Scalable Electrochemical NHK Reaction for High-Purity Eribulin Intermediate ERB Manufacturing
Introduction to Advanced Electrochemical Synthesis of Eribulin Precursors
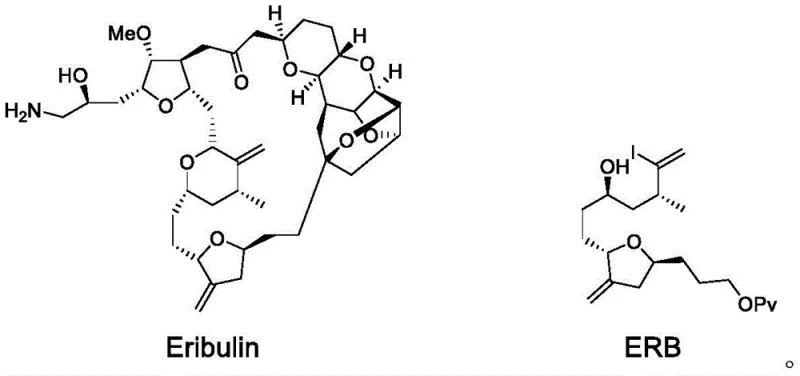
The pharmaceutical industry continuously seeks robust methodologies for constructing complex macrocyclic structures, particularly for oncology therapeutics like Eribulin mesylate. Patent CN113512733A introduces a groundbreaking approach for synthesizing the critical intermediate ERB (CAS: 157322-47-9) via an electrochemical Nozaki-Hiyama-Kishi (NHK) reaction. This technology addresses the inherent challenges of assembling the C1-C6 fragment of Eribulin, a molecule renowned for its structural complexity with 19 chiral centers. By leveraging electrochemical reduction, this novel pathway eliminates the need for stoichiometric amounts of hazardous metal reducing agents traditionally employed in NHK couplings. The innovation lies in the precise integration of a chiral oxazoline sulfonamide ligand with a dual chromium-cobalt catalytic system within an electrochemical cell. This strategic combination not only enhances the atom economy of the process but also drastically improves the operational safety profile by avoiding pyrophoric reagents. For global supply chains, this represents a pivotal shift towards greener, more sustainable manufacturing of high-value pharmaceutical intermediates.
The significance of this patent extends beyond mere academic interest; it offers a tangible solution for reliable pharmaceutical intermediate suppliers aiming to optimize their production lines. The electrochemical setup allows for fine-tuned control over the reduction potential, thereby minimizing side reactions that often plague traditional chemical reductions. Furthermore, the method demonstrates exceptional compatibility with various hydroxyl protecting groups, providing flexibility in synthetic route design. As regulatory pressures mount regarding heavy metal waste and solvent usage, adopting such electrochemical methodologies becomes a strategic imperative for maintaining compliance and competitiveness in the API manufacturing sector. This report delves into the technical nuances and commercial implications of this advanced synthesis route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the ERB intermediate has been fraught with inefficiencies that hinder scalable production. Prominent literature, such as the work by Phillips et al., describes a five-step sequence starting from compound 1, achieving a total yield of merely 37.6%. A critical bottleneck in these traditional routes is the asymmetric coupling of aldehydes and haloalkanes, which relies on classical NHK conditions. These conventional methods typically necessitate reaction times extending up to 42 hours, coupled with modest yields around 52% and poor diastereoselectivity (dr = 6:1). Moreover, the reliance on excessive amounts of metal simple substances as reducing agents generates substantial solid waste, complicating downstream processing and environmental compliance. The use of hazardous reagents like diazomethane in certain steps further exacerbates safety concerns, rendering these processes less attractive for commercial scale-up of complex intermediates.
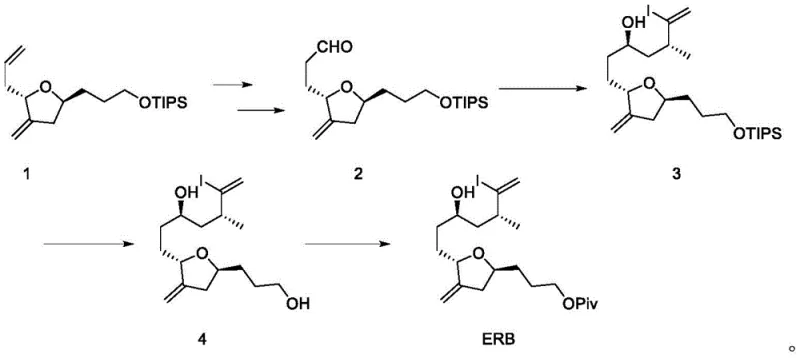
Beyond the immediate reaction metrics, the logistical burden of managing large quantities of metal waste cannot be overstated. Traditional NHK reactions often require rigorous quenching and purification steps to remove residual chromium and other metals to meet stringent pharmaceutical purity standards. This not only increases the cost of goods sold (COGS) but also extends the overall cycle time, impacting the agility of the supply chain. The low stereoselectivity observed in older methods often necessitates additional chromatographic separations or recrystallization steps, further eroding the overall process efficiency. Consequently, there is a pressing demand for a methodology that can deliver high purity and yield while mitigating the environmental footprint associated with heavy metal usage in fine chemical manufacturing.
The Novel Approach
The electrochemical NHK reaction detailed in CN113512733A offers a transformative alternative that directly addresses these legacy issues. By substituting chemical reducing agents with electrons supplied via an electrochemical cell, the process achieves a remarkable reduction in reaction time, completing the key coupling step in just 3 hours compared to the traditional 42 hours. This acceleration is achieved without compromising yield; in fact, the optimized electrochemical protocol delivers yields up to 55% with improved stereoselectivity (dr = 5:1). The core of this innovation is the regeneration of the active Cr(II) species at the cathode, which sustains the catalytic cycle without the need for stoichiometric metal consumption. This shift fundamentally alters the mass balance of the reaction, leading to a cleaner reaction profile and simplified workup procedures.
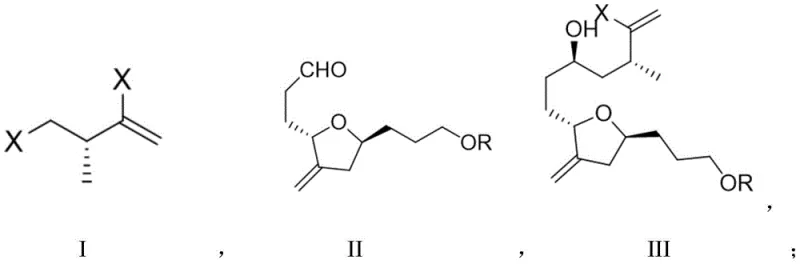
Furthermore, the novel approach incorporates a sophisticated ligand system that ensures high fidelity in stereocontrol. The use of specific oxazoline sulfonamide ligands, such as Ligand A, in conjunction with a cobalt co-catalyst, facilitates a highly selective radical coupling mechanism. This precision is crucial for constructing the intricate carbon framework of Eribulin. From a cost reduction in API manufacturing perspective, the elimination of bulk metal waste translates to significant savings in disposal costs and raw material procurement. The mild reaction conditions, operating effectively at room temperature or slightly elevated temperatures, also reduce energy consumption. This holistic improvement in efficiency and sustainability positions the electrochemical method as a superior choice for modern pharmaceutical intermediate production.
Mechanistic Insights into Electrochemical NHK Coupling
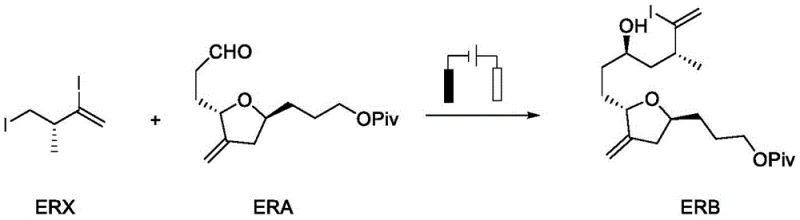
The mechanistic elegance of this electrochemical transformation lies in the synergistic interplay between chromium and cobalt catalysis driven by electron transfer. The cycle initiates with the reduction of Co(II) to Co(I) by Cr(II), followed by the oxidative addition of Co(I) to the carbon-halogen bond of the alkyl halide substrate (ERX). This step generates an alkyl-Co(III) intermediate, which undergoes homolytic cleavage to release an alkyl radical and regenerate Co(II). The transient alkyl radical is then trapped by Cr(II) to form a nucleophilic chromium-alkyl species. This organochromium intermediate subsequently attacks the aldehyde carbonyl of the ERA substrate, forming a chromium alkoxide. The presence of a dissociating agent, such as zirconocene dichloride, is critical here as it promotes the cleavage of the chromium-oxygen bond, releasing the product and regenerating Cr(III).
At the cathode, the spent Cr(III) is efficiently reduced back to the active Cr(II) state, closing the catalytic loop and driving the reaction forward continuously. This electrochemical regeneration is the key differentiator that allows for the use of catalytic rather than stoichiometric amounts of chromium. Meanwhile, the anode undergoes sacrificial oxidation, typically involving zinc or iron, which maintains charge balance without introducing interfering byproducts. The chiral environment provided by the oxazoline sulfonamide ligand coordinates with the chromium center, imposing steric constraints that direct the facial selectivity of the aldehyde attack. This precise control is essential for establishing the correct stereochemistry at the newly formed carbon-carbon bond, ensuring the biological activity of the final drug substance.
How to Synthesize Eribulin Intermediate ERB Efficiently
Implementing this electrochemical synthesis requires careful attention to reagent quality and cell configuration to maximize yield and selectivity. The process begins with the preparation of a chiral chelating agent by mixing the ligand, chromium catalyst, and an organic base like 1,8-bis-methylaminonaphthalene in tetrahydrofuran. This mixture is then introduced into an electrochemical cell containing the aldehyde substrate, electrolyte, and cobalt catalyst. The reaction is driven by a constant current, typically between 5 to 15 mA, under an inert atmosphere to prevent oxidation of sensitive intermediates. Following the reaction, the mixture is treated with a metal adsorbent such as Florisil to sequester residual metal species, followed by filtration and chromatographic purification. This streamlined workflow minimizes unit operations and enhances throughput.
- Prepare the chiral chelating agent by reacting an oxazoline sulfonamide ligand with a chromium catalyst and organic base in THF under alkaline conditions.
- Conduct the electrochemical coupling in an inert atmosphere using a mixed solvent system, electrolyte, and cobalt catalyst, applying constant current to couple the haloalkane ERX and aldehyde ERA.
- Quench the reaction with a metal adsorbent like Florisil, filter to remove metal residues, and purify the filtrate via column chromatography to isolate the ERB intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology presents compelling economic and operational benefits. The primary advantage stems from the drastic simplification of the reaction stoichiometry. By replacing expensive and hazardous stoichiometric metal reductants with electricity, the process significantly lowers raw material costs and eliminates the logistical complexities associated with handling pyrophoric metals. This shift not only reduces the direct cost of goods but also mitigates the risk of supply disruptions related to specialized metal reagents. Furthermore, the reduced reaction time from days to hours enhances asset utilization, allowing manufacturing facilities to increase throughput without capital expansion. This efficiency gain is critical for meeting the growing global demand for oncology treatments.
- Cost Reduction in Manufacturing: The elimination of stoichiometric metal waste leads to substantial savings in waste disposal and treatment costs. Traditional NHK reactions generate large volumes of heavy metal sludge that require specialized handling and disposal, incurring significant environmental fees. The electrochemical method produces minimal solid waste, primarily consisting of the sacrificial anode material which is easier to manage. Additionally, the higher yield and stereoselectivity reduce the loss of valuable starting materials, further optimizing the cost structure. The simplified workup procedure, requiring only filtration and standard chromatography, reduces solvent consumption and labor hours, contributing to overall process economy.
- Enhanced Supply Chain Reliability: The robustness of the electrochemical process enhances supply chain stability by reducing dependency on volatile reagent markets. Conventional methods often rely on specific grades of metal powders that can be subject to price fluctuations and availability issues. In contrast, the electrochemical setup uses readily available electricity and common salts as electrolytes, ensuring a stable supply of inputs. The shorter cycle time also allows for more responsive production scheduling, enabling manufacturers to adapt quickly to changes in demand. This agility is invaluable in the pharmaceutical sector where timely delivery of intermediates is crucial for downstream drug formulation and market launch.
- Scalability and Environmental Compliance: Electrochemical reactors are inherently scalable, allowing for seamless transition from laboratory to pilot and commercial scales. The modular nature of electrochemical cells facilitates capacity expansion without the need for massive reactor vessels. From a regulatory standpoint, the reduced generation of hazardous waste aligns with increasingly stringent environmental regulations, such as REACH and EPA guidelines. This compliance reduces the risk of regulatory penalties and enhances the corporate sustainability profile. The green chemistry credentials of this process can also be leveraged for marketing advantages, appealing to environmentally conscious stakeholders and customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of ERB. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this method into existing manufacturing workflows.
Q: How does the electrochemical method improve upon traditional NHK reactions for ERB synthesis?
A: The electrochemical method replaces stoichiometric metal reducing agents with electrons, significantly reducing solid waste and improving reaction greenness. It also shortens reaction time from over 40 hours to approximately 3 hours while maintaining high stereoselectivity.
Q: What specific ligands are required for high stereoselectivity in this process?
A: The process utilizes oxazoline sulfonamide derivatives, specifically Ligand A (formula V), which coordinates with chromium to effectively control the chirality of the coupling product, ensuring the correct configuration for the Eribulin backbone.
Q: Is this electrochemical process suitable for large-scale industrial production?
A: Yes, the method is designed for industrial application with mild reaction conditions, simple post-treatment involving metal adsorbents, and the use of scalable electrochemical cells, making it economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eribulin Intermediate ERB Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic technologies to deliver high-quality pharmaceutical intermediates. Our expertise in electrochemical organic synthesis allows us to leverage patents like CN113512733A to offer superior manufacturing solutions for complex molecules like Eribulin intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent supply regardless of volume requirements. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of ERB meets the highest industry standards for chirality and chemical purity.
We invite procurement leaders and R&D teams to collaborate with us on optimizing their supply chains for oncology drug precursors. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production needs. Our technical team is ready to provide specific COA data and route feasibility assessments to demonstrate how our electrochemical capabilities can drive value for your organization. Contact our technical procurement team today to discuss how we can support your long-term strategic goals with reliable, cost-effective, and sustainable manufacturing solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
