Advanced Manufacturing of Trifluoroacetamidine: A Safe and Scalable Route for Pharma Intermediates
Advanced Manufacturing of Trifluoroacetamidine: A Safe and Scalable Route for Pharma Intermediates
The global demand for fluorinated building blocks in the pharmaceutical and agrochemical sectors continues to surge, driven by the unique metabolic stability and lipophilicity that fluorine atoms impart to bioactive molecules. Within this critical landscape, trifluoroacetamidine stands out as a versatile intermediate, serving as a pivotal precursor for the synthesis of nitrogen-containing heterocyclic compounds and various amidine derivatives. The technical breakthrough detailed in patent CN102786440A introduces a robust preparation method that fundamentally alters the risk profile and economic viability of producing this essential chemical. By shifting away from the direct handling of hazardous gaseous reagents towards a controlled, in-situ generation strategy, this technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing portfolios. The structural integrity and reactivity of the target molecule, as visualized below, underscore its importance in constructing complex molecular architectures used in modern drug discovery.
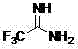
Furthermore, the versatility of trifluoroacetamidine extends beyond simple heterocycle formation; it serves as a core component in the development of advanced formamidine UV light absorbers, where specific substitutions on the nitrogen atoms yield materials with exceptional performance characteristics. Despite its widespread utility, the domestic industrial production of this compound has historically faced challenges, often necessitating reliance on external imports due to the complexities associated with traditional synthesis routes. The methodology outlined in the referenced patent addresses these bottlenecks by providing a streamlined, scalable process that enhances both safety and efficiency. For procurement managers and supply chain directors, understanding the nuances of this synthetic route is paramount, as it directly influences the continuity of supply and the overall cost structure of downstream API manufacturing. This report delves deep into the mechanistic advantages and commercial implications of adopting this novel preparation method.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amidines, including trifluoroacetamidine, has relied heavily on the direct reaction between nitriles and ammonia. Traditional protocols often involve dissolving the nitrile in absolute ethanol followed by the introduction of hydrogen chloride, or alternatively, conducting the synthesis under high-pressure conditions. While chemically feasible, these conventional approaches present severe logistical and safety hurdles when applied to trifluoroacetonitrile. The primary constraint lies in the physical nature of trifluoroacetonitrile itself; it is a highly toxic gaseous substance that poses significant risks during storage, transportation, and direct feeding into reaction vessels. Handling such volatile and hazardous raw materials requires specialized infrastructure, rigorous safety protocols, and often results in elevated operational costs due to the need for extensive containment systems. Moreover, the direct use of toxic gases complicates the reaction control, potentially leading to inconsistent yields and increased difficulty in managing exothermic events, thereby limiting the scalability of the process for large-scale commercial production.
The Novel Approach
In stark contrast to the perilous conventional methods, the innovative process described in patent CN102786440A circumvents the direct handling of trifluoroacetonitrile gas by generating it in situ. This novel approach utilizes trifluoroacetamide as a stable, solid starting material, which is subjected to a dehydration reaction in the presence of phosphorus pentoxide and polyphosphoric acid. As illustrated in the reaction scheme below, this transformation rapidly converts the amide into the reactive nitrile gas within a closed system, which is then immediately cooled and introduced into a liquid ammonia reactor. This strategic shift not only drastically reduces the production risk associated with toxic gas handling but also simplifies the operational workflow, making it far more accessible for industrial implementation. By integrating the generation and consumption of the reactive intermediate into a continuous or semi-continuous flow, the process minimizes exposure hazards and enhances the overall reaction yield, providing a safer and more efficient pathway for manufacturing high-purity trifluoroacetamidine.
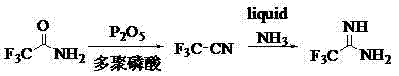
Mechanistic Insights into P2O5-Mediated Dehydration and Ammonolysis
The core of this technological advancement lies in the efficient dehydration mechanism facilitated by the synergistic action of phosphorus pentoxide (P2O5) and polyphosphoric acid (PPA). In the initial stage, trifluoroacetamide acts as the substrate, undergoing a dehydration reaction where the oxygen atom of the amide group is removed to form the carbon-nitrogen triple bond of the nitrile. Phosphorus pentoxide serves as a potent dehydrating agent, driving the equilibrium towards the formation of trifluoroacetonitrile gas, while polyphosphoric acid functions as both a solvent and a catalytic medium to ensure homogeneous reaction conditions. The reaction is typically initiated by preheating the polyphosphoric acid to approximately 50°C to enhance its flowability, followed by the addition of the solid reagents. As the temperature is slowly ramped up to the range of 146-150°C, the dehydration proceeds rapidly, releasing the trifluoroacetonitrile gas which is immediately swept out of the generation vessel. This precise thermal control is critical to preventing side reactions and ensuring the steady evolution of the gaseous intermediate.
Following the generation phase, the trifluoroacetonitrile gas is passed through a cooling system before being introduced into a reactor containing liquid ammonia maintained at cryogenic temperatures between -40 and -35°C. This low-temperature environment is essential for the subsequent ammonolysis reaction, where the nitrile reacts with ammonia to form the amidine functionality. The use of liquid ammonia not only provides the necessary nitrogen source but also acts as a solvent that facilitates the nucleophilic attack on the electrophilic carbon of the nitrile group. The reaction mixture is stirred for approximately three hours to ensure complete conversion, after which the excess ammonia is recycled, and the crude product is isolated via vacuum distillation. This meticulous control over reaction parameters allows for the suppression of impurities, resulting in a final product with a purity exceeding 95% and a yield greater than 85%, demonstrating the high selectivity of this catalytic system.
How to Synthesize Trifluoroacetamidine Efficiently
The practical implementation of this synthesis route requires careful attention to the stoichiometry and thermal profiles established in the patent embodiments. The process begins with the preparation of the reaction medium, where polyphosphoric acid is preheated to ensure optimal viscosity for mixing. Subsequently, trifluoroacetamide, phosphorus pentoxide, and the acid solvent are combined in a gas generation reactor, typically in a mass ratio that favors the complete conversion of the amide. The detailed standardized synthesis steps, including specific equipment setups and safety interlocks required for scaling this exothermic dehydration, are outlined in the technical guide below.
- Preheat polyphosphoric acid to 50°C to improve flowability, then mix with trifluoroacetamide and phosphorus pentoxide in a gas generation reactor.
- Slowly heat the mixture to 146-150°C to generate trifluoroacetonitrile gas in situ through dehydration.
- Pass the cooled gas into a liquid ammonia reactor maintained at -40 to -35°C, stir for 3 hours, and purify via vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the substantial reduction of operational risk, which directly correlates to lower insurance premiums and reduced liability exposure associated with handling highly toxic gaseous raw materials. By eliminating the need to procure, store, and transport bulk quantities of trifluoroacetonitrile gas, manufacturers can streamline their logistics and reduce the complexity of their hazard management protocols. This simplification of the supply chain enhances reliability, ensuring that production schedules are not disrupted by regulatory hurdles or safety incidents related to hazardous material transport. Furthermore, the ability to generate the reactive intermediate on-demand minimizes inventory holding costs and reduces the footprint required for safe storage, contributing to a more agile and responsive manufacturing operation.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive safety infrastructure required for direct gas handling and the utilization of cost-effective reagents like phosphorus pentoxide and polyphosphoric acid. Since the process avoids the use of transition metal catalysts or complex organometallic reagents, there is no need for costly downstream purification steps to remove heavy metal residues, which significantly lowers the overall processing costs. Additionally, the high reaction yield reported in the patent implies a more efficient utilization of raw materials, reducing the cost per kilogram of the final active intermediate. The qualitative improvement in process safety also leads to indirect cost savings by minimizing downtime associated with safety audits and maintenance of high-pressure gas systems.
- Enhanced Supply Chain Reliability: Sourcing stable solid raw materials like trifluoroacetamide is inherently more reliable than managing the supply of toxic gases, which are subject to stringent transportation regulations and potential supply disruptions. The robustness of this synthetic route ensures a consistent output of high-purity intermediates, allowing downstream API manufacturers to maintain steady production rates without the fear of raw material shortages. The modular nature of the gas generation and absorption setup allows for flexible scaling, meaning that suppliers can easily adjust production volumes to meet fluctuating market demands without significant capital investment in new infrastructure. This flexibility is crucial for maintaining continuity in the global supply of critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental perspective, this process offers a distinct advantage as the primary waste byproduct is phosphoric acid, which can be readily reclaimed and recycled by specialized producers, thereby minimizing the environmental footprint. Unlike processes that generate complex organic sludge or heavy metal waste, the waste stream here is well-defined and manageable, facilitating easier compliance with increasingly strict environmental regulations. The simplicity of the work-up procedure, involving vacuum distillation and ammonia recycling, further supports scalability, allowing the process to be transferred from pilot scale to multi-ton commercial production with minimal technical barriers. This alignment with green chemistry principles enhances the sustainability profile of the supply chain, a key metric for modern corporate responsibility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of trifluoroacetamidine, based on the specific details provided in the patent literature. These insights are designed to clarify the operational feasibility and safety profile of the method for potential partners and technical evaluators. Understanding these nuances is essential for making informed decisions regarding the integration of this intermediate into broader synthesis campaigns.
Q: Why is the in-situ generation of trifluoroacetonitrile preferred over direct usage?
A: Direct usage of trifluoroacetonitrile involves handling a highly toxic gaseous raw material, posing significant safety risks and operational difficulties. The patented in-situ method generates the gas safely within the reactor system.
Q: What are the purity and yield specifications of this method?
A: According to patent CN102786440A, this method achieves a product purity higher than 95% and a reaction yield greater than 85%, making it highly suitable for industrial applications.
Q: How does this process handle waste management?
A: The primary waste byproduct is phosphoric acid, which can be reclaimed and recycled by specialized producers, significantly reducing environmental impact compared to processes generating complex organic waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoroacetamidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates like trifluoroacetamidine play in the development of next-generation therapeutics and agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify every batch. Our capability to implement safe, in-situ generation technologies aligns perfectly with the industry's shift towards more sustainable and risk-mitigated manufacturing practices.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can enhance your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us support your journey from bench-scale discovery to full-scale commercialization with confidence and precision.
