Advanced Amisulpride Manufacturing: Scalable Two-Step Synthesis for Global Supply
The pharmaceutical industry constantly seeks robust manufacturing pathways that balance high purity with economic efficiency, and the synthesis of Amisulpride represents a critical area of innovation for antipsychotic medication production. Patent CN103819383A introduces a transformative approach that streamlines the creation of this vital compound through a concise two-step sequence, moving away from the cumbersome multi-stage processes that have historically plagued this sector. By leveraging 2-methoxy-4-amino-5-ethylsulfonyl benzoic acid as a foundational starting material, this method achieves exceptional atom utilization and yield rates while maintaining mild reaction conditions that are conducive to large-scale industrial operations. The strategic elimination of toxic catalysts and the adoption of recyclable solvents mark a significant leap forward in green chemistry principles, offering a sustainable solution for the reliable amisulpride supplier market. This technical breakthrough not only enhances the feasibility of commercial production but also addresses the growing demand for high-purity pharmaceutical intermediates that meet stringent global regulatory standards without compromising on cost-effectiveness or supply continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of Amisulpride has been hindered by complex synthetic routes that involve multiple reaction stages, each introducing potential points of failure and yield loss that accumulate to reduce overall efficiency. Traditional methods often rely on harsh sulfonation processes using chlorsulfonic acid or oxidation steps that require expensive and toxic reagents, creating significant challenges for waste management and environmental compliance in modern facilities. These legacy pathways frequently necessitate the use of unstable intermediates, such as disulfides or mercaptans, which demand rigorous safety protocols and specialized equipment to handle, thereby driving up capital expenditure and operational costs for manufacturers. Furthermore, the purification processes associated with these older techniques are often labor-intensive and time-consuming, involving multiple extraction and crystallization steps that extend lead times and reduce the throughput capacity of production lines. The reliance on non-recyclable solvents and harmful catalysts in these conventional methods also poses a substantial liability in terms of environmental impact, making it increasingly difficult for producers to align with the tightening global regulations regarding chemical manufacturing and disposal.
The Novel Approach
In stark contrast to these legacy challenges, the novel approach detailed in the patent data utilizes a direct esterification and condensation strategy that drastically simplifies the molecular construction of the target compound while maximizing resource efficiency. By initiating the synthesis with a stable, industrially available benzoic acid derivative, the process bypasses the need for preliminary sulfonation or oxidation, effectively collapsing the synthetic timeline and reducing the exposure of personnel and equipment to hazardous conditions. The use of thionyl chloride as a catalyst in the initial esterification step ensures high conversion rates under mild thermal conditions, while the subsequent condensation with N-ethyl-2-aminomethylpyrrolidine proceeds smoothly in common organic solvents like isopropanol or acetonitrile. This streamlined methodology not only enhances the overall yield of the final product but also simplifies the post-reaction workup, allowing for straightforward filtration and drying procedures that minimize solvent consumption and energy usage. The result is a manufacturing protocol that is inherently safer, more cost-effective, and significantly more scalable, providing a robust foundation for the commercial scale-up of complex pharmaceutical intermediates in a competitive global market.
Mechanistic Insights into Thionyl Chloride-Catalyzed Esterification and Condensation
The core of this synthetic innovation lies in the precise activation of the carboxylic acid group through thionyl chloride catalysis, which facilitates a highly efficient nucleophilic attack by the alcohol molecule to form the ester intermediate. This mechanism proceeds through the formation of an acyl chloride species in situ, which is significantly more reactive towards nucleophiles than the parent acid, thereby driving the equilibrium towards the ester product with minimal energy input. The reaction conditions are carefully controlled, typically maintaining temperatures between 25°C and reflux, which allows for the complete conversion of the starting material while preventing the degradation of sensitive functional groups such as the amino and sulfonyl moieties present on the aromatic ring. The subsequent neutralization and dilution steps are critical for isolating the ester intermediate in high purity, as they effectively remove acidic byproducts and unreacted reagents without the need for complex chromatographic separation techniques. This mechanistic efficiency is the key to the process's high atom economy, ensuring that the majority of the input mass is retained in the final product structure rather than being lost as waste.
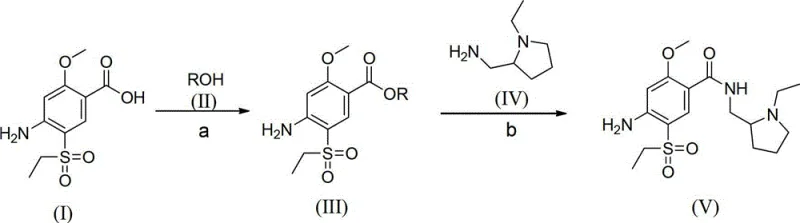
Following the formation of the ester intermediate, the condensation step involves a nucleophilic substitution where the amine group of N-ethyl-2-aminomethylpyrrolidine attacks the carbonyl carbon of the ester, displacing the alkoxide leaving group to form the final amide bond. This reaction is typically conducted at elevated temperatures ranging from 80°C to 125°C to overcome the activation energy barrier, ensuring that the reaction proceeds to completion within a reasonable timeframe of 20 to 60 hours. The choice of solvent plays a pivotal role in this stage, with polar aprotic solvents like acetonitrile or alcohols like isopropanol providing the necessary solvation environment to stabilize the transition state and facilitate the interaction between the reactants. The purification of the final Amisulpride product is achieved through a controlled cooling and crystallization process, where the addition of water induces precipitation of the product while keeping impurities in solution, followed by recrystallization to achieve the desired pharmaceutical grade purity. This detailed understanding of the reaction mechanism allows for precise optimization of parameters such as molar ratios and reaction times, ensuring consistent quality and reproducibility essential for cost reduction in pharmaceutical intermediates manufacturing.
How to Synthesize Amisulpride Efficiently
The practical implementation of this synthesis route requires careful attention to the stoichiometric ratios and thermal profiles defined in the patent to ensure optimal yield and purity outcomes. The process begins with the esterification of the benzoic acid derivative, where the mass ratio of acid to alcohol and thionyl chloride must be strictly maintained to prevent side reactions, followed by a controlled neutralization to isolate the intermediate ester. The subsequent condensation step demands precise temperature control and extended reaction times to ensure complete conversion, with the final isolation relying on differential solubility in water and organic solvents to separate the product from byproducts. Detailed standardized synthesis steps see the guide below for specific operational parameters and safety protocols required for industrial execution.
- Esterify 2-methoxy-4-amino-5-ethylsulfonyl benzoic acid with low alcohol using thionyl chloride catalyst.
- Condense the resulting ester intermediate with N-ethyl-2-aminomethylpyrrolidine under heated conditions.
- Purify the final product through water dilution, filtration, and recrystallization to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this streamlined synthesis route offers profound strategic benefits that extend far beyond simple chemical efficiency, directly impacting the bottom line and operational resilience of the organization. By reducing the number of synthetic steps from a complex multi-stage process to a concise two-step sequence, the method inherently lowers the consumption of raw materials, energy, and labor, resulting in substantial cost savings that can be passed down through the supply chain. The use of readily available starting materials eliminates the dependency on scarce or custom-synthesized precursors, thereby reducing lead time for high-purity pharmaceutical intermediates and mitigating the risk of supply disruptions caused by upstream bottlenecks. Furthermore, the mild reaction conditions and the absence of hazardous catalysts simplify the safety requirements for the manufacturing facility, lowering insurance costs and reducing the regulatory burden associated with handling toxic substances. These factors combine to create a supply profile that is not only more economical but also more reliable and sustainable, aligning perfectly with the long-term strategic goals of modern pharmaceutical enterprises seeking to optimize their sourcing strategies.
- Cost Reduction in Manufacturing: The elimination of multiple reaction steps and the removal of expensive, toxic catalysts from the process flow directly translate to a significant reduction in the overall cost of goods sold for the final active ingredient. By avoiding the need for complex purification techniques such as column chromatography and instead relying on simple filtration and crystallization, the process minimizes the consumption of high-purity solvents and reduces the waste disposal costs associated with hazardous chemical byproducts. The high atom utilization rate ensures that a greater proportion of the purchased raw materials is converted into saleable product, maximizing the return on investment for every kilogram of input material and driving down the unit cost of production. This economic efficiency is further enhanced by the ability to use common, recyclable solvents, which reduces the recurring expenditure on chemical consumables and supports a more sustainable manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on 2-methoxy-4-amino-5-ethylsulfonyl benzoic acid as a starting material leverages an existing industrial supply base, ensuring that raw material availability is stable and not subject to the volatility of niche chemical markets. The simplicity of the two-step process reduces the complexity of the manufacturing schedule, allowing for faster turnaround times and more flexible production planning that can adapt to fluctuating market demands without significant retooling or delays. The robustness of the reaction conditions means that the process is less susceptible to minor variations in temperature or pressure, leading to more consistent batch-to-batch quality and reducing the incidence of failed batches that can disrupt supply continuity. This reliability is crucial for maintaining the trust of downstream partners and ensuring that the pipeline of critical medications remains uninterrupted even in the face of global logistical challenges.
- Scalability and Environmental Compliance: The mild nature of the reaction conditions and the use of standard equipment make this synthesis route highly scalable, allowing for seamless transition from pilot plant quantities to multi-ton commercial production without the need for specialized high-pressure or cryogenic infrastructure. The avoidance of environmentally harmful catalysts and the generation of less hazardous waste streams simplify the compliance landscape, making it easier for manufacturers to meet strict environmental regulations and obtain necessary operating permits. The ability to recycle solvents and the reduced energy footprint of the shorter process contribute to a lower carbon footprint, aligning the manufacturing process with corporate sustainability goals and enhancing the brand reputation of the supplier in an increasingly eco-conscious market. This combination of scalability and compliance ensures that the production capacity can grow in tandem with market demand while maintaining adherence to the highest standards of environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, providing clarity on its operational feasibility and strategic value for industry stakeholders. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent data, ensuring that the information provided is accurate and relevant to decision-makers evaluating this technology. Understanding these details is essential for assessing the potential impact of this route on your specific manufacturing capabilities and supply chain objectives.
Q: What are the primary advantages of this synthesis route over conventional methods?
A: This method significantly reduces the number of reaction steps from multiple stages to just two, eliminating the need for harmful catalysts and complex purification processes associated with older sulfonation or oxidation routes.
Q: Is the starting material readily available for large-scale production?
A: Yes, the process utilizes 2-methoxy-4-amino-5-ethylsulfonyl benzoic acid, which is an existing industrialized product, ensuring stable sourcing and reducing supply chain risks for commercial scale-up.
Q: How does this method impact environmental compliance and waste management?
A: The route avoids environmentally harmful catalysts and uses common recyclable solvents like isopropanol or acetonitrile, drastically simplifying waste treatment and aligning with strict environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amisulpride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis routes like the one described in CN103819383A to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists and process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity Amisulpride that meets stringent purity specifications through our rigorous QC labs, providing our partners with the confidence that their supply chain is built on a foundation of quality and reliability. Our state-of-the-art facilities are equipped to handle the specific requirements of this two-step synthesis, from the precise control of esterification conditions to the efficient isolation of the final product, guaranteeing consistent output that aligns with your production schedules.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior synthetic methodology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can improve your margins and operational efficiency. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence and our proven track record of excellence in fine chemical manufacturing. Let us be your partner in driving innovation and efficiency in your pharmaceutical production processes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
