Advanced Phosphoramide Derivatives: Revolutionizing Catalytic Rearrangement for Industrial Ketone Production
The landscape of fine chemical manufacturing, particularly within the flavors and fragrances sector, is constantly evolving towards more efficient and sustainable catalytic processes. A significant breakthrough in this domain is documented in patent CN111004276A, which introduces a novel class of phosphoramide derivatives designed to act as potent organocatalysts. These compounds address longstanding inefficiencies in the synthesis of gamma-delta-unsaturated ketones, which are critical intermediates for high-value products like Vitamin E and premium fragrances. The core innovation lies in the unique molecular architecture of these derivatives, characterized by a central phosphorus atom bonded to phenoxy groups and a sulfonylamino moiety. 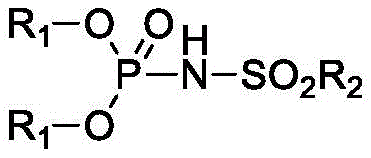 This structural configuration imparts exceptional acidity and solubility properties, distinguishing it from conventional inorganic acids. For procurement specialists and R&D directors seeking a reliable fine chemical intermediate supplier, understanding the mechanistic advantages of this technology is crucial for optimizing supply chains and reducing production costs in complex organic synthesis.
This structural configuration imparts exceptional acidity and solubility properties, distinguishing it from conventional inorganic acids. For procurement specialists and R&D directors seeking a reliable fine chemical intermediate supplier, understanding the mechanistic advantages of this technology is crucial for optimizing supply chains and reducing production costs in complex organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of gamma-delta-unsaturated ketones, such as methyl heptenone, has relied on acid-catalyzed rearrangement reactions that suffer from significant operational drawbacks. Early methodologies, dating back to the 1960s, utilized phosphoric acid but required excessively high temperatures ranging from 120 to 200 degrees Celsius and prolonged reaction times extending up to 16 hours, often resulting in mediocre yields around 41 percent. Subsequent improvements involving ammonium salts or specialized ionic liquids managed to improve yields but frequently introduced new complications, such as the need for extended reaction periods lasting between 12 to 40 hours or the requirement for high-pressure equipment. These traditional approaches not only consume substantial energy due to the thermal demands but also limit the throughput of manufacturing facilities, creating bottlenecks in the supply chain for key fragrance and pharmaceutical intermediates. Furthermore, the use of heterogeneous catalysts or corrosive mineral acids often complicates downstream processing, necessitating expensive purification steps to remove residual metals or acidic byproducts that could degrade product quality.
The Novel Approach
In stark contrast, the technology disclosed in CN111004276A presents a streamlined, one-pot synthetic route that fundamentally alters the economic and technical feasibility of producing these catalysts. The novel method involves the direct condensation of phosphorus oxychloride, phenols, and sulfonamides in a single reaction vessel, eliminating the need for multi-step isolation procedures that typically plague complex organophosphorus synthesis. 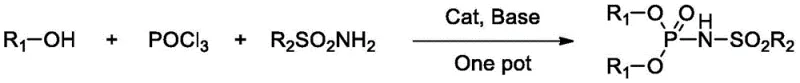 This approach not only simplifies the operational workflow but also significantly enhances the overall synthesis yield, with experimental data demonstrating efficiencies exceeding 85 percent under optimized conditions. By utilizing readily available starting materials and mild reaction conditions, this process drastically reduces the barrier to entry for manufacturing these high-performance catalysts. For a cost reduction in fine chemical manufacturing, this shift from multi-step, high-energy processes to a concise one-pot methodology represents a paradigm shift, offering a robust pathway to secure the supply of advanced catalytic materials needed for next-generation organic transformations.
This approach not only simplifies the operational workflow but also significantly enhances the overall synthesis yield, with experimental data demonstrating efficiencies exceeding 85 percent under optimized conditions. By utilizing readily available starting materials and mild reaction conditions, this process drastically reduces the barrier to entry for manufacturing these high-performance catalysts. For a cost reduction in fine chemical manufacturing, this shift from multi-step, high-energy processes to a concise one-pot methodology represents a paradigm shift, offering a robust pathway to secure the supply of advanced catalytic materials needed for next-generation organic transformations.
Mechanistic Insights into Phosphoramide-Catalyzed Rearrangement
The efficacy of these phosphoramide derivatives stems from their unique electronic properties, which function as strong Brønsted acids within organic media. Unlike traditional solid acids that may suffer from diffusion limitations or solubility issues, these derivatives are highly soluble in common organic solvents, ensuring homogeneous catalysis throughout the reaction mixture. The presence of the electron-withdrawing sulfonyl group adjacent to the nitrogen atom significantly increases the acidity of the P-N-H proton, allowing it to effectively activate the ether oxygen in 2-alkoxypropenes. This activation facilitates the cleavage of the carbon-oxygen bond and the subsequent rearrangement of the allylic system with remarkable precision. The mechanism avoids the formation of stable intermediate complexes that often lead to catalyst deactivation, thereby maintaining high turnover frequencies even at low catalyst loadings of 0.1 to 0.5 mol percent. This high level of activity ensures that the reaction proceeds rapidly, often reaching completion within just 1 to 3 hours, which is a fraction of the time required by legacy technologies.
From an impurity control perspective, the specificity of this catalytic system is paramount for producing high-purity OLED material precursors or fragrance ingredients. The strong acidity is balanced by the steric environment provided by the phenoxy substituents, which directs the reaction pathway towards the desired gamma-delta-unsaturated ketone with selectivity rates often surpassing 90 percent. This high selectivity minimizes the formation of isomeric byproducts or polymerization side reactions that are common in harsh acid environments. Consequently, the downstream purification burden is significantly reduced, leading to cleaner crude products that require less intensive distillation or chromatography. For R&D teams focused on the commercial scale-up of complex polymer additives or fine chemicals, this level of control over the impurity profile is essential for meeting stringent regulatory standards and ensuring consistent batch-to-batch quality in large-scale production runs.
How to Synthesize Diphenoxymethylsulfonylphosphamide Efficiently
The practical implementation of this technology relies on a straightforward protocol that balances reactivity with safety. The synthesis begins by establishing an inert atmosphere to prevent moisture interference, followed by the sequential addition of reagents to manage the exothermic nature of the phosphorylation steps. Detailed procedural nuances, such as the specific order of addition and temperature ramping, are critical for maximizing yield and minimizing the formation of hydrolysis byproducts. The following guide outlines the standardized approach derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale execution.
- Prepare the reaction vessel under nitrogen atmosphere with a polar aprotic solvent like dichloroethane and add the amine catalyst such as DMAP along with a base like triethylamine.
- Slowly add phosphorus oxychloride followed by the dropwise addition of the phenol component at room temperature, maintaining stirring for approximately two hours to ensure complete phosphorylation.
- Introduce the sulfonamide reagent to the mixture and heat under reflux conditions for several hours to finalize the formation of the phosphoramide derivative, followed by standard aqueous workup and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this phosphoramide technology offers tangible strategic benefits beyond mere technical performance. The reliance on commodity chemicals like phosphorus oxychloride and substituted phenols ensures a stable and resilient raw material supply base, mitigating the risks associated with sourcing exotic or proprietary reagents. The simplification of the synthesis process into a one-pot operation directly translates to reduced labor costs and lower capital expenditure on reactor infrastructure, as there is no need for complex filtration or separation units between steps. This operational efficiency allows manufacturers to respond more agilely to market demand fluctuations, reducing lead time for high-purity fine chemical intermediates and enhancing overall supply chain reliability. Furthermore, the ability to operate at lower temperatures and pressures compared to traditional methods contributes to a safer working environment and lower energy consumption, aligning with modern sustainability goals.
- Cost Reduction in Manufacturing: The elimination of multi-step purification and the use of inexpensive, bulk-available starting materials significantly lowers the cost of goods sold. By avoiding the need for expensive transition metal catalysts or complex ligand systems, the process removes the financial burden associated with metal removal and recovery protocols. The high yield of the one-pot synthesis ensures that raw material utilization is maximized, reducing waste generation and the associated disposal costs. Additionally, the shortened reaction times increase the asset utilization rate of existing manufacturing facilities, allowing for greater production volume without the need for additional capital investment in new reactors.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route means that production schedules are less susceptible to delays caused by difficult-to-source reagents or sensitive reaction conditions. Since the catalyst can be synthesized in-house using standard chemical engineering practices, companies can reduce their dependency on external suppliers for critical catalytic components. This vertical integration capability strengthens the supply chain against global disruptions and ensures a continuous flow of materials for downstream applications. The stability of the final phosphoramide product also simplifies logistics, as it can be stored and transported without special handling requirements, further streamlining the distribution network.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to tonnage-level commercial production without significant re-optimization. The use of common solvents like dichloroethane, which can be recovered and recycled, minimizes the environmental footprint of the manufacturing process. Moreover, the high selectivity of the catalyst reduces the generation of hazardous byproducts, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. This alignment with green chemistry principles not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phosphoramide technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for stakeholders evaluating the feasibility of adoption. Understanding these details is essential for making informed decisions about process integration and resource allocation.
Q: What are the primary advantages of this phosphoramide catalyst over traditional mineral acids?
A: Unlike traditional mineral acids which often require harsh conditions and long reaction times, these phosphoramide derivatives offer superior solubility in organic solvents and stronger acidity, enabling rearrangement reactions to complete in significantly shorter durations (1-3 hours) with higher selectivity.
Q: Can this synthesis method be scaled for commercial production of fragrance intermediates?
A: Yes, the one-pot synthesis utilizes readily available raw materials like phosphorus oxychloride and common phenols, operating under mild temperatures and atmospheric pressure, which simplifies equipment requirements and facilitates safe commercial scale-up.
Q: What specific substrates are compatible with this catalytic system?
A: The catalyst is highly effective for the rearrangement of various allyl alcohol compounds, including linalool and nerolidol, reacting with 2-alkoxypropenes to produce valuable gamma-delta-unsaturated ketones like methyl heptenone and geranylacetone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphoramide Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalysis in driving efficiency across the fine chemical and pharmaceutical sectors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the phosphoramide derivatives in CN111004276A can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of catalyst or intermediate meets the exacting standards required for high-value applications. Our commitment to technical excellence allows us to support clients in navigating the complexities of process optimization and regulatory compliance.
We invite you to collaborate with us to unlock the full commercial potential of this technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our solutions can enhance your operational efficiency and reduce your overall manufacturing costs. Let us be your partner in achieving sustainable growth and technological leadership in the global chemical market.
