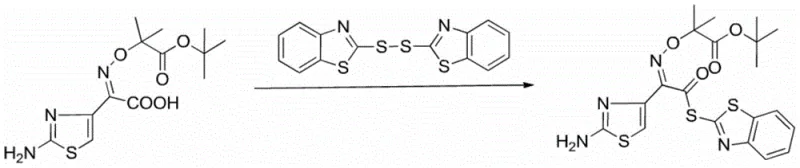
Advanced Microwave-Assisted Synthesis of Ceftazidime Side Chain Acid Active Ester for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for producing critical antibiotic intermediates, and Patent CN107513047B represents a significant leap forward in this domain. This patent discloses a green process for synthesizing ceftazidime side chain acid active ester by employing a microwave-assisted method, which fundamentally alters the kinetic profile of the reaction. By utilizing ceftazidime side chain acid and dithiodibenzothiazole as primary raw materials in a specific mixed solvent system, the process achieves remarkable efficiency. The core innovation lies in the substitution of traditional thermal heating with controlled microwave radiation at powers between 80-300W for durations of 30-60 min. This technological shift not only addresses the environmental concerns associated with long reaction times but also delivers a product with a yield exceeding 97 percent and a purity greater than 99 percent. For global procurement teams and R&D directors, this patent offers a validated route to high-quality intermediates essential for third-generation cephalosporin antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ceftazidime side chain acid active ester has been plagued by inefficiencies and high operational costs inherent to traditional thermal methods. Prior art techniques, such as those utilized by major pharmaceutical manufacturers, often rely on dichloromethane as a solvent and triphenylphosphine as a reagent, which presents multiple drawbacks. The use of dichloromethane poses significant environmental and safety challenges due to its toxicity and volatility, complicating waste treatment and increasing regulatory compliance costs. Furthermore, triphenylphosphine is an expensive reagent that drastically inflates the raw material bill, while often requiring reaction times as long as 3 to 8 hours to reach completion. Even with these extended durations, conventional methods frequently struggle with yields hovering around 79-90 percent and purity levels that necessitate complex purification steps to remove unreacted starting materials and byproducts dissolved in the solvent.
The Novel Approach
In stark contrast, the novel microwave-assisted approach described in the patent data introduces a paradigm shift by optimizing both the reagent profile and the energy input mechanism. By replacing the costly triphenylphosphine with inexpensive triethyl phosphite, the process immediately achieves a substantial reduction in production costs without compromising reaction efficacy. The implementation of microwave irradiation allows for rapid and uniform heating of the reaction mixture, accelerating the molecular interactions and reducing the total reaction time to merely 30-60 min. This method utilizes a tailored mixed solvent system of benzene and acetonitrile, which prevents the formation of sticky byproducts and ensures high solubility, leading to a streamlined post-treatment process. The result is a robust manufacturing protocol that consistently delivers yields above 97 percent and purity exceeding 99 percent, effectively solving the long-standing issues of low profitability and severe three-waste pollution associated with legacy processes.
Mechanistic Insights into Microwave-Assisted Activation
The success of this synthetic route relies heavily on the precise interaction between the microwave energy and the specific chemical environment created by the composite alkaline catalyst. The mechanism involves the activation of the ceftazidime side chain acid by the catalyst system, which typically comprises a combination of aniline, triethylamine, pyridine, or N,N-dimethylaniline. This composite catalyst acts as an acid-binding agent and activator, creating an optimal alkaline buffer range with a pH of 7.5-11.5 that is crucial for the nucleophilic attack. When microwave radiation is applied at 80-300W, the dielectric heating effect promotes the rapid formation of the active intermediate species. However, control is paramount; if the microwave power is too low, the reaction remains incomplete, whereas excessive power causes rapid temperature spikes leading to local carbonization of reactants. The patent specifies a narrow window of 30-60 min radiation time to ensure complete conversion while maintaining the structural integrity of the sensitive beta-lactam precursors.
Furthermore, the solvent dynamics play a critical mechanistic role in stabilizing the transition states and facilitating product isolation. The invention highlights that the mass ratio of acetonitrile to benzene must be carefully controlled, preferably at 4.5:5.5, to balance solubility and reaction rate. Pure benzene causes the material to become sticky, hindering filtration and lowering purity, while pure acetonitrile results in depressed yields. By maintaining the water content of this mixed solvent between 0.01-0.1 percent, the process prevents the hydrolysis of the active ester, which would otherwise degrade the product and reduce overall yield. This delicate balance of solvent polarity and moisture control ensures that the final active ester precipitates efficiently upon cooling, allowing for simple suction filtration and methanol washing to achieve the reported high purity specifications.
How to Synthesize Ceftazidime Side Chain Acid Active Ester Efficiently
Implementing this advanced synthesis route requires strict adherence to the optimized parameters regarding temperature, reagent addition rates, and microwave settings to ensure reproducibility at scale. The process begins with the preparation of the reaction mixture in a microwave-compatible reactor, where the precise ratio of solvents and catalysts sets the stage for high-yield conversion. Operators must monitor the dropwise addition of triethyl phosphite closely, maintaining the temperature between 15-25°C to prevent premature side reactions before the microwave energy is applied. Once the reagents are combined, the microwave reactor is engaged to drive the reaction to completion within the short 30-60 min window, followed by a controlled cooling phase to crystallize the product. For a detailed breakdown of the specific operational steps and safety protocols required for this synthesis, please refer to the standardized guide below.
- Prepare a mixed solvent system of benzene and acetonitrile (ratio 1: 7 to 7:1) containing ceftazidime side chain acid and dithiodibenzothiazole.
- Add a composite alkaline catalyst (e.g., triethylamine and pyridine) and maintain temperature between 15-25°C.
- Dropwise add triethyl phosphite over 30-120 minutes, then apply microwave radiation (80-300W) for 30-60 minutes.
- Cool the reaction mixture below 10°C, filter the crude product, and wash with methanol at 22-24°C to obtain the refined active ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this microwave-assisted process translates into tangible strategic advantages that extend beyond simple technical metrics. The primary driver of value is the significant cost reduction achieved by substituting expensive triphenylphosphine with cheap triethyl phosphite, which lowers the variable cost per kilogram of the active ester. This raw material optimization is compounded by the drastic reduction in reaction time, which increases the throughput capacity of existing manufacturing assets without the need for capital-intensive expansion. By shortening the production cycle from several hours to under one hour, facilities can respond more agilely to market demand fluctuations, thereby enhancing supply chain reliability and reducing the risk of stockouts for critical antibiotic components. Additionally, the simplified post-treatment process involving direct filtration and washing minimizes solvent consumption and waste generation, aligning with increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The replacement of high-cost reagents with economical alternatives like triethyl phosphite directly impacts the bottom line by lowering the bill of materials. Furthermore, the energy efficiency of microwave heating compared to prolonged thermal reflux reduces utility costs, while the high yield (>97 percent) minimizes the loss of valuable starting materials. These factors combine to create a highly cost-competitive manufacturing profile that allows for better pricing strategies in the global API market.
- Enhanced Supply Chain Reliability: The robustness of this process, characterized by its tolerance to specific solvent ratios and its rapid reaction kinetics, ensures consistent output quality and quantity. Shorter lead times for production batches mean that inventory levels can be optimized, reducing the working capital tied up in work-in-progress goods. This agility is crucial for maintaining continuous supply lines to downstream API manufacturers who depend on timely deliveries of ceftazidime intermediates to meet their own production schedules.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations like dropwise addition and filtration that are easily adapted from pilot to commercial scale. The reduction in hazardous solvent usage and the elimination of heavy metal catalysts simplify waste treatment procedures, ensuring compliance with green chemistry principles. This environmental stewardship not only mitigates regulatory risks but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this microwave-assisted synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production workflows.
Q: Why is triethyl phosphite preferred over triphenylphosphine in this synthesis?
A: Triethyl phosphite is significantly cheaper than triphenylphosphine, directly reducing raw material costs. Additionally, it facilitates a cleaner reaction profile under microwave conditions, contributing to higher yields (>97%) and purity (>99%) compared to conventional thermal methods.
Q: How does the microwave-assisted method improve productivity?
A: The microwave radiation power of 80-300W accelerates the reaction kinetics, reducing the reaction time from several hours (in traditional methods) to just 30-60 minutes. This drastic reduction in cycle time allows for higher throughput and improved productivity in manufacturing facilities.
Q: What is the role of the benzene-acetonitrile mixed solvent?
A: Using pure benzene leads to sticky materials affecting purity, while pure acetonitrile results in lower yields. A mixed solution (preferably 4.5:5.5 ratio) optimizes solubility and reaction efficiency, ensuring the product remains stable and easy to isolate with high purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceftazidime Side Chain Acid Active Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the production of life-saving antibiotics like ceftazidime. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative microwave-assisted techniques described in recent patents can be effectively translated into industrial reality. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of ceftazidime side chain acid active ester meets the highest international standards. Our commitment to quality assurance means that clients can rely on us for consistent supply without compromising on the chemical integrity required for downstream coupling reactions.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both cost and performance in the competitive pharmaceutical landscape.
