acylation strategy where a compound of Formula I reacts with 1,3-cyclohexanedione under basic conditions. This paradigm shift allows for a much more linear and efficient synthesis, drastically reducing the number of unit operations required. By avoiding the formation of unstable enol esters that require hazardous catalysis, the new method achieves a remarkable yield improvement, with experimental data indicating yields as high as 87.2 percent. This transition from a hazardous rearrangement to a straightforward condensation reaction represents a major leap forward in cost reduction in herbicide manufacturing, offering a cleaner, safer, and economically superior alternative for large-scale production facilities.
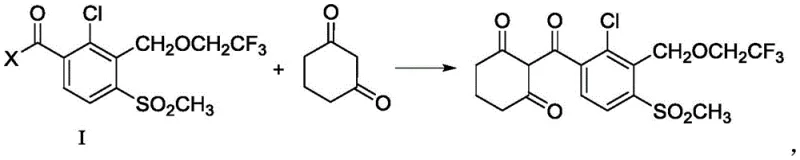
Mechanistic Insights into Base-Catalyzed C-Acylation
The success of this new synthetic route hinges on the precise control of reaction thermodynamics and kinetics through the selection of an appropriate base. The mechanism involves the deprotonation of 1,3-cyclohexanedione by a strong base, such as potassium tert-butoxide (t-BuOK), to generate a stable nucleophilic enolate species. The patent specifies that the pKa of the conjugate acid of the base must be greater than 12 at 25°C to ensure sufficient concentration of the reactive enolate. This enolate then attacks the carbonyl carbon of the benzoate derivative (Formula I), where X can be a halogen or an alkoxy group. The choice of solvent is equally critical; aprotic solvents like tetrahydrofuran (THF) are preferred because they solvate the cation effectively without interfering with the nucleophile, thereby enhancing the reaction rate and selectivity.
Furthermore, the process demonstrates remarkable versatility regarding the leaving group X on the benzoyl precursor. Whether utilizing an acid chloride (where X is Cl) generated in situ from the corresponding acid, or a methyl/ethyl ester (where X is OR1), the reaction proceeds efficiently under reflux conditions between 60°C and 75°C. This flexibility allows manufacturers to optimize their supply chain based on the availability and cost of starting materials. For instance, if acid chlorides are too corrosive for certain reactor setups, the ester route provides a viable, albeit slightly slower, alternative. The robustness of this mechanism ensures that impurity profiles remain clean, as the direct acylation minimizes side reactions common in multi-step rearrangement processes, ultimately delivering high-purity agrochemical intermediates that require less downstream purification.
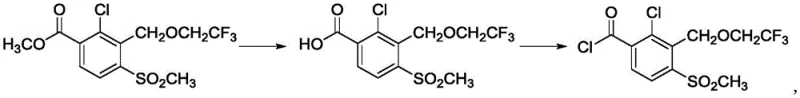
How to Synthesize Tembotrione Efficiently
Implementing this novel synthesis requires careful attention to stoichiometry and temperature control to maximize the benefits of the base-catalyzed mechanism. The process begins with the preparation of the key intermediate, typically involving the etherification of a bromomethyl-benzoate with trifluoroethanol, followed by the critical C-acylation step. Operators must ensure that the base is added to the 1,3-cyclohexanedione solution prior to the addition of the electrophile to pre-form the enolate, a technique that prevents self-condensation of the dione. The detailed standardized synthesis steps, including specific molar ratios and workup procedures derived from the patent examples, are outlined below to guide your technical team in replicating this high-yield process.
- Preparation of the key intermediate methyl 2-chloro-3-(2,2,2-trifluoroethoxy)methyl-4-methylsulfonylbenzoate via etherification.
- Generation of the 1,3-cyclohexanedione enolate using a strong base (pKa > 12) such as potassium tert-butoxide in an aprotic solvent like THF.
- Direct C-acylation reaction between the enolate and the benzoate intermediate under reflux conditions (60-75°C) followed by acidification and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patent technology translates into tangible strategic advantages beyond mere chemical efficiency. The primary benefit is the drastic simplification of the safety infrastructure required for production. By removing acetone cyanohydrin and hydrogen cyanide from the bill of materials, facilities can significantly reduce their insurance premiums, regulatory reporting burdens, and emergency response costs. This elimination of highly toxic reagents also streamlines the logistics of raw material sourcing, as suppliers of cyanide derivatives are fewer and subject to stricter controls compared to standard organic bases and solvents. Consequently, this leads to a more resilient supply chain that is less susceptible to regulatory shutdowns or transportation restrictions.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the substantial increase in reaction yield. Moving from a historical yield of roughly 28 percent to over 87 percent means that nearly three times less raw material is required to produce the same amount of active ingredient. This efficiency gain directly lowers the variable cost of goods sold (COGS). Additionally, the removal of the rearrangement step reduces energy consumption and solvent usage, further compounding the savings. The ability to use readily available esters or acid chlorides without specialized catalysts also contributes to a leaner, more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as potassium tert-butoxide, THF, and standard benzoate derivatives ensures a stable and diversified supply base. Unlike specialized toxic reagents that may face supply disruptions due to environmental crackdowns, these foundational chemicals are produced globally in vast quantities. This abundance guarantees commercial scale-up of complex agrochemical intermediates without the risk of bottlenecking on a single niche supplier. Furthermore, the simplified process flow shortens the overall production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is inherently superior. The absence of cyanide waste eliminates the need for costly detoxification units and reduces the hazard classification of the effluent. This makes the technology highly scalable, as expanding capacity does not proportionally increase the environmental risk profile. Facilities can confidently plan for reducing lead time for high-purity agrochemical intermediates knowing that the process meets modern green chemistry standards, thereby future-proofing the operation against tightening global environmental legislation.
Frequently Asked Questions (FAQ)
To assist your technical and commercial teams in evaluating this technology, we have compiled answers to common questions regarding the implementation and benefits of this synthesis method. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for your decision-making process. Understanding these nuances is essential for assessing the feasibility of integrating this route into your existing manufacturing portfolio.
Q: Why is the new base-catalyzed method superior to traditional Tembotrione synthesis routes?
A: Traditional routes rely on toxic acetone cyanohydrin or hydrogen cyanide for the rearrangement step, posing severe safety and environmental risks. The new method utilizes a direct C-acylation under basic conditions, eliminating hazardous reagents while achieving significantly higher yields (up to 87.2%).
Q: What are the critical reaction parameters for the C-acylation step?
A: The reaction requires a strong base with a conjugate acid pKa greater than 12, such as potassium tert-butoxide. It is typically conducted in an aprotic solvent like tetrahydrofuran (THF) at temperatures between 60°C and 75°C under reflux to ensure complete conversion.
Q: Can this process be scaled for industrial production?
A: Yes, the process is designed for industrial scalability. It avoids complex purification steps associated with toxic byproducts and uses commercially available solvents and reagents, making it suitable for large-scale manufacturing from 100 kgs to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tembotrione Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is not just a technical upgrade but a strategic imperative. Our team of expert chemists has extensively studied the base-catalyzed C-acylation pathway and possesses the capability to translate this patent knowledge into commercial reality. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this process are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of Tembotrione intermediate meets the exacting standards required by global agrochemical formulators.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can drive value and security for your herbicide production needs.
