Advanced Catalytic Dehydration Route for High-Purity Trifluoroacetamidine Manufacturing and Scale-Up
Advanced Catalytic Dehydration Route for High-Purity Trifluoroacetamidine Manufacturing and Scale-Up
The landscape of fluorinated pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic pathways. Patent CN112661667B introduces a groundbreaking methodology for the preparation of trifluoroacetamidine (CAS 354-37-0), a critical building block for bioactive heterocycles including pyrimidines, pyridines, and triazines. This innovation addresses the longstanding safety and environmental challenges associated with handling volatile trifluoroacetonitrile directly or utilizing corrosive dehydrating agents. By leveraging a reusable Lewis acid catalyst system, specifically tris(pentafluorophenyl)borane, the process achieves a seamless conversion from stable trifluoroacetamide to the target amidine with exceptional purity exceeding 99%. For R&D directors and procurement strategists, this represents a pivotal shift towards sustainable chemistry that does not compromise on yield or operational simplicity, offering a robust foundation for securing the supply chain of next-generation fluorinated drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoroacetamidine has been plagued by significant operational hazards and environmental burdens that complicate commercial viability. Traditional routes often rely on the direct use of trifluoroacetonitrile as a starting material; however, this reagent possesses high toxicity and a low melting point, making its storage, transport, and precise metering extremely difficult and dangerous in a plant setting. Alternative approaches utilizing phosphorus pentoxide (P2O5) for the dehydration of trifluoroacetamide present their own severe drawbacks, primarily the generation of massive quantities of acidic wastewater upon quenching, which imposes heavy costs on waste treatment facilities. Furthermore, P2O5 reactions are notoriously exothermic and difficult to control on a large scale, often leading to charring and reduced yields due to the formation of intractable phosphorus-containing byproducts that complicate downstream purification efforts.
The Novel Approach
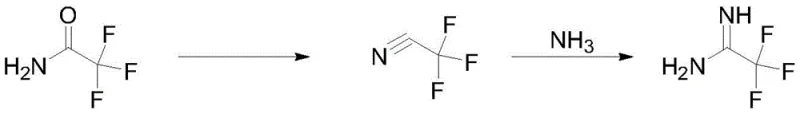
The novel methodology disclosed in the patent data circumvents these issues by employing a catalytic dehydration strategy that generates the reactive nitrile intermediate in situ, thereby eliminating the need to store or handle bulk quantities of hazardous gases. As illustrated in the reaction scheme below, trifluoroacetamide is subjected to reflux in the presence of a boron-based catalyst within a standard organic solvent matrix, allowing for the continuous evolution of dry trifluoroacetonitrile gas. This gas is immediately swept into a secondary reactor containing a DBU and liquid ammonia mixture, where it undergoes rapid ammonolysis to form the desired amidine. This telescoped approach not only enhances safety by minimizing the inventory of toxic intermediates but also streamlines the workflow into a coherent, operationally simple sequence that is highly amenable to automation and continuous processing technologies.
Mechanistic Insights into B(C6F5)3-Catalyzed Dehydration and Amidination
The core chemical innovation lies in the utilization of tris(pentafluorophenyl)borane, B(C6F5)3, as a potent yet recyclable Lewis acid catalyst to drive the dehydration equilibrium. Unlike stoichiometric dehydrating agents that are consumed and generate waste, this boron species activates the carbonyl oxygen of the trifluoroacetamide through coordination, significantly lowering the energy barrier for water elimination. The electron-deficient boron center withdraws electron density, facilitating the departure of the hydroxyl group as water, which is continuously removed via azeotropic distillation or a drying trap to push the reaction to completion. This mechanism ensures that the catalyst remains intact in the reaction loop, allowing it to be recovered and reused for subsequent batches, a feature that drastically alters the cost structure of the synthesis by amortizing the catalyst expense over multiple production cycles.
Following the generation of the nitrile, the process employs a dual-base system comprising 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) and liquid ammonia to effect the transformation to the amidine. DBU serves a dual purpose: it acts as a non-nucleophilic base to activate the ammonia and simultaneously functions as a high-boiling solvent that facilitates the subsequent distillation of the product. The reaction is conducted at low temperatures, typically between -10°C and 0°C, to control the exotherm and prevent side reactions. Crucially, the addition of polymerization inhibitors such as hydroquinone or 2,6-di-tert-butyl-4-methylphenol prior to the final distillation step is essential for impurity control. These additives scavenge free radicals that could otherwise initiate the polymerization of the electron-deficient amidine double bond during heating, thereby preserving the chemical integrity of the product and ensuring the final assay remains above the stringent 99% threshold required for pharmaceutical applications.
How to Synthesize Trifluoroacetamidine Efficiently
The implementation of this synthesis route requires careful attention to the physical setup, particularly the connection between the dehydration vessel and the ammonolysis reactor to ensure efficient gas transfer without leakage. The process begins with the suspension of the boron catalyst in a selected solvent such as toluene, n-heptane, or cyclohexane, followed by the addition of trifluoroacetamide. Upon heating to reflux, the system must be equipped with a water separator to continuously remove the byproduct water, driving the equilibrium forward. The evolved gas is passed through a drying train before bubbling into the chilled ammonia solution. Once the gas evolution ceases, indicating complete conversion of the amide, the reaction mixture is warmed, ammonia is vented, and the crude product is purified via fractional distillation under atmospheric pressure, collecting the fraction boiling between 56.2°C and 60.5°C. For detailed operational parameters and specific molar ratios, please refer to the standardized synthesis guide below.
- Dehydrate trifluoroacetamide using tris(pentafluorophenyl)borane catalyst in refluxing organic solvent to generate gaseous trifluoroacetonitrile.
- Pass the generated gas directly into a cooled solution of DBU and liquid ammonia to form the amidine intermediate.
- Add a polymerization inhibitor and perform atmospheric distillation at 56.2-60.5°C to isolate high-purity trifluoroacetamidine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this catalytic process offers profound advantages that directly impact the total cost of ownership and supply reliability for fluorinated intermediates. The shift from stoichiometric reagents to a catalytic cycle fundamentally changes the material cost profile, as the expensive boron catalyst is not consumed but rather retained within the process loop for repeated use. This eliminates the recurring purchase cost of dehydrating agents and, more importantly, removes the substantial logistical and financial burden associated with the disposal of phosphorus-rich hazardous waste. For procurement managers, this translates into a more predictable and stable cost structure that is less susceptible to fluctuations in the prices of bulk commodity chemicals used in waste neutralization.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the drastic simplification of the downstream processing and waste management phases. By avoiding the use of phosphorus pentoxide, the facility avoids the generation of tons of acidic sludge, which typically requires expensive neutralization, filtration, and specialized disposal services. Additionally, the ability to reuse the catalyst for more than 10 cycles significantly lowers the effective cost per kilogram of the active catalyst, contributing to substantial overall cost savings in the manufacturing of high-purity pharmaceutical intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of trifluoroacetamide as the starting material, which is a stable, solid commodity chemical with a well-established global supply chain, unlike the volatile and hazardous trifluoroacetonitrile. This stability reduces the risk of supply disruptions caused by transportation restrictions on dangerous goods. Furthermore, the process utilizes common, non-proprietary solvents like toluene and n-heptane, ensuring that raw material availability is not a bottleneck. The robustness of the reaction conditions also means that production schedules are less likely to be delayed by complex troubleshooting or equipment failures associated with handling corrosive slurries.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly smaller, aligning with increasingly stringent global regulations on industrial emissions and waste discharge. The reduction in three wastes (waste water, waste gas, and solid waste) simplifies the permitting process for new production lines and reduces the risk of regulatory shutdowns. From a scalability standpoint, the reaction operates at atmospheric pressure and uses standard glass-lined or stainless steel reactors, making the transition from pilot scale to multi-ton commercial production straightforward. The inherent safety of generating the toxic nitrile in situ and consuming it immediately further reduces the insurance and safety compliance costs associated with storing large volumes of hazardous intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this boron-catalyzed dehydration technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners and technical evaluators.
Q: Why is the B(C6F5)3 catalytic system superior to traditional P2O5 dehydration?
A: The boron-based catalyst system eliminates the massive wastewater generation associated with phosphorus pentoxide hydrolysis. Furthermore, the catalyst can be reused more than 10 times, significantly lowering raw material costs and simplifying the post-reaction workup compared to the slurry handling required for P2O5.
Q: How does this process ensure product stability during distillation?
A: The protocol explicitly incorporates polymerization inhibitors such as 2,6-di-tert-butyl-4-methylphenol or hydroquinone prior to distillation. This prevents the thermal polymerization of the reactive amidine species, ensuring the final product maintains a purity greater than 99% GC area.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes common industrial solvents like toluene or n-heptane and operates at atmospheric pressure with standard reflux conditions. The continuous gas-generation and capture design minimizes the inventory of toxic trifluoroacetonitrile, enhancing safety profiles for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoroacetamidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic methodologies like the B(C6F5)3-catalyzed route is essential for maintaining competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of trifluoroacetamidine meets the >99% purity benchmark required for sensitive drug synthesis applications.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this catalytic route for your supply chain. Please contact us to request specific COA data from our recent pilot runs and to discuss route feasibility assessments tailored to your volume requirements and timeline constraints.
