Scalable Synthesis of Obeticholic Acid Intermediates via Novel Oxidation and Olefination Strategies
The pharmaceutical landscape for treating cholestatic liver diseases has been significantly transformed by the advent of Obeticholic Acid (OCA), a potent farnesoid X receptor (FXR) agonist. As demand for this critical Active Pharmaceutical Ingredient (API) surges, the efficiency and safety of its supply chain have become paramount. Patent CN108264532B discloses a groundbreaking preparation method for Obeticholic Acid and its key intermediates, offering a robust alternative to legacy synthetic routes. This technical insight report analyzes the novel methodology, which leverages selective oxidation and Wittig olefination to bypass the severe limitations of traditional cryogenic alkylation. For R&D directors and procurement specialists seeking a reliable obeticholic acid intermediate supplier, understanding these mechanistic shifts is essential for securing a stable, cost-effective supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Obeticholic Acid has relied heavily on routes originating from Chenodeoxycholic Acid (CDCA) or 7-Ketolithocholic Acid (7-KLCA). Early methodologies, such as those described in WO02072598, necessitated the use of highly hazardous reagents like n-butyllithium (n-BuLi) or lithium diisopropylamide (LDA) to effect 6-alpha-ethylation. These reactions demand extreme cryogenic conditions, often reaching temperatures as low as -78°C, which imposes a massive burden on energy consumption and reactor infrastructure. Furthermore, the reliance on volatile alkyl halides like bromoethane or iodoethane introduces significant genotoxicity risks, requiring stringent and costly purification protocols to meet ICH M7 guidelines. The cumulative effect of these harsh conditions is a process with low overall yield, high operational risk, and limited scalability, making it ill-suited for the growing commercial demands of the global market.
The Novel Approach
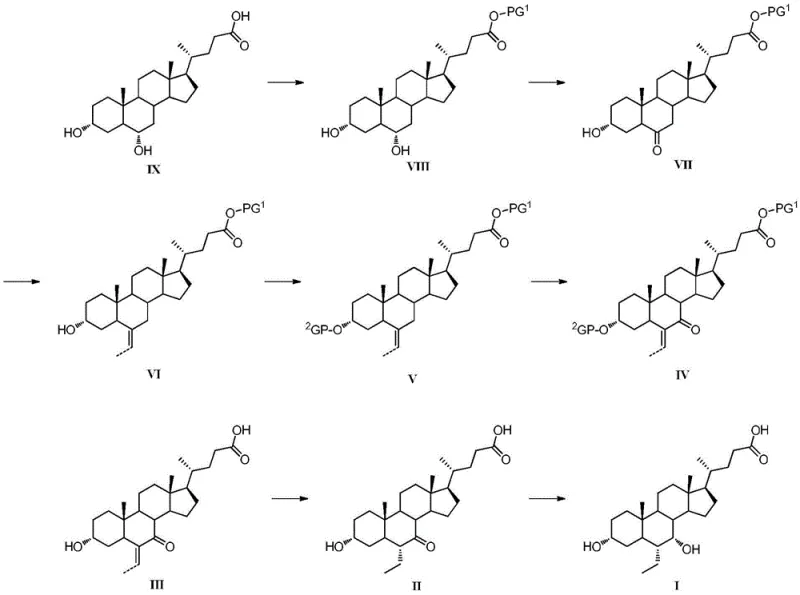
The methodology outlined in CN108264532B represents a paradigm shift by utilizing Hyodeoxycholic Acid (HDCA) as a starting material and employing a strategic sequence of oxidation and olefination. Instead of direct alkylation, the process introduces the ethyl side chain via a Wittig reaction to form a 6-ethylidene intermediate, which is subsequently hydrogenated. This approach completely eliminates the need for cryogenic alkylation and genotoxic alkyl halides. The oxidation of the 7-hydroxyl group is achieved using N-bromosuccinimide (NBS) or chromium-based oxidants under mild conditions, avoiding the instability associated with enol ether intermediates used in older routes. This streamlined pathway not only enhances safety but also improves the stereochemical control of the final product, ensuring high purity without the need for extensive chromatographic separation.
Mechanistic Insights into Selective Oxidation and Wittig Olefination
The core innovation of this synthesis lies in the precise manipulation of the steroid backbone, specifically at the C-6 and C-7 positions. The conversion of the 7-alpha-hydroxyl group to a 7-keto functionality is critical for enabling the subsequent olefination. In the preferred embodiments, N-bromosuccinimide (NBS) is utilized in a mixed solvent system of acetone and water. This reagent facilitates a selective oxidation that preserves the integrity of the 3-alpha-hydroxyl group (often protected as an ester or ether during earlier steps) while efficiently generating the ketone required for the Wittig reaction. The mechanistic advantage here is the avoidance of over-oxidation or degradation of the sensitive bile acid skeleton, which is a common pitfall in less selective oxidation protocols.
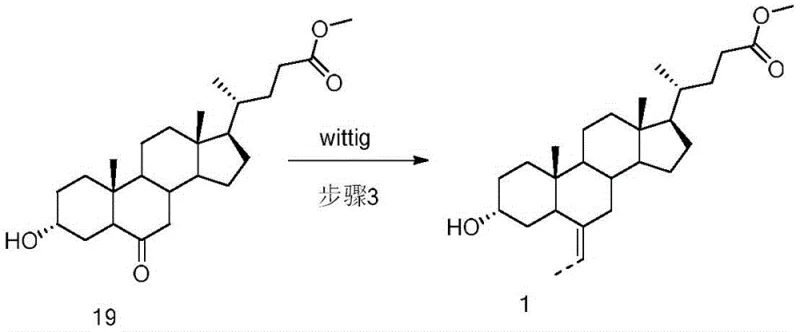
Following the formation of the 7-keto intermediate, the introduction of the carbon framework at the C-6 position is executed via a Wittig reaction. By reacting the 7-keto steroid with an ethyl phosphonium ylide, generated in situ from ethyltriphenylphosphonium bromide and a base like potassium tert-butoxide, a 6-ethylidene double bond is formed. This olefination step is thermodynamically favorable and proceeds under much milder thermal conditions compared to enolate alkylation. The resulting exocyclic double bond serves as a handle for the final stereochemical setup. Subsequent catalytic hydrogenation reduces the double bond to a single ethyl group, preferentially yielding the desired 6-alpha-configuration due to the steric environment of the steroid nucleus. This two-step "olefination-hydrogenation" sequence effectively replaces the risky direct alkylation, providing a cleaner reaction profile and superior impurity control.
How to Synthesize Obeticholic Acid Intermediates Efficiently
The implementation of this novel synthetic route requires careful attention to reaction parameters, particularly during the oxidation and olefination stages. The process begins with the protection of the carboxylic acid and the 3-hydroxyl group of Hyodeoxycholic Acid, followed by the critical NBS oxidation to install the 7-keto group. Once the ketone is established, the Wittig reaction is performed using a phosphorus ylide to generate the 6-ethylidene intermediate. The final stages involve the deprotection of functional groups, catalytic hydrogenation to set the 6-alpha-ethyl stereochemistry, and selective reduction of the 7-keto group back to the 7-alpha-hydroxyl moiety. Detailed standardized operating procedures for each of these transformations are critical for maintaining batch-to-batch consistency and ensuring the final API meets stringent pharmacopeial standards.
- Esterification of Hyodeoxycholic Acid followed by selective oxidation of the 7-alpha-hydroxyl group to a 7-keto group using N-bromosuccinimide (NBS).
- Introduction of the 6-ethylidene moiety via Wittig reaction using ethyltriphenylphosphonium bromide and a strong base.
- Final stereoselective hydrogenation of the 6-ethylidene double bond and reduction of the 7-keto group to yield the target 6-alpha-ethyl-7-alpha-hydroxy configuration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers substantial strategic benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the manufacturing infrastructure. By eliminating the requirement for deep-freeze reactors capable of sustaining -78°C, manufacturers can utilize standard glass-lined or stainless steel reactors, significantly lowering capital expenditure (CAPEX) and operational expenditure (OPEX). Furthermore, the removal of pyrophoric reagents like n-BuLi reduces the need for specialized handling protocols and inert atmosphere systems, thereby enhancing plant safety and reducing insurance and compliance costs. This process optimization translates directly into a more resilient supply chain capable of meeting high-volume demands without the bottlenecks associated with complex cryogenic operations.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous alkylating agents with stable phosphonium salts and solid oxidants like NBS leads to significant raw material cost savings. Additionally, the milder reaction conditions reduce energy consumption associated with cooling and heating cycles. The improved selectivity of the Wittig-hydrogenation sequence minimizes the formation of difficult-to-remove isomers, reducing the reliance on expensive preparative chromatography and increasing overall process yield. These factors combine to lower the cost of goods sold (COGS) for the final intermediate, offering better pricing stability for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Sourcing hazardous reagents like n-butyllithium often involves complex logistics and regulatory hurdles due to their classification as dangerous goods. In contrast, the reagents used in this novel route, such as NBS and ethyltriphenylphosphonium bromide, are commercially available, stable solids with straightforward shipping requirements. This simplifies the procurement process and reduces the risk of supply disruptions caused by transportation restrictions or vendor availability issues. A more robust raw material base ensures continuous production capability, safeguarding against market volatility.
- Scalability and Environmental Compliance: The environmental footprint of pharmaceutical manufacturing is increasingly scrutinized. This route avoids the generation of toxic borane gases associated with boron trifluoride-mediated reactions in older methods. The use of catalytic hydrogenation and standard workup procedures generates less hazardous waste, facilitating easier compliance with environmental regulations. The process is inherently more scalable, allowing for seamless transition from pilot plant to commercial tonnage production without the engineering challenges posed by cryogenic scaling, thus ensuring long-term supply security for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details helps stakeholders evaluate the feasibility of adopting this route for their specific supply chain needs. The answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the safety advantages of this new Obeticholic Acid synthesis route?
A: This route eliminates the need for cryogenic conditions (-78°C) and pyrophoric reagents like n-butyllithium or LDA, significantly reducing operational risks and equipment costs compared to traditional alkylation methods.
Q: How does this process address genotoxic impurity concerns?
A: By utilizing a Wittig olefination strategy followed by hydrogenation instead of direct alkylation with ethyl halides (bromoethane/iodoethane), the process avoids the introduction of genotoxic alkylating agents, simplifying impurity control.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the use of solid oxidants like NBS and standard hydrogenation catalysts allows for robust scale-up without the specialized cryogenic reactors required by earlier generations of synthesis technology.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Obeticholic Acid Intermediate Supplier
The technological advancements detailed in CN108264532B underscore the complexity and sophistication required to produce high-quality Obeticholic Acid intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel synthesis—such as improved safety and cost efficiency—are fully realized at an industrial level. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for FDA-approved drug substances.
We invite global pharmaceutical partners to collaborate with us to optimize their Obeticholic Acid supply chains. By leveraging our advanced synthesis capabilities, you can mitigate the risks associated with legacy manufacturing methods. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your project's economic and operational performance.
