Advanced Continuous Synthesis Strategy for High-Purity Sildenafil Intermediates and Commercial Scalability
Advanced Continuous Synthesis Strategy for High-Purity Sildenafil Intermediates and Commercial Scalability
The pharmaceutical landscape for erectile dysfunction treatments continues to evolve, driven by the need for more efficient and cost-effective manufacturing processes for key active pharmaceutical ingredients like Sildenafil. Patent CN111689969A, published on September 22, 2020, introduces a significant technological breakthrough in the preparation of Sildenafil by shifting from traditional step-by-step isolation methods to a streamlined continuous synthesis approach. This innovation addresses critical bottlenecks in the supply chain by drastically reducing reaction times and eliminating the need for intermediate purification, thereby enhancing overall production throughput. For R&D directors and procurement specialists, this patent represents a pivotal opportunity to optimize the manufacturing of high-purity pharmaceutical intermediates while mitigating the operational complexities associated with legacy synthetic routes. The core of this advancement lies in the telescoping of the amidation and chlorosulfonation steps, creating a seamless workflow that maintains rigorous quality standards without the burden of excessive processing time.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sildenafil has been plagued by inefficient protocols that impose severe constraints on manufacturing scalability and cost efficiency. As detailed in prior art such as US5955611, the conventional pathway involves a multi-step sequence starting with the chlorosulfonation of compound VII to obtain compound VIII, followed by a protracted N-sulfonation reaction. A major drawback of this legacy method is the excessive reaction duration; specifically, the chlorosulfonation step requires an impractical 18 hours to reach completion, while the subsequent N-sulfonation utilizes expensive activating agents like CDI and demands a staggering 72 hours of reaction time. Furthermore, these processes often necessitate stringent environmental controls, such as the use of dust-free flasks and dust-free hydrochloric acid, which significantly inflate operational expenditures and complicate facility management. The reliance on isolating and purifying intermediates at each stage not only increases material loss but also introduces multiple points of failure where impurities can be introduced, ultimately compromising the yield and economic viability of large-scale production runs.
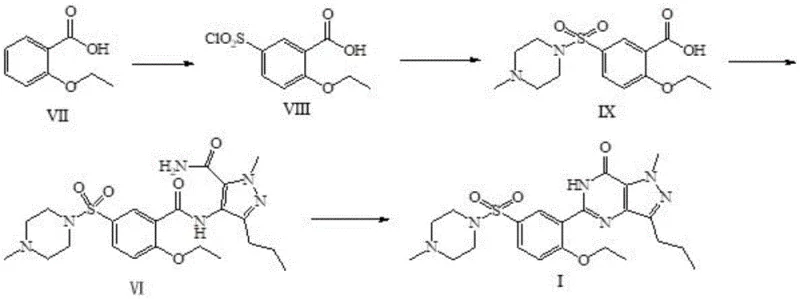
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the methodology disclosed in CN111689969A employs a continuous synthesis strategy that fundamentally restructures the reaction workflow to maximize efficiency. Instead of isolating the amidation product (Compound IV), the process directs the crude solution immediately into the chlorosulfonation reactor, effectively telescoping two distinct chemical transformations into a single operational sequence. This elimination of intermediate isolation not only saves substantial time but also reduces solvent consumption and waste generation, aligning with modern green chemistry principles. The new route bypasses the need for expensive activators and reduces the chlorosulfonation timeframe from nearly a day to a mere 0.5 to 4.0 hours, representing a monumental leap in process intensification. By simplifying the operational parameters and removing the requirement for specialized dust-free environments, this novel approach offers a robust platform for reliable sildenafil intermediate supplier networks seeking to enhance their competitive edge through superior process engineering and cost reduction in API manufacturing.
Mechanistic Insights into Telescoped Amidation and Chlorosulfonation
The chemical elegance of this new process is rooted in the precise control of reaction conditions that allow for the direct conversion of Compound III to Compound V without degradation. The initial step involves the amidation of Compound III with Compound II in a solvent system such as dichloromethane or tetrahydrofuran, facilitated by an aliphatic tertiary amine like triethylamine at temperatures between 0-40°C. Crucially, rather than quenching and isolating the resulting Compound IV, the reaction mixture is directly introduced into a mixture of chlorosulfonic acid and thionyl chloride. This direct addition is performed at a controlled temperature range of 25-60°C, ensuring that the highly reactive sulfonyl chloride functionality is generated efficiently within a 0.5 to 4.0 hour window. The mechanistic advantage here is the preservation of the reactive intermediate in solution, preventing hydrolysis or decomposition that might occur during workup procedures, thus maintaining high chemical integrity for the subsequent sulfonamide formation.
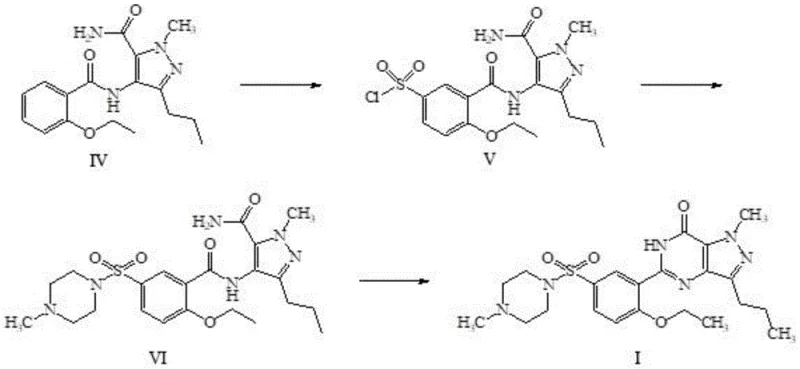
Following the formation of the sulfonyl chloride intermediate (Compound V), the process proceeds to the N-sulfonation and cyclization phases with equal precision. N-methylpiperazine is introduced to the crude Compound V solution to form the sulfonamide linkage, a reaction that completes within 0 to 4 hours under mild conditions of 5-60°C. The final cyclization to form the pyrazolo[4,3-d]pyrimidin-7-one core is triggered by the addition of a base, such as sodium hydroxide or potassium carbonate, in an alcoholic solvent at elevated temperatures of 60-100°C. This thermal activation drives the intramolecular condensation to closure, yielding the target Sildenafil structure. The impurity profile is tightly managed throughout this sequence because the continuous nature of the synthesis minimizes the exposure of sensitive intermediates to atmospheric moisture or contaminants, ensuring that the final product consistently meets stringent purity specifications required for pharmaceutical applications without the need for extensive downstream purification.
How to Synthesize Sildenafil Efficiently
The implementation of this continuous synthesis route requires careful attention to solvent selection and temperature gradients to ensure optimal yield and safety. The patent outlines a clear progression where the choice of organic solvents, such as dichloromethane for the initial steps and aliphatic alcohols for the cyclization, plays a critical role in solubility and reaction kinetics. Operators must maintain strict temperature controls, particularly during the exothermic chlorosulfonation phase, to prevent side reactions while maximizing the conversion rate. The detailed standardized synthesis steps below provide a comprehensive guide for replicating this high-efficiency protocol in a pilot or commercial setting, ensuring that the theoretical benefits of the patent are fully realized in practical application.
- Dissolve Compound III in an organic solvent like dichloromethane, add Compound II and a tertiary amine base to perform amidation at 0-40°C, yielding Compound IV solution.
- Directly add the crude Compound IV solution into a chlorosulfonic acid-thionyl chloride mixture for chlorosulfonation at 25-60°C without prior purification to obtain Compound V.
- React Compound V with N-methylpiperazine, concentrate, then add alkali and solvent to heat at 60-100°C for cyclization, followed by acidification to isolate Sildenafil.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this continuous synthesis methodology offers profound strategic advantages that extend far beyond simple chemical optimization. By fundamentally altering the production workflow to eliminate intermediate isolation, the process inherently reduces the number of unit operations required, which translates directly into lower labor costs and reduced equipment occupancy time. The removal of expensive activating agents like CDI, which were mandatory in previous generations of synthesis, results in a substantial reduction in raw material costs, allowing for more competitive pricing structures in the global market. Additionally, the drastic shortening of reaction times—from days to mere hours—significantly enhances the throughput capacity of existing manufacturing facilities, enabling suppliers to respond more agilely to fluctuating market demands without the need for capital-intensive plant expansions. This operational agility is crucial for maintaining supply continuity in the volatile pharmaceutical sector.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of costly reagents and the reduction of processing time. By removing the need for CDI and shortening the chlorosulfonation step from 18 hours to under 4 hours, the process achieves significant cost savings in both material consumption and energy usage. The telescoped nature of the reaction also minimizes solvent losses associated with multiple workup and drying stages, further driving down the variable cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route inherently reduces the risk of production delays caused by complex purification bottlenecks. With fewer isolation steps, there are fewer opportunities for yield loss or quality deviations, leading to a more predictable and stable output volume. This reliability is essential for securing long-term contracts with major pharmaceutical companies that require guaranteed delivery schedules and consistent quality batches for their own formulation lines.
- Scalability and Environmental Compliance: The process is explicitly designed for large-scale production, avoiding the strict dust-free conditions that limit the scalability of older methods. The reduction in reaction time and the use of standard solvents facilitate easier scale-up from pilot plants to multi-ton commercial reactors. Furthermore, the decreased generation of waste due to fewer purification steps aligns with increasingly stringent environmental regulations, reducing the burden on waste treatment facilities and lowering the overall environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Sildenafil synthesis route. These answers are derived directly from the experimental data and technical disclosures within patent CN111689969A, providing clarity on reaction parameters, yield expectations, and scalability factors. Understanding these details is vital for technical teams evaluating the feasibility of integrating this continuous process into their existing manufacturing portfolios to achieve higher efficiency and lower production costs.
Q: How does the new continuous method improve reaction time compared to conventional protocols?
A: The novel method drastically reduces the chlorosulfonation reaction time from 18 hours in prior art to merely 0.5-4.0 hours. Furthermore, it eliminates the 72-hour N-sulfonation step requiring expensive CDI activators found in older US patents, replacing it with a more efficient telescoped process.
Q: What are the critical purity specifications achieved in the described embodiments?
A: The patent embodiments demonstrate consistent high purity, with reported values ranging from 99.49% to 99.68%. This high quality is achieved through the continuous method which minimizes intermediate handling and potential contamination during isolation steps.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the invention explicitly states suitability for large-scale production. By adopting a continuous synthesis method that avoids separating and purifying intermediates, the process simplifies operations, improves production efficiency, and reduces the strict environmental controls (like dust-free flasks) required by previous methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sildenafil Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the continuous synthesis technologies described in CN111689969A and are fully equipped to leverage these advancements for our global partners. As a premier CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are designed to handle complex telescoped reactions with precision, adhering to stringent purity specifications and utilizing rigorous QC labs to guarantee that every batch of Sildenafil intermediate meets the highest international standards. We are committed to delivering not just a product, but a comprehensive supply solution that integrates cutting-edge process chemistry with reliable logistics.
We invite forward-thinking pharmaceutical companies to collaborate with us to unlock the full commercial potential of this optimized synthesis route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this continuous method can reduce your overall cost of goods sold. We encourage you to contact us today to discuss specific COA data and route feasibility assessments, ensuring that your supply chain is fortified with the most advanced and cost-effective manufacturing strategies available in the industry.
