Advanced Resin-Catalyzed Manufacturing of High-Purity 1,4-Dihydropyridine Derivatives for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes for critical cardiovascular intermediates, and Patent CN101508672B presents a transformative approach to manufacturing 1,4-dihydropyridine derivatives. This technology addresses the longstanding challenges associated with the esterification of 1,4-dihydropyridine-3,5-dicarboxylic acids, which serve as the core scaffolds for numerous calcium channel blockers. By leveraging specialized resin catalysts, the disclosed method achieves high conversion rates under remarkably mild thermal conditions, ranging from 0°C to 150°C, while eliminating the corrosive environments typical of traditional mineral acid or strong base catalysis. This innovation not only enhances the chemical integrity of the sensitive dihydropyridine ring but also streamlines the isolation process, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming for greener and more efficient production standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these vital intermediates has relied heavily on homogeneous catalysis using strong inorganic bases like sodium hydride or triethylamine, as well as mineral acids. These conventional pathways suffer from severe drawbacks, including the generation of large quantities of inorganic salt waste that complicates wastewater treatment and increases environmental compliance costs. Furthermore, the highly basic or acidic conditions often lead to equipment corrosion, necessitating expensive specialized reactor linings and increasing maintenance downtime. From a quality perspective, the harsh reaction environments can promote degradation of the dihydropyridine core, resulting in lower yields and complex impurity profiles that require rigorous and costly purification steps such as column chromatography to meet pharmacopeial standards.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes heterogeneous resin catalysts, such as Zeo-karb or anion exchange resins, to drive the esterification reaction with exceptional efficiency. This approach allows for the direct reaction of the dicarboxylic acid precursors with various alcohols or alkyl halides without the formation of troublesome salt byproducts. The heterogeneous nature of the catalyst enables simple physical separation via filtration, allowing the catalyst to be recovered, regenerated, and reused multiple times, which drastically simplifies the post-reaction workup. As illustrated in the reaction scheme below, this versatile platform accommodates a wide range of substituents while maintaining high selectivity and yield.
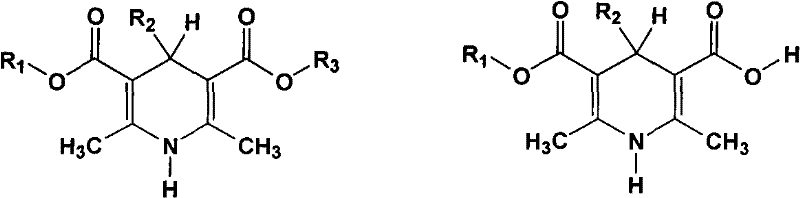
Mechanistic Insights into Resin-Catalyzed Esterification
The efficacy of this process lies in the unique surface chemistry of the ion-exchange resins, which provide active acidic or basic sites that activate the carboxylic acid group or the alcohol nucleophile without solubilizing into the reaction medium. For instance, when using cationic resins like D001 or metal-loaded variants such as AlCl3-D001, the Lewis acid sites coordinate with the carbonyl oxygen, increasing the electrophilicity of the carbonyl carbon and facilitating nucleophilic attack by the alcohol. This mechanism proceeds through a transition state that is stabilized by the resin matrix, lowering the activation energy required for ester bond formation while avoiding the high-energy intermediates that lead to ring oxidation or hydrolysis. The result is a clean reaction profile where the primary pathway dominates, ensuring that the valuable 1,4-dihydropyridine structure remains intact throughout the transformation.
Impurity control is another critical mechanistic advantage offered by this resin-based system. In traditional base-catalyzed methods, the presence of free alkoxides can lead to transesterification side reactions or Michael addition byproducts that are structurally similar to the target molecule and difficult to remove. The steric environment and specific pKa properties of the resin catalysts create a selective microenvironment that favors the desired esterification over these competing pathways. Experimental data from the patent embodiments confirms this, showing foreign matter content consistently below 1% and HPLC purity levels reaching up to 99.3%. This high level of chemical fidelity reduces the burden on downstream purification units and ensures a consistent quality profile essential for high-purity pharmaceutical intermediate manufacturing.
How to Synthesize 1,4-Dihydropyridine Derivatives Efficiently
To implement this advanced synthesis route, manufacturers must carefully select the appropriate resin catalyst based on the specific reactivity of the substrates involved, whether they are primary alcohols, secondary alcohols, or alkyl halides. The process generally involves suspending the resin in a suitable organic solvent such as acetonitrile, toluene, or DMF, followed by the addition of the dihydropyridine acid and the alkylating agent. Reaction temperatures are optimized between 50°C and reflux conditions depending on the boiling point of the solvent and the steric hindrance of the reactants, with reaction times typically ranging from 2 to 36 hours to ensure complete conversion. Detailed standardized operating procedures regarding catalyst loading ratios, solvent volumes, and crystallization protocols are essential for reproducibility.
- Mix the 1,4-dihydropyridine dicarboxylic acid substrate with the appropriate alcohol or alkyl halide reagent in a suitable organic solvent such as acetonitrile or toluene.
- Add the specific resin catalyst (e.g., D001 Zeo-karb or 717 anion exchange resin) at a weight ratio of 1/50 to 1/5 relative to the substrate.
- Heat the mixture to between 0°C and 150°C for 1 to 36 hours, then filter to recover the catalyst and crystallize the product from the filtrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resin-catalyzed technology offers profound strategic benefits that extend beyond simple yield improvements. The elimination of corrosive reagents means that production can be carried out in standard stainless steel reactors rather than requiring glass-lined or Hastelloy vessels, significantly lowering capital expenditure requirements for facility upgrades. Additionally, the ability to filter and reuse the catalyst multiple times reduces the recurring cost of raw materials, leading to substantial cost savings in API manufacturing over the long term. The simplified workup procedure, which avoids complex aqueous extractions and neutralization steps, shortens the overall batch cycle time, thereby enhancing production throughput and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The shift from homogeneous to heterogeneous catalysis removes the need for expensive neutralization agents and the disposal of large volumes of saline wastewater, directly lowering operational expenditures. By enabling catalyst regeneration, the process minimizes the consumption of catalytic materials, which is particularly advantageous when using specialized metal-loaded resins. This efficiency translates into a more competitive cost structure for the final active pharmaceutical ingredient, allowing partners to maintain healthy margins even in volatile raw material markets.
- Enhanced Supply Chain Reliability: The robustness of the resin catalysts ensures consistent batch-to-batch performance, reducing the risk of production failures or off-spec material that could disrupt supply continuity. Since the catalysts are stable and easily stored, there is no need for complex cold-chain logistics or immediate usage requirements associated with sensitive liquid catalysts. This stability supports a more resilient supply chain capable of meeting fluctuating demand schedules without compromising on the quality or availability of critical cardiovascular drug intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the patent examples ranging from gram-scale laboratory synthesis to multi-kilogram pilot runs, making it suitable for commercial scale-up of complex pharmaceutical intermediates. The reduction in hazardous waste generation and the absence of corrosive effluents simplify environmental permitting and compliance reporting, aligning with global sustainability goals. This eco-friendly profile enhances the corporate social responsibility standing of the manufacturing partner and mitigates regulatory risks associated with increasingly stringent environmental laws.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resin-catalyzed synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners. Understanding these nuances is crucial for evaluating the feasibility of integrating this technology into existing production lines.
Q: What are the primary advantages of using resin catalysts over traditional base catalysts for 1,4-dihydropyridine synthesis?
A: Resin catalysts eliminate the need for harsh mineral acids or strong bases like sodium hydride, thereby preventing equipment corrosion and reducing hazardous waste generation. This method allows for simple filtration to separate the catalyst, significantly simplifying downstream processing and improving overall product purity compared to conventional salt-forming methods.
Q: Can the resin catalyst be reused in this manufacturing process?
A: Yes, the patent data explicitly demonstrates that the resin catalysts, such as D001 or 717 types, can be recovered via filtration and regenerated for multiple cycles without significant loss of catalytic activity. This reusability contributes to substantial cost reductions and aligns with green chemistry principles by minimizing solid waste.
Q: How does this method impact the purity profile of the final API intermediate?
A: The mild reaction conditions and the heterogeneous nature of the resin catalyst minimize side reactions and decomposition often seen with aggressive homogeneous catalysts. Experimental data in the patent indicates HPLC purity levels consistently exceeding 98%, with impurity content effectively controlled below 1%, which is critical for regulatory compliance in pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Dihydropyridine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthetic routes in the modern pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and secure. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest international standards. Our infrastructure is designed to handle complex chemistries like the resin-catalyzed esterification described in CN101508672B, guaranteeing a steady supply of high-quality intermediates for your drug development pipelines.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this greener process can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can become a cornerstone of your supply chain strategy.
