Advanced Base-Catalyzed Synthesis of Quinoxaline-3-one Derivatives for Commercial Scale-up
Advanced Base-Catalyzed Synthesis of Quinoxaline-3-one Derivatives for Commercial Scale-up
The pharmaceutical and agrochemical industries constantly seek robust synthetic routes for heterocyclic scaffolds that offer both high purity and economic viability. A significant breakthrough in this domain is detailed in patent CN111018795B, which discloses a novel method for synthesizing quinoxaline-3-one derivatives under alkaline conditions. This technology addresses a longstanding challenge in organic synthesis: the preferential formation of the 3-keto isomer over the thermodynamically favored 2-keto isomer. By utilizing a simple organic base catalysis system, this process achieves exceptional regioselectivity ratios reaching up to 11:1, drastically reducing the burden of purification. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing of complex heterocyclic intermediates used in drug discovery and crop protection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoxaline derivatives has relied heavily on condensation reactions between o-phenylenediamines and various carbonyl sources. However, when asymmetric substrates are employed, traditional methods suffer from poor regioselectivity, often yielding nearly equimolar mixtures of isomers. For instance, earlier approaches utilized microwave-assisted reactions in methanol/acetic acid systems at high temperatures (160°C), which, while fast, resulted in a regioisomer ratio of approximately 1:1. This lack of selectivity necessitates energy-intensive chromatographic separations, inflating production costs and generating substantial chemical waste. Furthermore, alternative strategies involving in-situ iodination or aniline-mediated condensations introduce additional complexity, requiring expensive reagents, longer reaction times exceeding 24 hours, or toxic halogenated solvents, thereby complicating the supply chain and environmental compliance protocols.
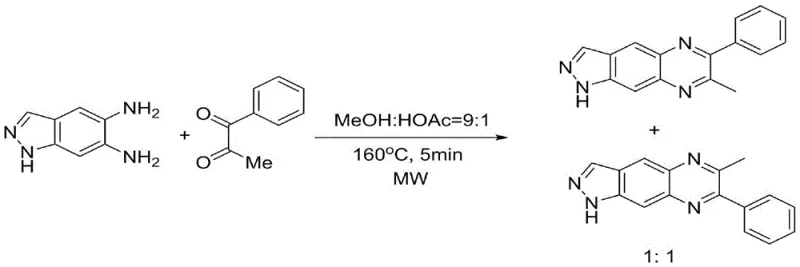
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in CN111018795B employs a streamlined base-catalyzed protocol that operates under remarkably mild conditions. By reacting 5-substituted o-phenylenediamines with alpha-ketoesters in the presence of organic bases such as triethylamine or DIPEA, the reaction pathway is kinetically controlled to favor the formation of the quinoxaline-3-one scaffold. This approach eliminates the need for harsh acids, transition metals, or microwave irradiation, allowing the reaction to proceed efficiently at temperatures between 20°C and 70°C. The simplicity of the reagent system—often just a common solvent like methanol and a cheap amine base—translates directly into operational ease and reduced raw material costs, making it an ideal candidate for reliable API intermediate supplier networks aiming to optimize their manufacturing portfolios.
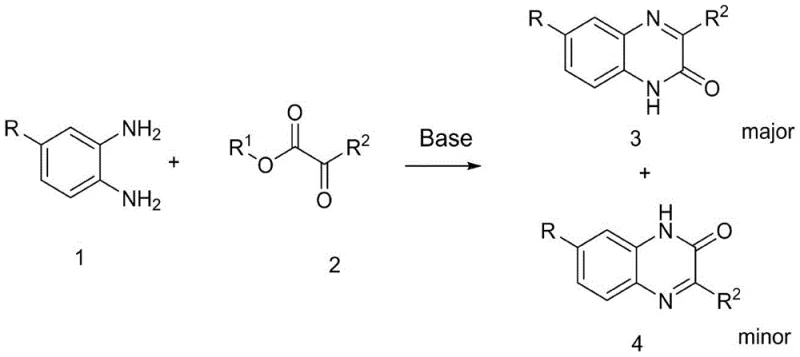
Mechanistic Insights into Base-Catalyzed Regioselective Cyclization
The core innovation of this technology lies in its ability to invert the typical regioselectivity observed in quinoxaline formation. In conventional acid-catalyzed or thermal condensations, the nucleophilic attack often favors the formation of the 2-keto isomer (Structure I) due to electronic factors. However, the alkaline environment provided by the organic base alters the nucleophilicity of the amine groups and the electrophilicity of the carbonyl centers in the alpha-ketoester. This subtle shift in reaction dynamics promotes the initial attack at the ester carbonyl or modifies the cyclization transition state to favor the 3-keto product (Structure II). Understanding this mechanistic nuance is critical for process chemists, as it allows for the rational design of substrates where the 3-position functionalization is required for specific biological activity, avoiding the costly separation of inseparable isomers that plague other synthetic routes.
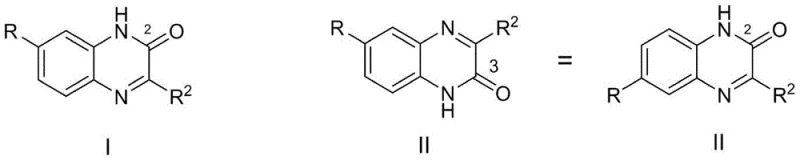
Furthermore, the choice of base and solvent plays a pivotal role in maintaining this high selectivity. The patent data indicates that inorganic bases like sodium hydroxide can lead to hydrolysis of the sensitive keto-ester starting materials, resulting in disordered reaction systems and lower yields. Conversely, organic bases provide a buffered environment that facilitates the condensation without degrading the substrate. The steric bulk of bases like DIPEA may also contribute to shielding specific reactive sites, further enhancing the ratio of the target 3-one product. This level of control over the impurity profile is essential for meeting the stringent purity specifications required in pharmaceutical manufacturing, ensuring that the final API intermediate contains minimal levels of structurally related impurities that could complicate regulatory filings.
How to Synthesize Quinoxaline-3-one Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation, making it accessible for both laboratory optimization and industrial production. The general procedure involves dissolving the substituted o-phenylenediamine in a protic solvent such as methanol or ethanol, followed by the addition of the organic base. Once the mixture is homogenized, the alpha-ketoester is introduced slowly to manage the exotherm, and the reaction is allowed to stir at ambient or slightly elevated temperatures. This straightforward workflow minimizes the need for specialized equipment, allowing facilities to leverage existing infrastructure for the commercial scale-up of complex pharmaceutical intermediates. Detailed standardized operating procedures for this transformation are outlined below to assist technical teams in replicating these high-selectivity results.
- Dissolve 5-substituted o-phenylenediamine in a suitable solvent like methanol or ethanol under stirring.
- Add an organic base such as triethylamine or DIPEA with a molar ratio of 3-5 equivalents relative to the diamine.
- Slowly add the alpha-ketoester substrate and stir at 20-70°C for 1-40 hours, followed by standard aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this base-catalyzed methodology offers compelling advantages that directly impact the bottom line and supply chain resilience. By shifting away from precious metal catalysts or hazardous halogenated reagents, manufacturers can significantly reduce the cost of goods sold (COGS) associated with raw material procurement and waste disposal. The elimination of heavy metals removes the necessity for expensive scavenging steps and rigorous residual metal testing, streamlining the quality control workflow. Moreover, the use of commodity chemicals like triethylamine and ethanol ensures a stable supply chain, mitigating the risks associated with sourcing specialized or regulated reagents that often face market volatility. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the drastic simplification of the reaction setup and workup. Since the reaction proceeds with high conversion and selectivity under mild conditions, energy consumption for heating and cooling is minimized compared to high-temperature microwave or reflux methods. Additionally, the avoidance of chromatographic purification in favor of simpler crystallization or extraction techniques reduces solvent usage and labor hours. These cumulative efficiencies result in substantial cost savings in API intermediate manufacturing, allowing for more competitive pricing structures without compromising on product quality or margin.
- Enhanced Supply Chain Reliability: The reliance on widely available, non-hazardous starting materials enhances the robustness of the supply chain. Unlike methods requiring custom-synthesized dicarbonyls or unstable iodine reagents, the alpha-ketoesters and diamines used here are commercially accessible from multiple vendors. This diversification of the supplier base reduces the risk of single-source bottlenecks and ensures business continuity. For procurement managers, this means reduced lead time for high-purity intermediates and greater flexibility in negotiating contracts, as the manufacturing process is not held hostage by the availability of exotic catalysts or reagents.
- Scalability and Environmental Compliance: The green chemistry attributes of this method align perfectly with modern environmental, social, and governance (ESG) goals. The absence of heavy metals and halogenated solvents simplifies wastewater treatment and reduces the generation of hazardous solid waste. This makes the process inherently safer and easier to scale from kilogram to multi-ton batches without triggering complex environmental permitting hurdles. For supply chain heads, this translates to faster technology transfer times and a lower regulatory burden, facilitating the rapid deployment of new intermediates into the commercial supply network.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled answers to common inquiries based on the patent data and industry standards. These questions address the specific advantages regarding selectivity, safety, and scalability that are most relevant to decision-makers evaluating new synthetic routes. Understanding these details helps in assessing the feasibility of integrating this technology into existing production lines for quinoxaline-based active ingredients.
Q: Why is regioselectivity critical in quinoxaline synthesis?
A: Regioselectivity determines whether the carbonyl group ends up at position 2 or 3 of the quinoxaline ring. Traditional methods often yield a 1:1 mixture of isomers, making purification difficult and increasing waste. High selectivity for the 3-one isomer simplifies downstream processing and ensures consistent biological activity profiles for API intermediates.
Q: What are the advantages of using organic bases over traditional acid catalysis?
A: Organic bases like triethylamine allow the reaction to proceed under mild temperatures (20-70°C) without the need for harsh acidic conditions or expensive transition metal catalysts. This reduces equipment corrosion risks, eliminates heavy metal contamination concerns, and significantly lowers the cost of raw materials and waste treatment.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly scalable. It utilizes readily available raw materials, operates at ambient to moderate pressures, and does not require specialized equipment like microwave reactors. The simple workup procedure involving extraction and crystallization facilitates easy adaptation to multi-ton manufacturing scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxaline-3-one Supplier
The technological advancements described in CN111018795B underscore the potential for producing high-value heterocyclic intermediates with unprecedented efficiency. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of quinoxaline-3-one derivative meets the exacting standards required for pharmaceutical applications. We understand the critical nature of supply continuity and are equipped to handle the complexities of regioselective synthesis to deliver consistent, high-quality products.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and reduce your overall manufacturing costs.
