Advanced Microwave-Assisted Synthesis for High-Purity Ceftazidime Intermediates
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of critical antibiotic intermediates, particularly for third-generation cephalosporins like Ceftazidime. A significant breakthrough in this domain is documented in patent CN107513047B, which discloses a novel green process for synthesizing ceftazidime side chain acid active ester using a microwave-assisted method. This technology represents a paradigm shift from traditional thermal heating, leveraging electromagnetic energy to drive the condensation reaction between ceftazidime side chain acid and dithiodibenzothiazole. By replacing expensive reagents with cost-effective alternatives and optimizing solvent systems, this process addresses long-standing inefficiencies in API intermediate manufacturing. For R&D directors and procurement managers alike, understanding the nuances of this patent is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials at a competitive cost structure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ceftazidime side chain acid active ester has been plagued by several technical and economic bottlenecks that hinder large-scale commercial viability. Traditional routes often rely heavily on triphenylphosphine as a key reagent, which is not only prohibitively expensive but also generates triphenylphosphine oxide as a stoichiometric byproduct that is notoriously difficult to remove from the final product matrix. Furthermore, conventional processes frequently utilize dichloromethane or pure acetonitrile as solvents; dichloromethane poses significant environmental and safety hazards due to its toxicity and volatility, while pure acetonitrile often results in poor product color and lower purity profiles. These legacy methods typically require extended reaction times, sometimes lasting up to eight hours, and operate at suboptimal temperatures that fail to drive the reaction to completion, resulting in yields that hover around 80-90% and necessitate complex, multi-step purification procedures that erode profit margins.
The Novel Approach
In stark contrast, the innovative methodology outlined in the patent data introduces a streamlined workflow that fundamentally alters the reaction landscape through the strategic application of microwave chemistry. By substituting triphenylphosphine with triethyl phosphite, the process drastically reduces raw material costs while simultaneously simplifying the downstream workup, as the byproducts are far easier to manage. The implementation of a microwave reactor allows for precise control over energy input, with radiation power tuned between 80-300W, enabling the reaction to reach completion in a mere 30-60 minutes. This approach not only enhances productivity but also utilizes a tailored mixed solvent system of benzene and acetonitrile, which optimizes the solubility of reactants and facilitates superior crystallization of the final active ester, consistently achieving purity levels exceeding 99% and yields greater than 97%.
Mechanistic Insights into Microwave-Assisted Activation and Catalysis
The core of this technological advancement lies in the synergistic interaction between microwave irradiation and the specific catalytic system employed. Unlike conventional conductive heating, which relies on thermal gradients that can lead to localized overheating or incomplete reaction zones, microwave energy penetrates the reaction mixture uniformly, exciting polar molecules and accelerating the nucleophilic attack of the phosphite on the activated acid species. This rapid energy transfer is critical for activating the ceftazidime side chain acid in the presence of dithiodibenzothiazole, ensuring that the formation of the active thioester bond proceeds with high kinetic efficiency. The use of a composite alkaline catalyst, comprising a blend of agents such as triethylamine, pyridine, or aniline, creates a buffered pH environment between 7.5 and 11.5, which is essential for maintaining the stability of the reactive intermediates while promoting the desired condensation pathway over potential hydrolysis side reactions.
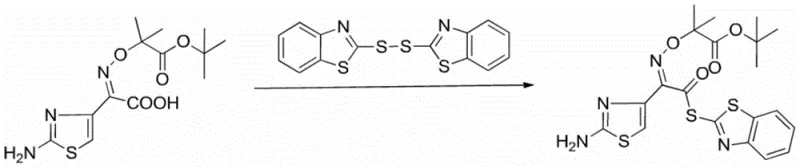
Furthermore, the meticulous control of solvent composition plays a pivotal mechanistic role in determining the physical state of the reaction mass and the ultimate quality of the isolate. The patent specifies a mass ratio of acetonitrile to benzene ranging from 1:7 to 7:1, with a preferred balance around 4.5:5.5, to prevent the formation of sticky, amorphous solids that occur in pure benzene or the low-yield outcomes observed in pure acetonitrile. This solvent engineering ensures that the product precipitates cleanly upon cooling, allowing for a simple suction filtration step followed by a methanol wash to remove residual impurities. The result is a highly crystalline product with a defined impurity profile, making it an ideal precursor for the subsequent coupling steps in the total synthesis of ceftazidime, thereby satisfying the rigorous quality standards demanded by regulatory bodies for antibiotic production.
How to Synthesize Ceftazidime Side Chain Acid Active Ester Efficiently
Implementing this microwave-assisted protocol requires precise adherence to the reaction parameters defined in the intellectual property to ensure reproducibility and safety. The process begins with the preparation of the solvent system and the addition of the solid reactants, followed by the controlled introduction of the liquid reagents under inert conditions. Operators must carefully monitor the microwave power output and internal temperature to avoid local carbonization, which can occur if the energy input is too aggressive. The following guide outlines the standardized operational procedure derived from the patent examples, serving as a foundational reference for process chemists aiming to replicate this high-efficiency route in a pilot or production setting.
- Prepare a mixed solvent system of benzene and acetonitrile with controlled water content (0.01-0.1%) and add ceftazidime side chain acid and dithiodibenzothiazole.
- Introduce a composite alkaline catalyst system and dropwise add triethyl phosphite while maintaining the temperature between 15-25°C.
- Execute the reaction in a microwave reactor at 80-300W power for 30-60 minutes, followed by cooling, filtration, and methanol washing.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this microwave-assisted synthesis route offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The substitution of high-cost reagents with commoditized chemicals directly impacts the bill of materials, creating a leaner cost structure that enhances competitiveness in the global API market. Moreover, the drastic reduction in reaction time translates to higher throughput capacity without the need for additional reactor volume, effectively debottlenecking production lines and improving asset utilization rates. These factors combined create a robust supply chain proposition that mitigates the risks associated with raw material price volatility and production delays.
- Cost Reduction in Manufacturing: The replacement of expensive triphenylphosphine with triethyl phosphite serves as a primary driver for cost optimization, eliminating the need for costly purification steps to remove phosphine oxides. Additionally, the energy efficiency of microwave heating compared to prolonged conventional reflux significantly lowers utility consumption per kilogram of product. The simplified workup procedure, involving direct filtration and washing rather than complex chromatographic separations, further reduces labor and solvent recovery costs, leading to substantial overall savings in the manufacturing budget.
- Enhanced Supply Chain Reliability: By utilizing readily available raw materials such as triethyl phosphite and common solvents like benzene and acetonitrile, the process minimizes dependency on specialized or scarce reagents that often cause supply disruptions. The robustness of the reaction conditions, with a wide tolerance for microwave power and time variations, ensures consistent batch-to-batch quality, reducing the incidence of out-of-specification results that can delay shipments. This reliability allows for more accurate forecasting and inventory planning, ensuring a steady flow of high-purity intermediates to downstream formulation partners.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this process, such as the avoidance of chlorinated solvents like dichloromethane and the reduction of hazardous waste generation, align perfectly with modern environmental regulations. The ability to scale microwave technology from laboratory to industrial reactors enables seamless technology transfer, facilitating rapid commercialization. Furthermore, the high purity of the crude product reduces the load on wastewater treatment facilities, supporting sustainable manufacturing practices and lowering the environmental compliance burden for production sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this microwave-assisted synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the operational advantages and quality benchmarks associated with this method. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: Why is triethyl phosphite preferred over triphenylphosphine in this synthesis?
A: Triethyl phosphite is significantly cheaper than triphenylphosphine and avoids the formation of difficult-to-remove triphenylphosphine oxide byproducts, thereby simplifying purification and reducing overall production costs.
Q: How does microwave assistance improve the yield of the active ester?
A: Microwave irradiation provides uniform and rapid heating, which accelerates the reaction kinetics and ensures complete conversion within 30-60 minutes, achieving yields exceeding 97% compared to traditional heating methods.
Q: What is the critical role of the benzene-acetonitrile solvent mixture?
A: The specific ratio of benzene to acetonitrile prevents the reaction mass from becoming sticky (a issue with pure benzene) and avoids the low yields associated with pure acetonitrile, ensuring optimal product crystallization and purity above 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceftazidime Side Chain Acid Active Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain a competitive edge in the pharmaceutical intermediate market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of ceftazidime side chain acid active ester adheres to the highest international standards for antibiotic synthesis.
We invite you to collaborate with us to leverage this cutting-edge microwave technology for your supply chain needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green process can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to drive efficiency and quality in your cephalosporin production.
