Advanced Hydroxy Cyclohexyl Acetophenone Photoinitiators for High-Efficiency LED Curing Applications
Introduction to Next-Generation LED Photoinitiators
The rapid evolution of ultraviolet curing technology has necessitated the development of photoinitiators specifically tailored for modern light-emitting diode (LED) sources. As detailed in patent CN111574350B, a significant technological gap exists between traditional photoinitiators like compound 184, which absorb maximally at 333nm, and contemporary LED systems that emit primarily above 365nm. This spectral mismatch severely limits polymerization efficiency in industrial applications. The present invention addresses this critical challenge by introducing a novel class of hydroxy cyclohexyl acetophenone photoinitiators designed through precise molecular engineering. By extending the conjugated system via a condensation reaction, these new compounds achieve maximum absorption wavelengths exceeding 365nm, ensuring optimal energy capture from LED sources. This breakthrough not only enhances the double bond conversion rates of polymerized monomers but also opens new avenues for high-performance coatings, inks, and electronic materials where deep curing and rapid processing are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the industry has relied heavily on Type I photoinitiators such as 1-hydroxycyclohexyl phenyl ketone (PI 184) due to their established performance profiles. However, the fundamental limitation of PI 184 lies in its absorption spectrum, which peaks at 333nm and drops significantly in the 365nm to 405nm range utilized by energy-efficient LED curing lamps. When subjected to LED irradiation, conventional initiators fail to generate sufficient free radicals, resulting in incomplete polymerization and poor mechanical properties of the final cured film. Comparative data indicates that under 365nm LED exposure, traditional PI 184 achieves a mere 10% double bond conversion within 100 seconds, rendering it inefficient for high-speed manufacturing lines. Furthermore, attempting to compensate for this inefficiency often requires higher loading levels or longer exposure times, which increases energy consumption and production costs while potentially compromising the thermal stability of the substrate.
The Novel Approach
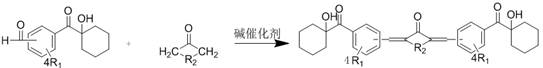
The innovative strategy outlined in the patent involves a structural modification of the hydroxycyclohexylacetophenone core to redshift its absorption characteristics. This is achieved through a condensation reaction between an aldehyde-functionalized hydroxycyclohexylacetophenone intermediate and various ketones containing alpha-hydrogen atoms. This chemical transformation introduces an extended conjugated pi-system across the molecule, effectively shifting the maximum absorption wavelength to above 365nm, with some derivatives reaching up to 392nm. For instance, the reaction with acetone yields a product with a maximum absorption at 381nm and a double bond conversion rate of 85% within 300 seconds under 365nm LED light. This approach allows for the fine-tuning of photophysical properties by simply varying the ketone reactant, providing a versatile platform for developing specialized photoinitiators that perfectly match the emission spectra of specific LED curing equipment.
Mechanistic Insights into Base-Catalyzed Aldol Condensation
The core chemical transformation driving this synthesis is a base-catalyzed aldol condensation, a robust and well-understood organic reaction mechanism that facilitates carbon-carbon bond formation. In this process, a basic catalyst such as sodium hydroxide, potassium hydroxide, or lithium diisopropylamide deprotonates the alpha-carbon of the ketone reactant to form a nucleophilic enolate ion. This enolate subsequently attacks the electrophilic carbonyl carbon of the aldehyde-functionalized hydroxycyclohexylacetophenone intermediate. Following the initial addition, a dehydration step occurs, eliminating a water molecule to form an alpha,beta-unsaturated ketone structure. This newly formed double bond is conjugated with both the aromatic ring and the carbonyl group, creating an extended chromophore responsible for the redshifted absorption. The reaction conditions are mild, typically proceeding at temperatures between 0°C and 80°C with a reaction time ranging from 0.5 to 5 hours, depending on the steric hindrance of the specific ketone employed.
Impurity control is a critical aspect of this mechanistic pathway, particularly given the potential for side reactions such as self-condensation of the ketone or over-reaction of the aldehyde. The patent specifies a molar ratio of the aldehyde intermediate to the ketone of 1-2:1, which helps drive the reaction towards the desired cross-condensation product while minimizing homopolymerization. Furthermore, the choice of solvent plays a pivotal role in managing solubility and reaction kinetics; polar protic solvents like ethanol and methanol, as well as polar aprotic solvents like DMF and DMSO, are effective in stabilizing the transition states. Post-reaction purification is streamlined by the tendency of the final products to crystallize upon cooling, allowing for efficient isolation via filtration and recrystallization. This inherent crystallinity not only simplifies downstream processing but also ensures high chemical purity, which is essential for preventing yellowing and maintaining the long-term stability of the cured polymer matrix.
How to Synthesize Hydroxy Cyclohexyl Acetophenone Photoinitiator Efficiently
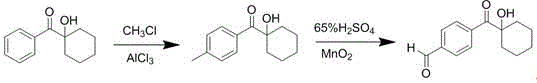
The synthesis protocol described in the patent offers a practical and scalable route for producing these advanced photoinitiators, beginning with the preparation of the key aldehyde intermediate. This precursor is synthesized by introducing a formyl group onto the benzene ring of hydroxycyclohexylacetophenone, typically via a Friedel-Crafts alkylation with methyl chloride followed by oxidation with manganese dioxide in the presence of sulfuric acid. Once the aldehyde intermediate is secured, the subsequent condensation step is straightforward, requiring only the mixing of the intermediate with the chosen ketone in a suitable alcoholic solvent. The reaction is initiated by the addition of a basic catalyst to adjust the pH to between 8 and 12, facilitating the formation of the enolate species necessary for the condensation. Detailed standardized synthesis steps for specific derivatives are provided in the guide below to assist R&D teams in replicating these results.
- Introduce an aldehyde group onto the benzene ring of hydroxycyclohexylacetophenone via Friedel-Crafts alkylation followed by oxidation.
- React the aldehyde-functionalized intermediate with a ketone containing alpha-hydrogen atoms using a basic catalyst at 0-80°C.
- Purify the resulting solid product through filtration, washing, recrystallization, and vacuum drying to obtain the final photoinitiator.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this novel synthesis route presents significant advantages for procurement managers and supply chain directors seeking to optimize costs and ensure material availability. The primary benefit lies in the simplicity of the chemical process, which eliminates the need for expensive transition metal catalysts or complex multi-step sequences often associated with high-performance photoinitiators. By utilizing commodity chemicals such as acetone, cyclohexanone, and common bases like sodium hydroxide, the raw material costs are drastically reduced compared to specialized proprietary initiators. Additionally, the reaction proceeds under mild conditions without the requirement for high-pressure equipment or cryogenic temperatures, further lowering the capital expenditure and operational energy costs associated with manufacturing facilities.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of inexpensive, widely available starting materials directly translate to lower production costs. The one-step condensation reaction minimizes processing time and labor, while the high yield and ease of purification via crystallization reduce waste disposal costs. This economic efficiency allows for competitive pricing strategies in the global market for UV curing additives, making high-performance LED-compatible initiators accessible for cost-sensitive applications.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals ensures a stable and resilient supply chain, mitigating the risks associated with sourcing niche intermediates that may be subject to geopolitical disruptions or capacity constraints. Since the synthesis does not depend on single-source proprietary reagents, manufacturers can easily qualify multiple suppliers for raw materials, thereby enhancing supply security. This robustness is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of large-scale industrial customers in the coatings and electronics sectors.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from laboratory bench scale to pilot production without significant modification. The use of common organic solvents like ethanol and methanol simplifies solvent recovery and recycling systems, aligning with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. Furthermore, the absence of heavy metals in the final product simplifies regulatory compliance for end-use applications in food packaging and medical devices, expanding the market potential for these photoinitiators.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating these new photoinitiators into existing formulation workflows.
Q: Why is this new photoinitiator better than traditional PI 184 for LED curing?
A: Traditional PI 184 has a maximum absorption at 333nm, which mismatches modern LED sources emitting above 365nm. This novel initiator shifts absorption to over 365nm, significantly improving double bond conversion rates from 10% to over 80% under LED irradiation.
Q: What types of ketones can be used in this synthesis?
A: The process is highly versatile and accommodates various ketones containing alpha-hydrogen atoms, including acetone, 3-pentanone, cyclohexanone, acetophenone, N-vinylpyrrolidone, and N-methylpyrrolidone, allowing for tunable properties.
Q: Is the manufacturing process scalable for industrial production?
A: Yes, the synthesis involves a straightforward one-step condensation reaction with readily available raw materials and simple purification via recrystallization, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxy Cyclohexyl Acetophenone Photoinitiator Supplier
As a leading CDMO expert in the fine chemical industry, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure required to bring this innovative photoinitiator technology to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume demands of global multinational corporations. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the highest standards of quality and consistency required for sensitive electronic and coating applications.
We invite you to collaborate with us to explore the full potential of these LED-curing photoinitiators for your specific product lines. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current formulation needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and product performance.
