Advanced Molybdenum-Copper Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
Advanced Molybdenum-Copper Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust synthetic methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are pivotal for enhancing metabolic stability and bioavailability in modern drug design. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in current manufacturing processes. This technology leverages a unique molybdenum-copper co-catalytic system to facilitate the cycloaddition of trifluoroethylimidoyl chloride and functionalized isonitriles under remarkably mild conditions. The significance of this innovation is underscored by the prevalence of the 1,2,4-triazole scaffold in blockbuster medications such as Sitagliptin and Deferasirox, where the trifluoromethyl group plays a crucial role in optimizing physicochemical properties.
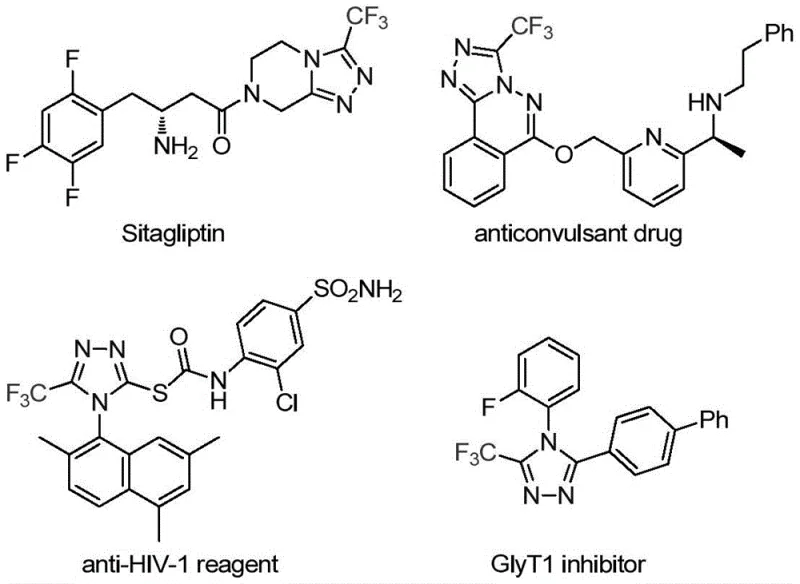
For R&D directors and process chemists, the ability to access these privileged structures efficiently is paramount. The disclosed method not only simplifies the synthetic route but also expands the chemical space available for medicinal chemistry campaigns by tolerating a wide array of functional groups. By shifting away from hazardous hydrazine-based traditional routes, this patent offers a safer, more sustainable pathway that aligns with modern green chemistry principles while maintaining high reaction efficiency and selectivity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on methodologies that present significant operational and safety challenges for industrial application. Traditional literature reports predominantly describe the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These classical approaches often necessitate the use of unstable or hazardous hydrazine derivatives, which pose severe safety risks during handling and storage on a large scale. Furthermore, alternative copper-catalyzed multi-component reactions involving diazonium salts and trifluorodiazoethane require stringent control over reactive intermediates that can be difficult to manage in a commercial setting. The reliance on such volatile precursors frequently leads to inconsistent batch-to-batch reproducibility and complicates the purification process due to the formation of complex impurity profiles.
The Novel Approach
In stark contrast to these legacy methods, the invention detailed in CN113307778A utilizes a sophisticated dual-catalyst system comprising molybdenum hexacarbonyl and cuprous acetate to drive the reaction forward with exceptional precision. This novel approach employs readily available trifluoroethylimidoyl chloride and functionalized isonitriles as starting materials, effectively bypassing the need for dangerous hydrazine reagents. The reaction proceeds smoothly in common organic solvents like THF at moderate temperatures ranging from 70°C to 90°C, eliminating the need for extreme thermal conditions or high-pressure equipment. This shift in strategy not only enhances the safety profile of the manufacturing process but also significantly streamlines the workflow, allowing for a direct one-pot transformation that minimizes waste generation and reduces the overall environmental footprint of the synthesis.
Mechanistic Insights into Mo-Cu Co-Catalyzed Cycloaddition
The mechanistic elegance of this transformation lies in the synergistic interaction between the molybdenum and copper species, which orchestrate the assembly of the triazole ring through a concerted [3+2] cycloaddition pathway. Initially, the molybdenum hexacarbonyl acts as a potent activator for the functionalized isonitrile, forming a transient metal-isocyanide complex that increases the nucleophilicity of the carbon center. Subsequently, the cuprous acetate promotes the attack of this activated species onto the electrophilic carbon of the trifluoroethylimidoyl chloride, facilitating the formation of a five-membered ring intermediate. This cooperative catalysis ensures that the reaction barrier is sufficiently lowered to allow the process to occur under mild thermal conditions, thereby preserving sensitive functional groups that might otherwise decompose under harsher regimes.
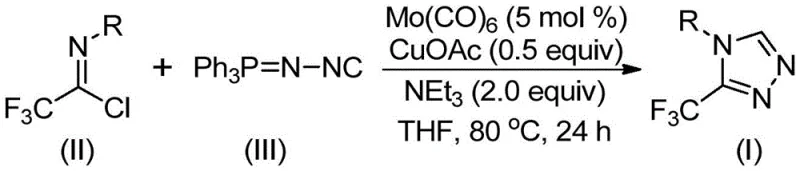
Following the cyclization event, the intermediate undergoes a spontaneous elimination of triphenylphosphine oxide, driven by the thermodynamic stability of the resulting aromatic triazole system. From an impurity control perspective, the mildness of the reaction conditions is a distinct advantage, as it suppresses common side reactions such as polymerization or hydrolysis of the imidoyl chloride starting material. The use of molecular sieves in the reaction mixture further aids in scavenging trace moisture, ensuring that the hydrolytic degradation of reagents is minimized. This results in a cleaner crude reaction mixture, which simplifies downstream purification and ultimately leads to higher isolated yields of the target pharmaceutical intermediate with superior purity specifications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it highly attractive for both laboratory-scale optimization and pilot plant production. The protocol involves a straightforward charging of reagents into a reaction vessel, followed by heating and stirring, which requires no specialized equipment beyond standard glassware or stainless steel reactors. The detailed standardized synthesis steps provided below outline the precise stoichiometry and conditions required to achieve optimal conversion rates as demonstrated in the patent examples.
- Combine molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70°C to 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix the crude sample with silica gel, and perform column chromatography purification to isolate the pure 3-trifluoromethyl-substituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology translates into tangible strategic benefits regarding cost structure and supply continuity. The reliance on commodity chemicals such as triethylamine, cuprous acetate, and commercially available imidoyl chlorides means that the raw material supply chain is robust and less susceptible to geopolitical disruptions or single-source bottlenecks. Unlike processes requiring bespoke ligands or exotic catalysts, the reagents here are bulk chemicals with established global supply networks, ensuring consistent availability and pricing stability for long-term production planning.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the use of inexpensive copper salts and the elimination of costly purification steps associated with removing toxic hydrazine byproducts. By avoiding the need for cryogenic conditions or high-pressure reactors, the capital expenditure (CAPEX) for setting up production lines is drastically reduced, while operational expenditures (OPEX) are lowered through energy savings at moderate reaction temperatures. Furthermore, the high atom economy of the cycloaddition reaction minimizes raw material waste, contributing to substantial cost savings in waste disposal and raw material procurement.
- Enhanced Supply Chain Reliability: The simplicity of the reaction setup and the stability of the starting materials contribute to a more reliable manufacturing schedule with reduced risk of batch failures. Since the starting materials are widely sourced and the reaction tolerates a broad range of substrates, manufacturers can quickly adapt to changing demand for different triazole derivatives without extensive re-validation of the process. This flexibility allows for a more agile response to market needs, ensuring that critical API intermediates can be delivered to customers with consistent lead times and quality.
- Scalability and Environmental Compliance: The patent explicitly notes that the method can be expanded to gram-level reactions and potentially beyond, indicating a clear path to commercial scale-up without encountering the non-linear scaling issues often seen in complex catalytic systems. The absence of heavy metal contaminants that are difficult to remove, combined with the use of standard organic solvents, simplifies compliance with stringent environmental regulations regarding effluent discharge. This eco-friendly profile not only reduces regulatory burden but also aligns with the sustainability goals of major pharmaceutical clients who prioritize green manufacturing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scalability, substrate scope, and purification of the 3-trifluoromethyl-1,2,4-triazole synthesis described in the patent. These insights are derived directly from the experimental data and beneficial effects reported, providing clarity for technical teams evaluating this route for potential integration into their manufacturing portfolios.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies reacting at 70-90°C for 18-30 hours in THF solvent, utilizing Mo(CO)6 and CuOAc as co-catalysts with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates wide substrate tolerance, successfully synthesizing derivatives with methyl, methoxy, fluoro, chloro, and nitro substituents on the aromatic ring.
Q: Is this process suitable for large-scale production?
A: The invention explicitly states that the method can be expanded to gram-level reactions and offers simple post-treatment, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this molybdenum-copper co-catalyzed technology in advancing the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this innovative synthesis for your next drug development program. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your overall budget. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global pharmaceutical market.
