Revolutionizing Eribulin Intermediate Synthesis via Electrochemical NHK Reaction for Commercial Scale
Introduction to Advanced Electrochemical Synthesis of Eribulin Intermediates
The pharmaceutical industry continuously seeks robust methodologies for constructing complex macrocyclic structures, particularly for oncology therapeutics like Eribulin. Patent CN113512733B introduces a groundbreaking approach to synthesizing the critical intermediate ERB (CAS: 157322-47-9) through an electrochemical Nozaki-Hiyama-Kishi (NHK) reaction. This technology represents a significant paradigm shift from traditional stoichiometric metal-mediated couplings, addressing long-standing challenges in efficiency and environmental impact. By leveraging electrochemical reduction to regenerate active catalytic species in situ, this method circumvents the need for excessive metallic reductants, thereby streamlining the production workflow. For R&D directors and process chemists, this innovation offers a viable pathway to enhance the purity profile and reduce the impurity burden associated with heavy metal residues in the final API. The strategic implementation of this electrochemical protocol ensures a more sustainable manufacturing process, aligning with modern green chemistry principles while maintaining the rigorous stereochemical control required for such a complex molecule containing 19 chiral centers.
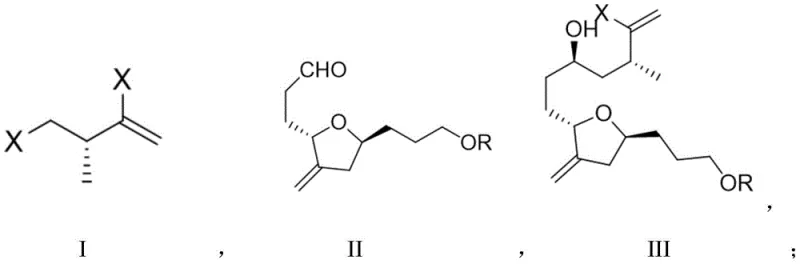
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the ERB intermediate has relied on classical NHK reaction conditions which necessitate the use of stoichiometric amounts of metal powders, typically manganese or zinc, to drive the reduction of chromium species. As documented in prior art such as Angew. Chem. Int. Ed. 2009, these traditional protocols suffer from prolonged reaction times often exceeding 42 hours, which creates significant bottlenecks in production scheduling. Furthermore, the reliance on bulk metal reductants generates substantial quantities of solid waste, complicating downstream processing and increasing the environmental footprint of the manufacturing facility. The economic implications are profound, as the disposal of metal sludge and the extended reactor occupancy time drive up the cost of goods sold. Additionally, the stereoselectivity in conventional thermal NHK reactions can be inconsistent, often yielding diastereomeric ratios that require extensive and yield-eroding purification steps. These factors collectively render the traditional approach suboptimal for the high-volume, cost-sensitive production of complex pharmaceutical intermediates required for global supply chains.
The Novel Approach
The novel electrochemical methodology described in the patent fundamentally reengineers the reduction step by replacing chemical reductants with electrons supplied via a controlled current. This approach allows for the precise regeneration of the active Cr(II) species at the cathode surface, eliminating the need for excess metal powder and the associated waste streams. The reaction time is dramatically compressed from over 40 hours to merely 3 hours under optimized conditions, representing a massive increase in throughput capacity. By utilizing a sacrificial anode system paired with a cobalt co-catalyst and specialized oxazoline sulfonamide ligands, the process achieves superior diastereoselectivity, consistently delivering dr values around 5:1. This enhancement in selectivity simplifies the purification landscape, allowing for more efficient isolation of the desired isomer. The transition to an electrochemical platform not only improves the economic viability through reduced material costs and faster cycle times but also enhances the safety profile by removing pyrophoric metal handling from the operation.
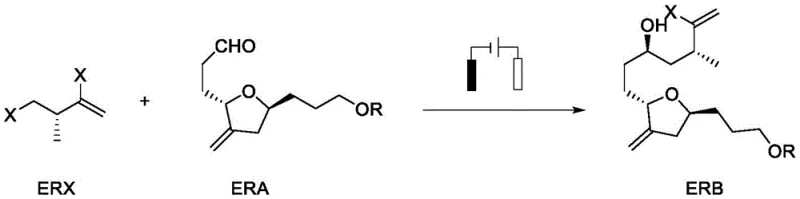
Mechanistic Insights into Electrochemical NHK Coupling
The core of this synthetic advancement lies in the intricate interplay between the chromium and cobalt catalytic cycles driven by electrochemical potential. Mechanistically, the process initiates with the formation of a chiral chromium-ligand complex which serves as the active coupling agent. In the electrochemical cell, Cr(III) species generated during the coupling cycle are reduced back to the active Cr(II) state at the cathode, sustaining the catalytic turnover without external chemical reductants. Simultaneously, the cobalt catalyst undergoes a redox cycle where Co(I) species facilitate the activation of the alkyl halide substrate (ERX) through oxidative addition, generating alkyl radicals that are subsequently trapped by the chromium center. This radical-mediated pathway is crucial for forming the carbon-carbon bond with the aldehyde substrate (ERA). The use of a dissociating agent, such as zirconocene dichloride or TMSCl, plays a pivotal role in cleaving the chromium-oxygen bond in the intermediate alkoxide, releasing the product and regenerating the chromium species for the next cycle. This elegant mechanistic design ensures that the reaction proceeds with high atom economy and minimal byproduct formation, providing a clean reaction profile that is highly desirable for GMP manufacturing environments.
Controlling the impurity profile in such complex couplings is paramount, and the electrochemical method offers distinct advantages in this regard. The precise control over current density and potential prevents over-reduction side reactions that are common when using strong chemical reductants like manganese. Furthermore, the specific coordination environment provided by the chiral oxazoline sulfonamide ligands effectively shields the reactive centers, minimizing non-selective background reactions that lead to diastereomeric impurities. The patent data indicates that optimizing the molar ratios of the ligand, chromium, and cobalt catalysts is critical; deviations can lead to incomplete conversion or erosion of stereocontrol. By maintaining strict control over these parameters and utilizing a sacrificial anode system that does not introduce interfering metal ions into the solution, the process ensures a high-purity output. This level of control is essential for meeting the stringent regulatory requirements for oncology drug intermediates, where impurity thresholds are exceptionally low.
How to Synthesize Eribulin Intermediate ERB Efficiently
Implementing this electrochemical synthesis requires careful attention to the preparation of the catalytic system and the configuration of the electrochemical cell. The process begins with the pre-formation of the chromium-ligand chelate in anhydrous THF, ensuring that the chiral environment is established before the coupling event. Subsequently, the reaction mixture is transferred to an undivided cell equipped with appropriate electrodes, typically zinc as the sacrificial anode and nickel or platinum as the cathode. The addition of supporting electrolytes like tetrabutylammonium tetrafluoroborate is necessary to maintain conductivity, while the choice of solvent, preferably acetonitrile, balances solubility and electrochemical stability. Operators must monitor the constant current application closely, as the rate of electron transfer directly influences the concentration of the active Cr(II) species. Following the reaction, a straightforward workup involving metal adsorbents like Florisil effectively removes residual metal catalysts, yielding the crude product which can be purified via standard chromatographic techniques. For detailed operational parameters and specific reagent quantities, please refer to the standardized synthesis guide below.
- Prepare the chelating agent by reacting a chiral oxazoline sulfonamide ligand with a chromium catalyst and organic base in THF under inert atmosphere.
- Set up an electrochemical cell with a sacrificial anode (e.g., Zinc) and cathode (e.g., Nickel), adding electrolyte, cobalt catalyst, and dissociating agent in acetonitrile.
- Introduce the halide substrate (ERX) and aldehyde substrate (ERA) to the cell, apply constant current (5-15mA) at controlled temperature to effect coupling, then purify the resulting ERB.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this electrochemical technology offers transformative benefits that extend beyond simple yield improvements. The elimination of stoichiometric metal reductants translates directly into substantial cost reductions in raw material procurement and waste disposal logistics. Traditional NHK reactions generate tons of metal sludge that require specialized hazardous waste handling, incurring significant environmental compliance costs. By switching to an electron-driven process, manufacturers can drastically simplify their waste management protocols and reduce the overall environmental levy associated with production. Furthermore, the reduction in reaction time from days to hours significantly enhances asset utilization, allowing existing reactor infrastructure to produce significantly more batches per year without capital expansion. This increased throughput capability provides a buffer against supply disruptions, ensuring a more reliable supply of this critical intermediate for downstream API synthesis. The robustness of the electrochemical parameters also suggests easier scale-up potential, reducing the risk associated with technology transfer from pilot to commercial scale.
- Cost Reduction in Manufacturing: The shift from chemical reductants to electrochemical reduction removes the recurring cost of purchasing large volumes of activated metal powders, which are subject to market price volatility. Additionally, the simplified workup procedure reduces the consumption of purification media and solvents, further lowering the variable cost per kilogram. The ability to run the reaction at ambient or mild temperatures also reduces energy consumption related to heating or cryogenic cooling, contributing to overall operational expenditure savings. These cumulative efficiencies result in a more competitive cost structure for the final intermediate, providing a strategic advantage in pricing negotiations with downstream API manufacturers.
- Enhanced Supply Chain Reliability: The reliance on electricity as the primary "reagent" decouples the production process from the supply chain vulnerabilities associated with specialty metal reagents. Metal powders often face availability issues and strict transportation regulations due to their pyrophoric nature. In contrast, the electrochemical setup utilizes stable, shelf-stable catalysts and common electrode materials that are readily available globally. This stability ensures consistent production schedules and minimizes the risk of batch failures due to reagent quality variations. The shorter cycle time also means that inventory turnover is faster, allowing for a more agile response to fluctuating demand signals from the pharmaceutical market without the need for excessive safety stock.
- Scalability and Environmental Compliance: Electrochemical processes are inherently scalable through the numbering-up of cells or the use of continuous flow electrochemical reactors, which offer superior heat and mass transfer characteristics. This scalability aligns perfectly with the growing global demand for Eribulin, allowing manufacturers to ramp up production capacity smoothly. Moreover, the green chemistry credentials of this method, characterized by reduced E-factors and lower toxicity profiles, facilitate smoother regulatory approvals and audits. Meeting increasingly stringent environmental standards is no longer just a compliance issue but a market differentiator, and this technology positions the supplier as a leader in sustainable pharmaceutical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process robustness and optimization strategies. Understanding these nuances is critical for process engineers evaluating the feasibility of integrating this technology into existing production lines. The answers reflect the specific conditions and outcomes observed during the development of this method, ensuring accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of the electrochemical NHK method over traditional methods?
A: The electrochemical method eliminates the need for large quantities of stoichiometric metal reducing agents like manganese or zinc, significantly reducing solid waste. It also drastically shortens reaction time from over 40 hours to approximately 3 hours while maintaining high stereoselectivity.
Q: Which ligands are most effective for controlling chirality in this synthesis?
A: Oxazoline sulfonamide derivatives, particularly Ligand A (as defined in the patent), demonstrate superior performance in coordinating with chromium to control the stereochemistry of the coupling reaction, achieving diastereomeric ratios up to 5:1.
Q: Is this electrochemical process suitable for industrial scale-up?
A: Yes, the process utilizes standard electrochemical parameters such as constant current and common electrode materials (Zinc/Nickel), making it highly adaptable for continuous flow reactors and large-scale manufacturing without the safety risks associated with pyrophoric reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eribulin Intermediate ERB Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the synthesis of life-saving oncology drugs like Eribulin. Our technical team has extensively analyzed the electrochemical NHK pathway and possesses the expertise to implement this advanced methodology at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. Our facilities are equipped with state-of-the-art electrochemical reactors and stringent purity specifications are enforced through our rigorous QC labs, guaranteeing that every batch of ERB meets the highest standards of stereochemical integrity and chemical purity. We are committed to being a partner that not only supplies materials but also drives process innovation to secure your supply chain.
We invite you to engage with our technical procurement team to discuss how this electrochemical technology can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate the superiority of this method against your current supply benchmarks. Let us collaborate to optimize your Eribulin supply chain with cutting-edge chemistry and unwavering reliability.
