Revolutionizing Alkenyl Silane Production via Air-Tolerant Manganese Catalysis for Commercial Scale
Introduction to Breakthrough Manganese Catalysis Technology
The landscape of organosilicon chemistry is undergoing a significant transformation driven by the urgent need for sustainable and cost-effective synthetic methodologies. A pivotal advancement in this field is documented in Chinese Patent CN110229180B, which discloses a highly efficient method for the selective preparation of alkenyl silanes. This technology leverages the unique properties of manganese, an earth-abundant and non-toxic transition metal, to catalyze the dehydrosilylation of terminal olefins with unprecedented selectivity. Unlike conventional approaches that often struggle with byproduct formation and harsh reaction conditions, this novel protocol achieves single-mode selectivity for trans-configuration dehydrosilylation products. The process is remarkably robust, operating effectively under air atmosphere without the necessity for inert gas protection or additional hydrogen acceptors. For R&D directors and procurement specialists in the fine chemical sector, this represents a paradigm shift towards greener, more economical manufacturing of high-value silicon-containing intermediates used extensively in pharmaceuticals and advanced materials.
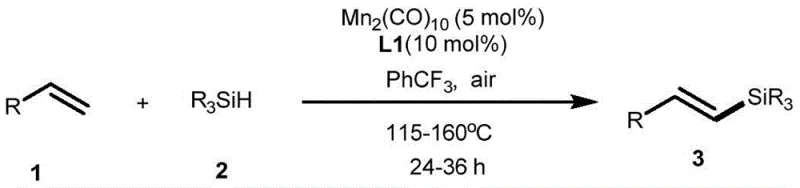
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkenyl silanes has been dominated by the use of precious transition metal catalysts such as rhodium, platinum, and iridium. These traditional methods are fraught with significant economic and operational drawbacks that hinder large-scale adoption. Firstly, the reliance on scarce noble metals drives up the raw material costs substantially, creating volatility in supply chains and impacting the final pricing of downstream intermediates. Secondly, these catalytic systems are typically extremely sensitive to oxygen and moisture, necessitating rigorous inert atmosphere conditions (such as nitrogen or argon shielding) which adds layers of complexity and equipment costs to the manufacturing process. Furthermore, conventional silylation reactions often suffer from poor selectivity, frequently yielding mixtures of hydrosilylation and dehydrosilylation products, or generating cis/trans isomeric mixtures that require difficult and yield-reducing separation processes. The atom economy is also frequently compromised by the need for stoichiometric oxidants or hydrogen acceptors to drive the dehydrosilylation pathway, generating substantial chemical waste.
The Novel Approach
The methodology outlined in the patent data introduces a disruptive alternative that directly addresses these systemic inefficiencies. By utilizing dimanganese decacarbonyl (Mn2(CO)10) in conjunction with a specifically designed ligand (Ligand 1), the reaction achieves high-efficiency synthesis of dehydrosilylation products with exceptional selectivity. A key differentiator is the system's tolerance to air, eliminating the need for expensive inert gas setups and allowing for simpler reactor configurations. The reaction proceeds in trifluorotoluene solvent at elevated temperatures (115-160°C), typically around 140°C, to deliver the trans-alkenyl silane as the sole product. This eliminates the downstream burden of separating isomers or removing hydrosilylation byproducts. Moreover, the absence of external oxidants or hydrogen acceptors improves the overall atom economy, aligning perfectly with modern green chemistry principles. This approach not only simplifies the operational workflow but also drastically reduces the environmental footprint associated with silicon-functionalized intermediate production.
Mechanistic Insights into Ligand-Regulated Manganese Catalysis
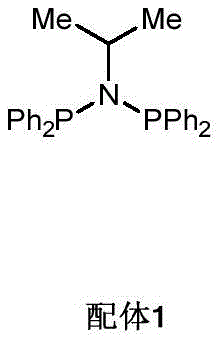
The core innovation of this technology lies in the precise regulation of the catalytic cycle through ligand design. The specific ligand employed, identified as Ligand 1 in the patent documentation, plays a critical role in modulating the electronic and steric environment around the manganese center. This modulation is essential for overriding the thermodynamic preference for hydrosilylation, which is the typical outcome when olefins react with silanes in the presence of many transition metals. The ligand facilitates a pathway where the activation of the Si-H bond and the subsequent insertion of the olefin occur in a manner that favors beta-hydride elimination, leading to the dehydrosilylated product. The mechanistic pathway ensures that the reaction stops at the alkenyl silane stage without further reduction or isomerization. This level of control is rare in base metal catalysis and rivals the selectivity often seen only with much more expensive noble metal systems.
From an impurity control perspective, this mechanism offers distinct advantages for pharmaceutical intermediate manufacturing. The high trans-selectivity means that the crude reaction mixture is significantly cleaner, containing minimal amounts of the cis-isomer or the saturated alkyl silane byproduct. This purity profile simplifies the purification process, often allowing for straightforward column chromatography or even crystallization to achieve the required specifications. For complex substrates, such as those containing sensitive functional groups or sterically hindered environments, the catalyst system demonstrates remarkable compatibility. The ability to tolerate air suggests that the active manganese species is sufficiently stable against oxidation, or that the catalytic turnover is fast enough to outcompete deactivation pathways. This robustness translates to consistent batch-to-batch quality, a critical parameter for regulatory compliance in the production of API intermediates.
How to Synthesize Trans-Alkenyl Silanes Efficiently
Implementing this synthesis route requires careful attention to the catalyst activation phase to ensure optimal performance. The standard protocol involves pre-stirring the manganese source and the ligand in the solvent to generate the active catalytic species before introducing the substrates. This activation step is crucial for achieving the high yields reported in the patent examples, which often exceed 70-80% even for challenging substrates. The reaction conditions are relatively straightforward, utilizing a sealed tube or autoclave capable of withstanding temperatures up to 160°C. The molar ratio of olefin to silane is typically maintained at 1:1, maximizing atom efficiency. While the reaction time can range from 24 to 36 hours depending on the substrate reactivity, the simplicity of the workup compensates for the duration. Detailed standardized synthesis steps for specific substrates are provided in the guide below.
- Prepare the catalyst system by mixing dimanganese decacarbonyl (5 mol%) and Ligand 1 (10 mol%) in trifluorotoluene solvent at 80°C for 1 hour.
- Add the terminal olefin substrate and trisubstituted silane (1: 1 molar ratio) to the reaction mixture without the need for inert gas protection.
- Heat the reaction system to 140°C and maintain for 24-36 hours to achieve high-yield trans-selective dehydrosilylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this manganese-catalyzed process offers tangible strategic benefits that extend beyond simple chemistry. The shift from precious metals to earth-abundant manganese fundamentally alters the cost structure of alkenyl silane production. By eliminating the dependency on volatile noble metal markets, manufacturers can secure more stable pricing and reduce the risk of supply disruptions caused by geopolitical factors affecting platinum group metal mining. Additionally, the operational simplicity of running reactions under air rather than inert atmosphere reduces the capital expenditure required for specialized reactor infrastructure and lowers the ongoing utility costs associated with nitrogen or argon consumption.
- Cost Reduction in Manufacturing: The replacement of expensive rhodium or iridium catalysts with low-cost manganese salts results in a direct and significant reduction in raw material costs. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials into byproduct streams, thereby improving the overall yield and effective cost per kilogram of the final product. The elimination of stoichiometric oxidants or hydrogen acceptors further reduces the bill of materials, contributing to substantial cost savings in the manufacturing process without compromising product quality.
- Enhanced Supply Chain Reliability: Manganese is one of the most abundant transition metals in the earth's crust, ensuring a secure and long-term supply of the primary catalyst component. This abundance contrasts sharply with the supply constraints often faced with noble metals. The robustness of the reaction conditions, particularly the tolerance to air and moisture, simplifies logistics and storage requirements for reagents, reducing the risk of batch failures due to environmental exposure during handling. This reliability ensures consistent delivery schedules for downstream customers relying on these intermediates for their own production lines.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by its applicability to gaseous olefins like ethylene and propylene in autoclave settings. The absence of toxic heavy metals and the reduction in chemical waste due to high atom economy simplify waste treatment protocols, aiding in compliance with increasingly stringent environmental regulations. The straightforward workup procedure, often involving simple concentration and chromatography, facilitates rapid scale-up from laboratory to pilot and commercial production scales, reducing the time-to-market for new silicon-based products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this manganese-catalyzed dehydrosilylation technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the process viability. Understanding these details is essential for technical teams assessing the feasibility of integrating this method into existing manufacturing workflows.
Q: What distinguishes this manganese-catalyzed method from traditional noble metal catalysis?
A: Unlike traditional methods requiring expensive Rh, Pt, or Ir catalysts under strict inert conditions, this patent utilizes earth-abundant manganese which operates effectively under air atmosphere, significantly reducing raw material costs and operational complexity.
Q: How does the ligand regulation improve product selectivity?
A: The specific PNP-type ligand (Ligand 1) regulates the catalytic cycle to favor dehydrosilylation over hydrosilylation, ensuring the exclusive formation of the trans-alkenyl silane configuration without needing additional oxidants or hydrogen acceptors.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process demonstrates excellent substrate applicability including complex molecules and gases, operates under air without sensitive additives, and uses simple workup procedures, making it highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkenyl Silane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this manganese-catalyzed technology for the production of high-purity alkenyl silanes. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this innovative chemistry can be realized at an industrial level. Our facilities are equipped with state-of-the-art reactors capable of handling high-temperature and pressure conditions safely, and our rigorous QC labs enforce stringent purity specifications to meet the demanding requirements of the pharmaceutical and electronic chemical industries. We are committed to delivering consistent quality and supply continuity for all our clients.
We invite you to collaborate with us to leverage this cost-effective and sustainable synthetic route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this technology can optimize your budget. Please contact our technical procurement team today to request specific COA data for our available alkenyl silane inventory and to discuss route feasibility assessments for your custom synthesis projects. Let us help you secure a competitive advantage through superior chemical manufacturing.
