Advanced Manufacturing of Amisulpride: A Technical Breakthrough for Global Supply Chains
Introduction to Novel Amisulpride Manufacturing Technologies
The global demand for high-quality antipsychotic medications continues to drive innovation in pharmaceutical intermediate synthesis. A significant advancement in this field is detailed in patent CN110577482B, which outlines a highly efficient preparation method for Amisulpride, a critical active pharmaceutical ingredient (API) used primarily in the treatment of schizophrenia. This patent introduces a streamlined synthetic route that addresses long-standing challenges in yield optimization and process safety. By utilizing 2-methoxy-4-amino-5-ethylsulfonyl benzoic acid and thionyl chloride as primary raw materials, the method achieves a direct acylation followed by a seamless amidation reaction. This approach not only simplifies the operational workflow but also ensures the production of the target molecule with exceptional purity levels, often exceeding industry standards. For R&D directors and procurement specialists, understanding this technological shift is vital for securing a reliable amisulpride supplier capable of meeting rigorous quality specifications.
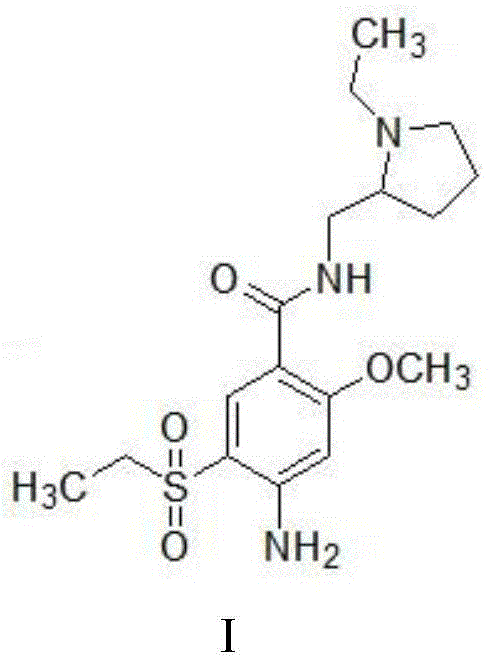
The structural integrity of Amisulpride, as depicted in the chemical formula, relies on the precise arrangement of the ethylsulfonyl group and the pyrrolidine moiety. The novelty of the patented process lies in its ability to construct this complex architecture through a robust two-step sequence that avoids the pitfalls of traditional multi-step syntheses. By focusing on reaction selectivity and minimizing side products, this method represents a paradigm shift towards greener and more cost-effective pharmaceutical intermediates manufacturing. The implications for supply chain stability are profound, as simplified processes generally translate to reduced lead times and lower production costs, ultimately benefiting the entire value chain from raw material sourcing to final drug formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN110577482B, the synthesis of Amisulpride was plagued by several inefficiencies that hindered large-scale production. For instance, earlier patents such as CN102807516A disclosed a method involving the use of di-tert-butyl dicarbonate to introduce a Boc protecting group. This necessitated a three-step sequence including protection, condensation, and subsequent deprotection. Such a lengthy pathway inherently increases the risk of yield loss at each stage, with reported maximum yields hovering around only 76 percent. Furthermore, the use of protecting groups adds significant material costs and generates additional waste streams, complicating the environmental compliance profile of the manufacturing process. Other methods, like those found in CN102838520A, attempted one-step synthesis under alkaline conditions using phenyl chloroformate or oxalyl chloride. However, these approaches suffered from poor selectivity, leading to substantial side reactions between the activating agents and the amine component, thereby compromising the purity of the final product.
The Novel Approach
In stark contrast, the novel approach presented in the subject patent revolutionizes the synthesis by adopting a direct acylation-amidation strategy. This method bypasses the need for cumbersome protecting groups and avoids the harsh conditions associated with alkaline amidation. Instead, it leverages the high reactivity of the in-situ generated acyl chloride intermediate. By reacting 2-methoxy-4-amino-5-ethylsulfonyl benzoic acid with thionyl chloride, the process efficiently generates the reactive acid chloride species. This intermediate is then immediately subjected to amidation with N-ethyl-2-aminomethyl pyrrolidine. The result is a dramatic improvement in process efficiency, characterized by a shorter preparation period and significantly higher yields. The elimination of isolation steps between the acylation and amidation phases creates a "one-pot" advantage that drastically reduces solvent usage and labor intensity, positioning this technology as a superior choice for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Acylation and Amidation Cascade
The core of this technological breakthrough lies in the precise control of the reaction mechanism, specifically the conversion of the carboxylic acid to the acid chloride and its subsequent coupling with the amine. The first stage involves the activation of the benzoic acid derivative using thionyl chloride (SOCl2) in the presence of a catalytic amount of N,N-dimethylformamide (DMF). The DMF acts as a nucleophilic catalyst, forming a highly reactive Vilsmeier-Haack type intermediate that accelerates the chlorination of the carboxyl group. This step is critical; the patent specifies a temperature range of 30-45°C, preferably 35-40°C, to ensure complete conversion while preventing thermal degradation. The stoichiometry is carefully balanced with a slight excess of thionyl chloride (molar ratio 1:1.0 to 1.1) to drive the equilibrium forward without leaving excessive residual reagents that could complicate downstream processing.
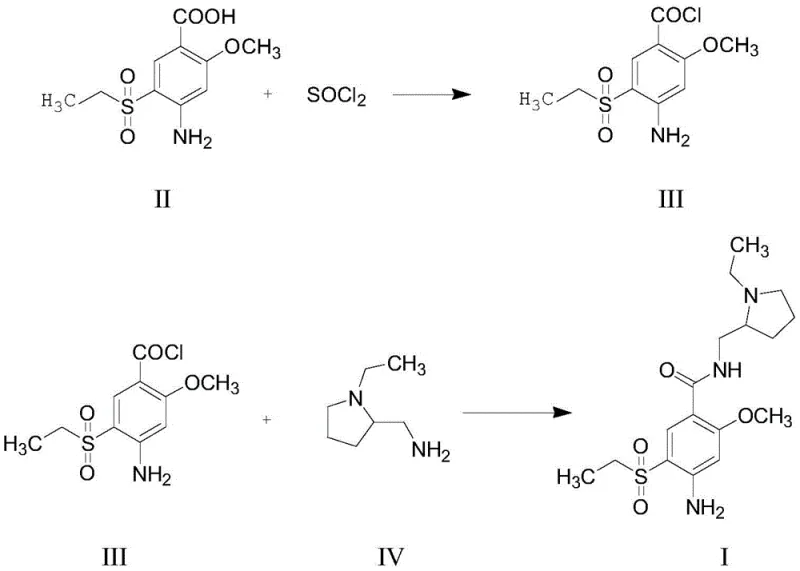
Following the formation of the acid chloride (Compound III), the process transitions seamlessly into the amidation phase without isolating the intermediate. This "telescoped" operation is key to maintaining high purity. The reaction mixture is cooled to 20-25°C before the addition of N-ethyl-2-aminomethyl pyrrolidine (Compound IV). Crucially, Compound IV serves a dual role: it acts as the nucleophile to form the amide bond and simultaneously functions as an acid scavenger to neutralize the HCl generated during the reaction. The patent recommends using a molar excess of Compound IV (2.0-3.0 equivalents relative to the acid) to ensure complete consumption of the acid chloride and to buffer the reaction medium. The temperature is then raised back to 35-40°C to facilitate the amidation kinetics. This controlled thermal profile minimizes side reactions, such as the hydrolysis of the acid chloride or over-alkylation, ensuring that the final high-purity amisulpride is obtained with a purity of over 98 percent after simple recrystallization.
How to Synthesize Amisulpride Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters regarding solvent selection, reagent addition rates, and temperature control to maximize yield and safety. The process utilizes common organic solvents like dichloromethane, which allows for easy handling and recovery. The following guide outlines the standardized operational procedure derived from the patent examples, designed to assist process chemists in replicating these high-efficiency results in a pilot or production setting. Detailed standard operating procedures for scale-up should always be validated against local safety regulations and equipment capabilities.
- Perform acylation of 2-methoxy-4-amino-5-ethylsulfonyl benzoic acid with thionyl chloride in dichloromethane using catalytic DMF at 35-40°C.
- Without isolating the intermediate, cool the reaction mixture and add N-ethyl-2-aminomethyl pyrrolidine to initiate amidation.
- Work up the reaction by washing with saturated sodium bicarbonate, followed by solvent removal and recrystallization from acetone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the significant simplification of the supply chain for raw materials. The starting materials, specifically 2-methoxy-4-amino-5-ethylsulfonyl benzoic acid and N-ethyl-2-aminomethyl pyrrolidine, are readily available commodity chemicals, reducing the risk of supply bottlenecks. Furthermore, the elimination of specialized reagents like di-tert-butyl dicarbonate or phenyl chloroformate removes dependency on niche suppliers, thereby enhancing overall supply chain resilience. The streamlined nature of the process also implies a drastic reduction in manufacturing cycle time. By combining two reaction steps into a single vessel operation, facilities can increase throughput capacity without requiring additional capital investment in reactor hardware, directly contributing to commercial scale-up of complex psychiatric drug intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the reduction in unit operations. Traditional methods requiring isolation and purification of intermediates consume vast amounts of solvents and energy for drying and distillation. By employing a one-pot technique, the consumption of utilities and solvents is substantially lowered. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which reduces the burden on downstream purification units. This efficiency translates into a lower cost of goods sold (COGS), allowing for more competitive pricing strategies in the global market without compromising margin.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to consistent batch-to-batch quality. The tolerance for mild temperatures (30-45°C) and the use of stable solvents like dichloromethane reduce the likelihood of batch failures due to thermal runaway or equipment corrosion. This reliability is crucial for maintaining continuous supply to API manufacturers. Moreover, the high yield (reported up to 89.4% in examples) ensures that less raw material is wasted per kilogram of product, optimizing inventory turnover and reducing the working capital tied up in raw material stockpiles.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method is superior. The patent explicitly notes the absence of highly toxic substances, which simplifies waste treatment protocols and lowers disposal costs. The ability to recycle solvents like dichloromethane further aligns with green chemistry principles. Scalability is enhanced by the dropwise addition protocols described, which manage exotherms effectively, making the transition from laboratory grams to multi-ton production safer and more predictable. This facilitates reducing lead time for high-purity amisulpride delivery to customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, purity profiles, and operational safety. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of the one-pot synthesis method for Amisulpride?
A: The one-pot method eliminates the need for isolating the unstable acyl chloride intermediate, significantly reducing processing time and solvent consumption while maintaining high purity (>98%).
Q: How does this method compare to prior art regarding yield and safety?
A: Unlike previous methods requiring toxic reagents or complex protection/deprotection steps with yields around 76%, this novel route achieves molar yields exceeding 88% without using highly toxic substances.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes common solvents like dichloromethane and operates at mild temperatures (30-45°C), making it highly suitable for large-scale commercial manufacturing with minimal side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amisulpride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the pharmaceutical sector. Our team of expert chemists has thoroughly analyzed the technological potential of the route described in CN110577482B and is fully prepared to leverage these insights for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Amisulpride meets the highest international standards for safety and efficacy.
We invite global partners to collaborate with us to unlock the full commercial potential of this efficient synthesis route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and operational reliability.
