Advanced Catalytic Synthesis of Quinoxaline Compounds for Commercial Scale-up
Introduction to Green Catalytic Synthesis of Quinoxalines
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for sustainable and efficient synthetic routes. A pivotal advancement in this domain is documented in patent CN103145630A, which introduces a novel method for the catalytic synthesis of quinoxaline compounds. Quinoxalines are a critical class of nitrogen-containing benzoheterocyclic scaffolds widely utilized in the pharmaceutical and agrochemical industries due to their potent biological activities, including antibacterial and anticancer properties. Traditionally, the synthesis of these valuable intermediates has relied heavily on organic solvents and transition metal catalysts, which pose substantial environmental and operational challenges. The technology described in this patent represents a paradigm shift by utilizing a specialized polysulfonate ionic liquid catalyst in an aqueous medium. This approach not only aligns with the principles of green chemistry but also offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. By leveraging this innovation, manufacturers can achieve high purity standards while drastically reducing the ecological footprint associated with traditional heterocyclic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of quinoxaline derivatives has been achieved through the condensation of o-phenylenediamine with 1,2-dicarbonyl compounds in organic solvents such as acetonitrile, ethanol, or acetic acid. These conventional protocols typically employ catalysts like copper sulfate pentahydrate (CuSO4·5H2O), gallium triflate (Ga(OTf)3), or molecular iodine (I2). While effective in a laboratory setting, these methods present severe drawbacks when translated to industrial production. The reliance on volatile organic compounds (VOCs) creates significant safety hazards regarding flammability and toxicity, necessitating expensive containment and ventilation systems. Furthermore, the use of heavy metal catalysts introduces the risk of metal residue in the final product, which is unacceptable for pharmaceutical applications and requires costly downstream purification steps to meet regulatory limits. The corrosive nature of some of these catalysts and solvents also accelerates equipment degradation, leading to increased maintenance costs and potential production downtime. Consequently, there is a pressing industry demand for a cleaner, safer, and more economically viable alternative that eliminates these bottlenecks.
The Novel Approach
The methodology outlined in patent CN103145630A addresses these critical pain points by introducing a task-specific ionic liquid system that functions as both a catalyst and a phase-transfer agent in water. As illustrated in the general reaction scheme below, the process facilitates the condensation of o-phenylenediamine and various 1,2-dicarbonyl compounds under remarkably mild conditions.
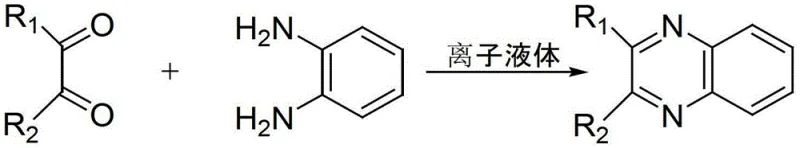
This novel approach utilizes water as the primary reaction solvent, comprising 50-90% of the total material mass, which inherently eliminates the fire risks and environmental toxicity associated with organic solvents. The reaction proceeds efficiently at temperatures between 40-60°C and atmospheric pressure, significantly lowering energy consumption compared to high-temperature reflux methods. The simplicity of the workup procedure—filtration followed by ethanol recrystallization—streamlines the production workflow, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing. By replacing hazardous reagents with a benign aqueous system, this technology offers a sustainable solution that maintains high yields while ensuring operator safety and environmental compliance.
Mechanistic Insights into Polysulfonate Ionic Liquid Catalysis
The cornerstone of this innovative synthesis is the unique structure of the polysulfonate ionic liquid catalyst. Unlike traditional Brønsted acidic ionic liquids that may possess only a single acidic site, the catalyst employed in this patent features a multi-sulfonic acid functionalized architecture. The structural formula, depicted below, reveals a cationic core decorated with multiple sulfonic acid (-SO3H) groups attached via alkyl chains.
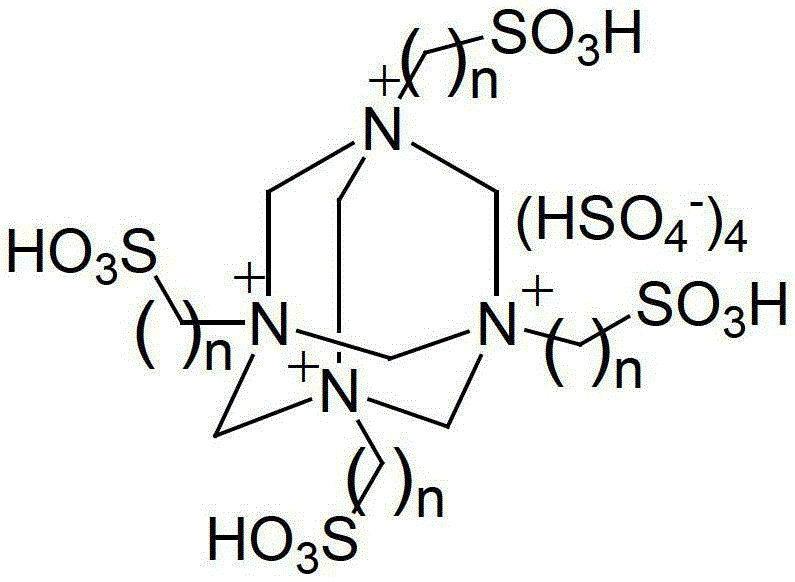
This multi-functional design results in a significantly higher acid density, which translates to superior catalytic activity. The presence of multiple acidic protons allows for the simultaneous activation of both the carbonyl oxygen of the 1,2-dicarbonyl substrate and the amino groups of the o-phenylenediamine. This dual activation lowers the energy barrier for the nucleophilic attack and subsequent cyclization steps, driving the reaction to completion rapidly even at lower temperatures. Furthermore, the ionic nature of the catalyst ensures excellent solubility in the aqueous reaction medium while remaining immiscible with the precipitating quinoxaline product, facilitating easy separation. From an impurity control perspective, the mild reaction conditions (40-60°C) minimize the formation of thermal degradation byproducts and polymerization side reactions that often plague high-temperature syntheses. The specific structure where n equals 3 or 4 provides an optimal balance between steric accessibility and acidity, ensuring that the catalyst remains stable and active throughout multiple reaction cycles.
How to Synthesize Quinoxaline Derivatives Efficiently
Implementing this synthesis route in a production environment requires adherence to specific stoichiometric and operational parameters to maximize yield and catalyst recovery. The process is designed to be operationally simple, requiring standard reactor equipment without the need for specialized high-pressure or cryogenic setups. The key to success lies in the precise control of the catalyst loading and the efficient management of the mother liquor for recycling purposes. The following guide outlines the standardized procedure derived from the patent examples to ensure consistent quality and performance.
- Prepare the reaction mixture by combining o-phenylenediamine and a 1,2-dicarbonyl compound in a 1: 1 molar ratio with 5-10% molar equivalent of the polysulfonate ionic liquid catalyst in water.
- Heat the mixture to a temperature between 40-60°C and stir vigorously for 20 to 120 minutes to allow the condensation reaction to proceed to completion.
- Filter the reaction product, wash the filter cake with water, and recrystallize the residue using ethanol to obtain the pure quinoxaline compound while recycling the filtrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this polysulfonate ionic liquid technology offers transformative economic and logistical benefits. The shift from organic solvents to water fundamentally alters the cost structure of quinoxaline production by removing the expenses associated with solvent purchase, recovery, and hazardous waste disposal. Moreover, the elimination of heavy metal catalysts removes the need for expensive scavenging resins or complex extraction processes to meet residual metal specifications, thereby simplifying the supply chain for raw materials.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic simplification of the downstream processing workflow. Since the product precipitates directly from the aqueous reaction mixture, the isolation step is reduced to a simple filtration, bypassing energy-intensive distillation or chromatography. Additionally, the catalyst exhibits exceptional stability, allowing the filtrate containing the ionic liquid and unreacted starting materials to be recycled directly for subsequent batches. This closed-loop capability means that the effective consumption of the catalyst is negligible over time, leading to substantial long-term savings on reagent costs without compromising on reaction efficiency or yield.
- Enhanced Supply Chain Reliability: Relying on water as a solvent mitigates the supply chain risks associated with volatile organic solvents, which are often subject to strict transportation regulations and price volatility. The raw materials required, specifically o-phenylenediamine and various 1,2-dicarbonyl compounds, are commodity chemicals with robust global availability. The mild reaction conditions also reduce the wear and tear on production equipment, minimizing unplanned maintenance shutdowns and ensuring a more consistent output schedule. This reliability is crucial for maintaining the continuity of supply for downstream API manufacturers who depend on timely delivery of high-quality intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often introduces safety and environmental hurdles, particularly when dealing with exothermic reactions in organic media. This aqueous-based protocol inherently possesses a higher heat capacity, making temperature control easier and safer on a large scale. The absence of VOC emissions aligns perfectly with increasingly stringent environmental regulations, reducing the burden on waste treatment facilities. The ability to recycle the catalyst system up to five times without treatment further minimizes the generation of chemical waste, positioning this method as a leader in sustainable manufacturing practices for high-purity quinoxaline derivatives.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its integration into existing production lines. The following questions address common inquiries regarding the catalyst's performance, the scope of applicable substrates, and the practical aspects of implementation. These insights are derived directly from the experimental data and technical specifications provided in the patent literature to ensure accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of using polysulfonate ionic liquids over traditional metal catalysts?
A: Polysulfonate ionic liquids offer superior acid density and catalytic activity without the risk of heavy metal contamination. Unlike traditional catalysts like CuSO4 or Ga(OTf)3, they operate effectively in water, eliminating the need for corrosive organic solvents and simplifying the purification process.
Q: Can the ionic liquid catalyst be recovered and reused in this synthesis method?
A: Yes, the patent data confirms that the polysulfonate ionic liquid catalyst along with unreacted raw materials in the filtrate can be recycled and reused up to 5 times without significant treatment, maintaining high catalytic yields throughout the cycles.
Q: What represents the optimal reaction conditions for this quinoxaline synthesis?
A: The optimal conditions involve a reaction temperature of 40-60°C and a reaction time of 20-120 minutes. The process uses water as the solvent (50-90% of total mass) and operates at atmospheric pressure, making it highly energy-efficient and safe for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxaline Supplier
The technological advancements detailed in patent CN103145630A underscore the immense potential for optimizing the production of quinoxaline scaffolds. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory methodologies into robust, commercial-grade manufacturing processes. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling aqueous and ionic liquid systems safely and efficiently. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of quinoxaline intermediate meets the exacting standards required by the global pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this green synthesis technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this catalytic route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for our quinoxaline portfolio and to discuss route feasibility assessments for your target molecules. Let us help you secure a sustainable and cost-effective supply chain for your critical intermediates.
