Advanced Cyclization Technology for Sildenafil: Enhancing Commercial Scalability and Purity
Advanced Cyclization Technology for Sildenafil: Enhancing Commercial Scalability and Purity
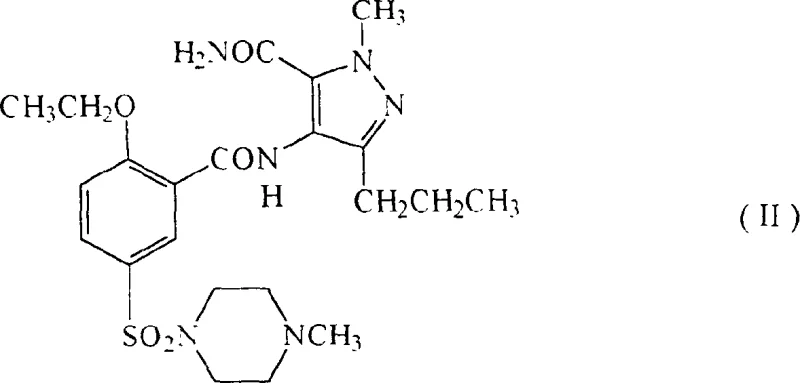
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with economic efficiency, particularly for high-volume active pharmaceutical ingredients like Sildenafil. Patent CN1106399C introduces a transformative approach to the preparation of 5-[2-ethoxy-5-(4-methylpiperazin-1-ylsulfonyl)phenyl]-1-methyl-3-n-propyl-1,6-dihydro-7H-pyrazolo[4,3-d]pyrimidin-7-one. This specific patent data reveals a novel cyclization strategy that converts a diamide precursor directly into the final therapeutic agent with exceptional efficiency. Unlike traditional methods that often require multiple purification stages to remove impurities and by-products, this innovation allows for the direct isolation of the product meeting clinical quality standards. The technical breakthrough lies in the specific selection of reaction conditions, including the use of defined bases and solvents, which drive the cyclization of the intermediate of structural formula (II) to completion. For R&D directors and procurement managers, understanding the nuances of this patent is critical for evaluating potential supply chain partners who can leverage this intellectual property to deliver cost-effective and high-quality intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sildenafil and related pyrazolo-pyrimidinone derivatives has been plagued by inefficient cyclization steps that result in suboptimal yields and complex impurity profiles. Conventional processes, such as those disclosed in earlier patents like EP-A-0463756, often rely on harsh conditions or multi-step sequences that degrade the overall economic viability of the manufacturing process. In these traditional routes, the conversion of the acyclic precursor to the cyclic final product frequently generates significant amounts of side products, necessitating extensive downstream purification. This not only increases the consumption of solvents and reagents but also extends the production cycle time, creating bottlenecks in the supply chain. Furthermore, the reliance on chromatographic purification for final polishing is often impractical on a multi-ton commercial scale due to the high costs associated with silica gel and solvent recovery. These limitations directly impact the cost of goods sold and can lead to supply inconsistencies, which are critical concerns for procurement managers managing global API inventories.
The Novel Approach
In stark contrast, the methodology outlined in patent CN1106399C offers a streamlined pathway that addresses these historical inefficiencies through a highly optimized cyclization reaction. The core of this innovation is the treatment of the diamide intermediate of structural formula (II) under specific basic conditions, which promotes rapid and clean ring closure. The patent data indicates that by utilizing bases such as potassium tert-butoxide in solvents like tert-butanol, the reaction can proceed with yields as high as 95% under optimized concentrations. This represents a substantial improvement over the 27.6% overall yield reported in prior art when calculated from similar starting materials. The ability to obtain the product directly in clinical quality without subsequent purification steps is a game-changer for commercial manufacturing. It implies a drastic reduction in unit operations, lower energy consumption, and a smaller environmental footprint. For supply chain heads, this translates to a more reliable and scalable production process that can meet fluctuating market demands without the risk of yield losses associated with complex purification workflows.
Mechanistic Insights into Base-Catalyzed Cyclization
The chemical elegance of this process lies in the mechanistic pathway facilitated by the chosen base and solvent system. The cyclization involves the intramolecular nucleophilic attack of the amide nitrogen onto the adjacent carbonyl carbon, followed by dehydration to form the pyrimidinone ring. The presence of a strong base, such as an alkali metal alkoxide, is crucial for deprotonating the amide nitrogen, thereby increasing its nucleophilicity and driving the equilibrium towards the cyclic product. The patent specifies a wide range of suitable bases, including alkali metal hydrides, hydroxides, and carbonates, allowing for flexibility in process optimization based on available raw materials. The solvent choice also plays a pivotal role; polar aprotic solvents or alcohols like tert-butanol help stabilize the transition state and solubilize the ionic intermediates. This mechanistic understanding is vital for R&D teams aiming to replicate or scale this process, as slight deviations in base strength or solvent polarity can significantly impact the reaction kinetics and impurity profile. The robustness of this mechanism ensures that the process remains consistent even when scaled from laboratory to pilot plant levels.
Controlling the impurity profile is another critical aspect of this mechanistic design. In many cyclization reactions, over-reaction or decomposition of the sensitive pyrazole moiety can lead to difficult-to-remove impurities. However, the conditions described in the patent, such as maintaining specific temperature ranges between 60°C to 105°C and controlling the molar equivalents of the base, minimize these side reactions. The data shows that using 1.1 to 2.0 molar equivalents of base is sufficient to drive the reaction without causing significant degradation. Furthermore, the work-up procedure involving neutralization and filtration is designed to precipitate the product while leaving soluble impurities in the mother liquor. This selective precipitation is a key factor in achieving high purity without chromatography. For quality assurance teams, this means that the critical quality attributes of the API can be controlled primarily through the reaction parameters rather than relying solely on end-of-line testing and reprocessing. This proactive approach to quality by design is essential for maintaining regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Sildenafil Efficiently
Implementing this synthesis route requires a precise adherence to the reaction parameters defined in the patent to ensure optimal yield and purity. The process begins with the preparation of the diamide intermediate, which must be of high quality to prevent the carryover of impurities into the final cyclization step. Once the intermediate is ready, it is suspended in a suitable solvent such as tert-butanol, and a pre-measured amount of base like potassium tert-butoxide is added under stirring. The mixture is then heated to reflux for a specified duration, typically around 8 hours, to ensure complete conversion. Following the reaction, the mixture is cooled, and water is added to dissolve the inorganic salts. The product is then precipitated by adjusting the pH to neutral using dilute acid, followed by filtration and drying. This straightforward protocol eliminates the need for complex extraction or chromatographic steps, making it highly attractive for industrial application. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this efficient process.
- Prepare the diamide intermediate of structural formula (II) through chlorosulfonation and subsequent coupling reactions.
- Conduct the cyclization reaction in the presence of a suitable base such as potassium tert-butoxide in a solvent like tert-butanol.
- Isolate the final Sildenafil product directly from the reaction mixture via neutralization and filtration, achieving clinical quality standards.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this cyclization technology offers significant strategic advantages for procurement and supply chain management. The primary benefit is the substantial reduction in manufacturing costs driven by the simplified process flow. By eliminating the need for extensive purification steps such as column chromatography, manufacturers can save significantly on solvent usage, silica gel, and labor hours. This efficiency gain directly translates to a lower cost of goods sold, allowing for more competitive pricing in the global API market. Additionally, the high yield reported in the patent means that less raw material is required to produce the same amount of final product, further enhancing cost efficiency. For procurement managers, this implies a more stable pricing structure and the potential for long-term cost savings agreements with suppliers who utilize this technology. The reduction in process complexity also lowers the risk of batch failures, ensuring a more consistent supply of material.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and complex purification sequences significantly lowers the operational expenditure associated with Sildenafil production. Traditional methods often require expensive catalysts and multiple recrystallization steps to achieve pharmaceutical grade purity, which adds considerable cost to the final product. In contrast, this base-catalyzed method uses readily available and inexpensive reagents like potassium tert-butoxide and tert-butanol. The direct isolation of the product from the reaction mixture reduces the consumption of utilities such as steam for distillation and electricity for extended processing times. Furthermore, the reduction in solvent waste lowers the costs associated with environmental compliance and waste disposal. These cumulative savings create a robust economic model that supports sustainable manufacturing practices while maintaining high profit margins.
- Enhanced Supply Chain Reliability: The simplicity and robustness of this synthetic route contribute to a more reliable supply chain by reducing the number of potential failure points. Complex multi-step syntheses are prone to delays caused by equipment downtime, reagent shortages, or quality deviations at intermediate stages. By consolidating the final steps into a single, high-yield cyclization, the overall lead time for production is drastically shortened. This agility allows manufacturers to respond more quickly to changes in market demand, ensuring that inventory levels are maintained without the need for excessive safety stock. For supply chain heads, this reliability is crucial for maintaining continuous production schedules for downstream formulation plants. The use of common, commercially available solvents and reagents also mitigates the risk of supply disruptions caused by the scarcity of specialized chemicals.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often introduces challenges related to heat transfer, mixing, and safety. The conditions described in this patent, such as moderate temperatures and the use of non-hazardous bases, are inherently safer and easier to scale. The absence of toxic heavy metals simplifies the environmental compliance process, as there is no need for rigorous testing and removal of metal residues to meet strict regulatory limits. This aligns with the growing industry trend towards green chemistry and sustainable manufacturing. The reduced solvent load and waste generation make the process more environmentally friendly, which is increasingly important for corporate social responsibility goals. For organizations looking to expand their production capacity, this technology offers a clear path to commercial scale-up of complex pharmaceutical intermediates without significant capital investment in specialized waste treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation. They are intended to provide clarity for technical teams evaluating the feasibility of this route for their specific manufacturing contexts. Understanding these details is essential for making informed decisions about process adoption and supplier qualification. The information covers key aspects such as yield optimization, reagent selection, and quality control measures that are critical for successful implementation.
Q: What is the primary advantage of the cyclization method in patent CN1106399C?
A: The primary advantage is the ability to obtain Sildenafil meeting clinical quality standards directly from the reaction mixture without the need for subsequent complex purification steps, significantly improving overall yield.
Q: Which bases are preferred for this cyclization reaction?
A: Preferred bases include alkali metal alkoxides such as potassium tert-butoxide and sodium ethoxide, as well as metal hydrides like sodium hydride, which facilitate high conversion rates.
Q: How does this process impact commercial scalability?
A: By simplifying the work-up procedure and eliminating extensive chromatography, the process reduces processing time and solvent consumption, making it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sildenafil Supplier
The technical potential of the cyclization process described in patent CN1106399C is immense, offering a pathway to high-purity Sildenafil with superior economic efficiency. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to life. Our facility is equipped with rigorous QC labs and stringent purity specifications that ensure every batch meets the highest international standards. We understand the critical nature of API intermediates in the pharmaceutical supply chain and are committed to delivering materials that facilitate the seamless production of final drug products. Our team of expert chemists is well-versed in the nuances of base-catalyzed cyclizations and can optimize this process to meet your specific volume and quality requirements.
We invite you to engage with our technical procurement team to discuss how we can support your supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this optimized manufacturing route. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project timelines. Our goal is to be your strategic partner in reducing lead time for high-purity pharmaceutical intermediates and ensuring the long-term success of your product portfolio. Let us help you engineer a more efficient and reliable supply chain for your critical pharmaceutical ingredients.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
