Optimized Industrial Synthesis of 2-Cyano-3-Chloro-5-Trifluoromethylpyridine for High-Performance Agrochemicals
The global demand for high-efficiency pyrazole amide bactericides has driven significant innovation in the synthesis of their key precursors. Patent CN107286087B, published in early 2020, introduces a robust and industrially viable synthetic method for 2-cyano-3-chloro-5-trifluoromethylpyridine, a critical building block in modern agrochemistry. This technology addresses long-standing challenges in halogen exchange and nucleophilic substitution reactions, offering a pathway that combines high selectivity with operational simplicity. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the nuances of this patent is essential for securing a stable supply chain. The method leverages a strategic two-step sequence involving selective fluorination followed by cyanidation, utilizing cost-effective inorganic reagents instead of expensive organic catalysts. This shift not only enhances the economic profile of the manufacturing process but also aligns with stricter environmental compliance standards by reducing waste generation associated with catalyst recovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industry largely relied on methods such as those described in US Patent 66993, which utilized 2,3-dichloro-5-trifluoromethylpyridine reacting with 4-dimethylaminopyridine (DMAP) followed by cyanidation. While chemically feasible, this legacy approach presents severe bottlenecks for commercial scale-up of complex agrochemical intermediates. The primary drawback lies in the recovery and recycling of DMAP, an organic base that is both costly and difficult to separate from the reaction mixture efficiently. Low recovery rates of this catalyst lead to inflated raw material costs and generate significant organic waste streams that require expensive treatment. Furthermore, the reaction conditions in these older protocols often lack the robustness required for consistent multi-ton production, resulting in variable yields typically hovering around 80%. For procurement managers focused on cost reduction in agrochemical manufacturing, these inefficiencies translate directly into higher unit prices and supply volatility.
The Novel Approach
The methodology disclosed in CN107286087B fundamentally re-engineers the synthetic route to eliminate these dependencies on difficult-to-recycle organic catalysts. By substituting the DMAP-mediated pathway with a phase-transfer catalyzed halogen exchange using inorganic fluorides, the process achieves a dramatic improvement in operational efficiency. The new route begins with the conversion of the starting dichloro compound into a fluoro-intermediate, which is subsequently cyanated under mild conditions. This strategy allows for the use of inexpensive reagents like potassium fluoride (KF) and sodium cyanide (NaCN), which are globally available and economically stable. The elimination of complex catalyst recovery steps streamlines the workflow, reducing the overall processing time and energy consumption. Consequently, this approach offers a compelling value proposition for supply chain heads, ensuring that the production of high-purity OLED material or agrochemical precursors remains uninterrupted and cost-effective.
Mechanistic Insights into Phase-Transfer Catalyzed Halogen Exchange
The core of this technological breakthrough lies in the precise control of nucleophilic aromatic substitution reactions through phase-transfer catalysis (PTC). In the first step, the chlorine atom at the 2-position of the pyridine ring is selectively replaced by a fluorine atom using anhydrous KF or NaF. The presence of quaternary ammonium salts, such as benzyltriethylammonium chloride, facilitates the transport of the fluoride anion into the organic phase, significantly enhancing its nucleophilicity. This mechanism allows the reaction to proceed at temperatures between 140-170°C in polar aprotic solvents like DMAC, ensuring complete conversion while minimizing side reactions. The selectivity is crucial, as it prevents the displacement of the chlorine at the 3-position or the trifluoromethyl group, preserving the structural integrity required for downstream applications. Understanding this mechanistic detail is vital for R&D teams aiming to replicate or optimize the process for specific purity specifications.
Following the fluorination, the second step involves the displacement of the newly introduced fluorine atom with a cyano group using sodium cyanide. This transformation is highly sensitive to temperature and solvent choice, with the patent specifying a range of 15-30°C in solvents like dichloroethane. The lower temperature is critical to prevent the hydrolysis of the cyanide reagent, which would otherwise lead to the formation of carboxylic acid impurities and reduce the overall yield. The phase transfer catalyst continues to play a pivotal role here, ensuring efficient contact between the inorganic cyanide salt and the organic substrate. This careful balancing of reaction parameters results in a final product with exceptional purity, reaching up to 99.5% as confirmed by gas chromatography. Such high purity is indispensable for the synthesis of potent bactericides, where even trace impurities can affect biological activity or regulatory approval.
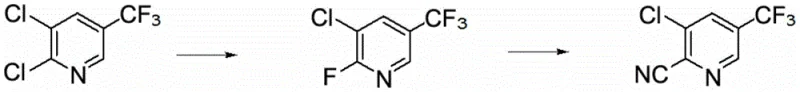
How to Synthesize 2-Cyano-3-Chloro-5-Trifluoromethylpyridine Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding solvent ratios, catalyst loading, and thermal profiles to ensure reproducibility. The process is divided into two distinct stages: the initial fluorination which sets the stage for activation, and the subsequent cyanidation which installs the functional handle. Operators must pay close attention to the distillation steps, particularly the collection of fractions under reduced pressure to isolate the colorless liquid product without thermal degradation. The detailed standardized synthesis steps below outline the precise quantities and conditions necessary to achieve the reported 90% yield and 99.5% purity.
- Fluorination of 2,3-dichloro-5-trifluoromethylpyridine using anhydrous KF and a phase transfer catalyst in DMAC at 140-170°C.
- Cyanidation of the resulting 2-fluoro-3-chloro-5-trifluoromethylpyridine using sodium cyanide and a catalyst in dichloroethane at 15-30°C.
- Post-treatment involving water washing, phase separation, and vacuum distillation to isolate the final product with 99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the bottom line and logistical continuity, the shift to this new synthetic protocol offers substantial strategic benefits beyond mere chemical elegance. The transition from expensive, hard-to-recycle organic catalysts to abundant inorganic salts drastically simplifies the supply chain dynamics. By removing the dependency on specialized reagents like DMAP, manufacturers can mitigate the risk of price spikes and availability shortages that often plague the fine chemical sector. Furthermore, the simplified workup procedure, which involves basic water washing and distillation rather than complex chromatographic separations, significantly reduces the operational overhead. This efficiency gain translates directly into a more competitive pricing structure for the final intermediate, allowing downstream formulators to maintain healthy margins.
- Cost Reduction in Manufacturing: The replacement of 4-dimethylaminopyridine with inexpensive inorganic fluorides and cyanides eliminates a major cost center associated with catalyst procurement and loss. Since the new reagents are commodity chemicals with stable global markets, the volatility in raw material expenses is significantly dampened. Additionally, the high yield of 90% means less starting material is wasted per kilogram of product, further driving down the cost of goods sold. The ability to recover and reuse solvents like DMAC and dichloroethane efficiently adds another layer of economic optimization, ensuring that the process remains lean and profitable even at fluctuating market rates.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit immensely from the use of universally available reagents such as potassium fluoride and sodium cyanide, which are produced at massive scales for various industries. This ubiquity ensures that production schedules are not held hostage by the limited capacity of niche catalyst suppliers. The robustness of the reaction conditions also means that manufacturing can be scaled up or shifted between different facilities with minimal re-validation effort. For supply chain heads, this flexibility is paramount in maintaining continuous delivery schedules to pharmaceutical and agrochemical clients who operate on just-in-time inventory models.
- Scalability and Environmental Compliance: The mild reaction temperatures and straightforward isolation techniques make this process inherently safer and easier to scale from pilot plants to multi-ton reactors. The reduction in organic waste, specifically the avoidance of large volumes of spent DMAP, simplifies effluent treatment and lowers the environmental footprint of the facility. This alignment with green chemistry principles not only reduces disposal costs but also future-proofs the manufacturing site against increasingly stringent environmental regulations. Companies adopting this method demonstrate a commitment to sustainable practices, which is becoming a key differentiator in B2B procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating the feasibility of adoption. Understanding these specifics helps in making informed decisions about process integration and quality assurance protocols.
Q: What are the key advantages of this synthesis method over prior art US 66993?
A: Unlike the prior art which relies on 4-dimethylaminopyridine (DMAP) that is difficult to recover and recycle, this novel method utilizes inexpensive inorganic fluorinating agents and phase transfer catalysts. This significantly simplifies the post-treatment process, improves the overall yield to 90%, and enhances industrial feasibility.
Q: What is the achievable purity and yield for 2-cyano-3-chloro-5-trifluoromethylpyridine?
A: According to patent CN107286087B, the optimized process achieves a product purity of up to 99.5% (GC) and a total two-step yield of approximately 90%. This high quality is critical for downstream pesticide synthesis.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method is specifically designed for industrial scale-up. It employs mild reaction conditions, readily available solvents like DMAC and dichloroethane, and avoids complex catalyst recovery steps, making it highly suitable for reliable agrochemical intermediate supplier operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyano-3-Chloro-5-Trifluoromethylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, backed by our rigorous QC labs which utilize advanced analytical techniques to verify every batch. Our capability to implement the efficient synthesis methods described in CN107286087B allows us to offer a superior balance of quality and value to our global partners.
We invite you to engage with our technical procurement team to discuss how our optimized manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to our supply chain. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a partnership built on transparency and technical excellence.
