Advanced Hydroxy Cyclohexyl Acetophenone Photoinitiators for High-Efficiency LED Curing Systems
Introduction to Next-Generation LED Photoinitiators
The rapid transition from traditional mercury-vapor lamps to energy-efficient LED light sources in the UV curing industry has created a critical demand for photoinitiators with tailored absorption spectra. According to patent CN111574350B, a significant technological breakthrough has been achieved in the development of novel hydroxy cyclohexyl acetophenone photoinitiators designed specifically to bridge the spectral gap between conventional initiators and modern LED emission profiles. Traditional photoinitiators, such as the widely used Type 184, exhibit a maximum absorption wavelength around 333nm, which falls short of the 365nm to 770nm emission range typical of current industrial LED systems. This mismatch results in suboptimal polymerization efficiency and lower double bond conversion rates, ultimately compromising the mechanical properties of the cured coating. The innovation described in this patent addresses this fundamental limitation by chemically modifying the hydroxycyclohexylacetophenone scaffold to extend its conjugation system, thereby red-shifting its absorption maximum to align perfectly with LED outputs.
This strategic molecular redesign not only enhances the photochemical performance under 365nm irradiation but also maintains the favorable solubility and stability characteristics required for high-performance coating formulations. For R&D directors and formulation chemists, this represents a viable pathway to upgrade existing product lines without overhauling entire curing infrastructure. The ability to achieve high conversion rates, reported in specific embodiments to reach up to 95% within 300 seconds under 365nm LED exposure, underscores the practical viability of these new compounds. Furthermore, the synthetic route outlined in the patent relies on robust, scalable organic transformations, suggesting that these advanced materials can be integrated into the supply chain with minimal friction. As the global market increasingly mandates lower energy consumption and higher throughput, adopting photoinitiators that maximize photon utilization becomes a competitive necessity for manufacturers of electronic chemicals and specialty coatings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on legacy photoinitiators like 184 in an era dominated by LED curing technology presents a multifaceted challenge for the coatings and adhesives industry. The primary deficiency lies in the spectral mismatch; when a photoinitiator with a peak absorption at 333nm is subjected to an LED source emitting primarily at 365nm or higher, the molar extinction coefficient at the excitation wavelength is significantly reduced. This physical limitation means that a substantial portion of the incident light energy is not absorbed by the initiator, leading to inefficient radical generation. Consequently, formulators are often forced to increase the loading level of the photoinitiator to compensate, which can introduce issues related to migration, odor, and yellowing in the final cured film. Moreover, incomplete curing due to poor light absorption can result in tacky surfaces and reduced chemical resistance, necessitating post-curing thermal treatments that negate the energy-saving benefits of LED technology. From a supply chain perspective, continuing to optimize formulations around inefficient initiators limits the potential for process speed improvements and increases the total cost of ownership for end-users who require faster line speeds and deeper cure depths.
The Novel Approach
The innovative strategy presented in patent CN111574350B circumvents these limitations through a rational design approach that extends the pi-conjugation of the hydroxycyclohexylacetophenone core. By introducing an aldehyde functional group onto the benzene ring and subsequently undergoing a condensation reaction with ketones containing alpha-hydrogen atoms, the resulting molecule possesses an enlarged conjugated system. This structural modification effectively lowers the energy gap between the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO), shifting the absorption maximum into the near-UV and visible region compatible with LED sources. 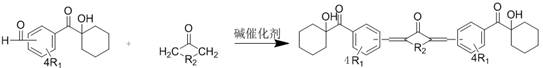 . This one-step condensation reaction is remarkably efficient, allowing for the rapid assembly of diverse derivatives by simply varying the ketone component. The versatility of this approach enables the fine-tuning of solubility, reactivity, and absorption characteristics to meet specific application requirements, whether for rigid substrates or flexible films. Unlike complex multi-step syntheses involving expensive transition metal catalysts, this method utilizes straightforward base-catalyzed chemistry, making it highly attractive for cost reduction in electronic chemical manufacturing and large-scale industrial production.
. This one-step condensation reaction is remarkably efficient, allowing for the rapid assembly of diverse derivatives by simply varying the ketone component. The versatility of this approach enables the fine-tuning of solubility, reactivity, and absorption characteristics to meet specific application requirements, whether for rigid substrates or flexible films. Unlike complex multi-step syntheses involving expensive transition metal catalysts, this method utilizes straightforward base-catalyzed chemistry, making it highly attractive for cost reduction in electronic chemical manufacturing and large-scale industrial production.
Mechanistic Insights into Base-Catalyzed Aldol Condensation
The core chemical transformation driving the synthesis of these advanced photoinitiators is a base-catalyzed aldol condensation, a fundamental reaction in organic synthesis known for its ability to form carbon-carbon bonds and extend conjugation. The mechanism initiates with the deprotonation of the alpha-carbon of the ketone reactant by a basic catalyst, such as sodium hydroxide, potassium hydroxide, or lithium diisopropylamide, generating a reactive enolate ion. This nucleophilic enolate then attacks the electrophilic carbonyl carbon of the aldehyde-functionalized hydroxycyclohexylacetophenone intermediate, forming a beta-hydroxy ketone adduct. Under the reaction conditions specified in the patent, typically ranging from 0°C to 80°C, this intermediate undergoes dehydration to yield the alpha,beta-unsaturated ketone product. The formation of this double bond is crucial as it serves as the conduit for electron delocalization across the molecule, which is directly responsible for the observed bathochromic shift in absorption. The choice of base and solvent plays a pivotal role in controlling the reaction kinetics and selectivity; for instance, using polar protic solvents like ethanol or methanol facilitates the dissolution of inorganic bases, while aprotic solvents like THF may be preferred for stronger, non-nucleophilic bases like LDA to prevent side reactions.
Impurity control is another critical aspect of this mechanistic pathway, particularly given the sensitivity of photoinitiators to trace contaminants that can act as radical scavengers. The patent specifies a pH range of 8 to 12 for the reaction system, which is optimized to drive the condensation forward while minimizing the risk of Cannizzaro reactions or self-polymerization of the aldehyde starting material. Post-reaction processing involves careful crystallization and recrystallization steps, which leverage the differences in solubility between the desired conjugated product and unreacted starting materials or oligomeric byproducts. The structural integrity of the final product is confirmed through rigorous analytical techniques such as 1H-NMR and mass spectrometry, ensuring that the conjugated system is intact and free from structural defects that could compromise photolytic efficiency. For procurement managers, understanding that the synthesis relies on well-understood equilibrium processes rather than exotic catalytic cycles provides confidence in the reproducibility and robustness of the supply. The ability to purify the product via simple filtration and drying further simplifies the downstream processing, reducing the environmental footprint associated with solvent-intensive chromatographic separations often required for more complex pharmaceutical intermediates.
How to Synthesize Hydroxy Cyclohexyl Acetophenone Photoinitiator Efficiently
The synthesis protocol detailed in the patent offers a streamlined pathway for producing these high-performance materials, beginning with the preparation of the key aldehyde intermediate. This precursor is typically generated by functionalizing a standard hydroxycyclohexylacetophenone molecule, often involving a Friedel-Crafts acylation followed by oxidation to install the formyl group at the para-position. Once the aldehyde building block is secured, the core condensation step can be executed under mild conditions using commercially available ketones. The reaction parameters are flexible, allowing operators to adjust temperature and catalyst loading to optimize yield and reaction time based on the specific steric and electronic properties of the chosen ketone. For a comprehensive guide on executing this synthesis with precision, please refer to the standardized operating procedures below.
- Functionalize the benzene ring of hydroxycyclohexylacetophenone with an aldehyde group via methylation and subsequent oxidation.
- Perform a condensation reaction between the aldehyde-functionalized intermediate and a ketone containing alpha-hydrogen atoms using a basic catalyst.
- Isolate the final photoinitiator product through filtration, recrystallization, and vacuum drying to ensure high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial standpoint, the adoption of this novel photoinitiator synthesis route offers compelling advantages for supply chain optimization and cost management. The primary driver for cost reduction lies in the simplicity of the chemical transformation; the condensation reaction proceeds in a single step with high atom economy, utilizing inexpensive and widely available feedstocks such as acetone, cyclohexanone, and acetophenone. This eliminates the need for costly proprietary reagents or precious metal catalysts that often characterize newer photochemical technologies, thereby insulating the production cost from volatility in the rare earth or precious metal markets. Furthermore, the reaction conditions are mild, typically requiring temperatures between 0°C and 80°C, which translates to lower energy consumption for heating and cooling compared to high-temperature pyrolysis or high-pressure hydrogenation processes. The ease of product isolation through crystallization rather than complex distillation or chromatography significantly reduces processing time and solvent usage, contributing to substantial cost savings in utility and waste treatment operations.
Enhanced supply chain reliability is another critical benefit derived from this technology. The starting materials are commodity chemicals with established global supply networks, reducing the risk of bottlenecks that can occur with specialized fine chemical intermediates. The robustness of the base-catalyzed reaction means that manufacturing can be scaled up rapidly from pilot batches to multi-ton production runs without requiring specialized reactor configurations or exotic containment systems. This scalability ensures that suppliers can respond quickly to surges in demand from the coatings and electronics sectors, maintaining continuity of supply even during periods of market fluctuation. Additionally, the environmental profile of the process is favorable; the use of aqueous or alcoholic solvents and the absence of heavy metals simplify wastewater treatment and regulatory compliance, mitigating the risk of production shutdowns due to environmental violations. For logistics planners, the solid nature of the final product facilitates safe and efficient storage and transportation, further enhancing the overall resilience of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photoinitiator technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding the specific performance metrics and synthesis requirements is essential for evaluating the feasibility of integrating these materials into existing manufacturing workflows. The answers below clarify the operational boundaries and potential benefits associated with this novel class of compounds.
Q: Why is this new photoinitiator better than traditional Type 184 for LED applications?
A: Traditional photoinitiator 184 has a maximum absorption wavelength of 333nm, which mismatches modern LED sources emitting above 365nm. This novel derivative extends conjugation to shift absorption above 365nm, significantly improving double bond conversion rates.
Q: What are the key raw materials required for this synthesis?
A: The process utilizes readily available hydroxycyclohexylacetophenone derivatives and common ketones such as acetone, cyclohexanone, or acetophenone, ensuring a stable and cost-effective supply chain.
Q: Is the manufacturing process scalable for industrial production?
A: Yes, the synthesis involves standard organic reactions like Friedel-Crafts acylation and aldol condensation, which are well-established and easily scalable from laboratory to multi-ton commercial production without complex equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxy Cyclohexyl Acetophenone Photoinitiator Supplier
As the demand for LED-curable materials continues to surge, partnering with an experienced chemical manufacturer is crucial for securing a stable supply of high-quality photoinitiators. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of global coating and adhesive formulators. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against critical performance parameters such as absorption maxima and initiation efficiency. We understand that consistency is key in industrial applications, and our advanced manufacturing facilities are equipped to handle the precise temperature and pH controls required for the base-catalyzed condensation reactions described in patent CN111574350B.
We invite potential partners to engage with our technical procurement team to discuss how these advanced photoinitiators can enhance your product portfolio. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of switching to our LED-compatible initiators. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your specific formulation needs. Whether you are developing high-speed printing inks, protective electronic coatings, or durable wood finishes, our expertise in fine chemical synthesis positions us as a strategic partner capable of delivering both innovation and reliability in equal measure.
