Advanced Manufacturing of Pyrimidylcyclopentane Intermediates for AKT Inhibitors
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex small molecules, particularly for oncology targets like AKT protein kinase inhibitors. Patent CN110590606B discloses a groundbreaking methodology for the preparation of pyrimidylcyclopentane compounds, specifically serving as critical intermediates for drugs such as ipatasertib. This technology represents a significant leap forward in process chemistry, addressing long-standing challenges related to stereocontrol, purification efficiency, and environmental safety. By integrating transition metal catalysis with biocatalysis, the disclosed process offers a reliable pharmaceutical intermediate supplier pathway that ensures high optical purity without the burden of extensive downstream processing. For R&D directors and procurement managers alike, understanding the nuances of this patented approach is essential for securing a stable supply chain for next-generation cancer therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral amino acid derivatives and hydroxylated heterocycles required for AKT inhibitors has been plagued by inefficiencies and safety concerns. Traditional routes often relied on diastereoselective additions using chiral auxiliaries, which necessitated additional steps for attachment and removal, inevitably leading to significant material loss and reduced overall yields. Furthermore, prior art methods frequently employed coupling reagents like HBTU, which are known to cause severe occupational allergic contact dermatitis, posing serious industrial hygiene risks. The reliance on hazardous solvents such as dichloromethane, classified as a Hazardous Air Pollutant (HAP), further complicated regulatory compliance and waste disposal. Perhaps most critically, conventional approaches often failed to achieve sufficient stereoselectivity, forcing manufacturers to resort to preparative chiral chromatography, a technique that is prohibitively expensive and difficult to scale for commercial manufacturing due to high solvent consumption and low throughput.
The Novel Approach
In stark contrast, the novel methodology outlined in the patent introduces a convergent strategy that couples two highly optimized chiral fragments. The process begins with the asymmetric hydrogenation of an acrylic acid derivative using a specialized Ruthenium complex catalyst, which establishes the first stereocenter with exceptional enantiomeric excess. Simultaneously, a ketone precursor is subjected to enzymatic reduction using engineered ketoreductases, setting the second stereocenter with high diastereoselectivity under mild aqueous conditions. These two fragments are then joined via an amide coupling reaction using propylphosphonic anhydride (T3P). This integrated approach not only streamlines the synthetic sequence by reducing the total number of steps but also fundamentally alters the safety and environmental profile of the manufacturing process. 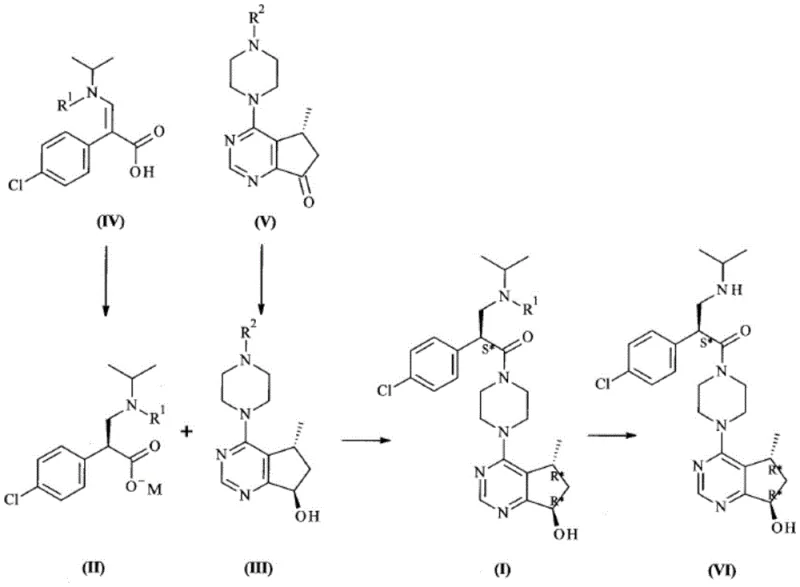
Mechanistic Insights into Ruthenium-Catalyzed Asymmetric Hydrogenation
The cornerstone of the first fragment's synthesis is the asymmetric hydrogenation of the dehydro-amino acid precursor. This transformation is mediated by a cationic or neutral Ruthenium complex containing chiral diphosphine ligands, such as BINAP or MeOBIPHEP. The mechanism involves the coordination of the olefinic substrate to the metal center, followed by the migratory insertion of hydride species generated from molecular hydrogen. The chiral environment provided by the bulky phosphine ligands dictates the facial selectivity of the hydrogen addition, ensuring the formation of the desired (S)-enantiomer. Crucially, the patent highlights that specific catalyst formulations allow for substrate-to-catalyst ratios (S/C) as high as 10,000, a dramatic improvement over standard systems that typically operate at S/C ratios of 200 to 250. This high turnover number significantly reduces the residual metal content in the final product, simplifying purification and lowering the cost of goods.
Following the hydrogenation, the process employs a unique salt formation step where the crude acid is treated with a metal alkoxide, such as sodium ethoxide, to precipitate the product directly as a sodium salt. This crystallization-induced diastereomeric enrichment further enhances the optical purity, often achieving >99.9% ee without the need for recrystallization or chromatography. The second fragment synthesis utilizes a completely different mechanistic paradigm: biocatalysis. Engineered ketoreductases (KREDs) facilitate the NADPH-dependent reduction of a cyclopentanone moiety. The enzyme's active site provides a rigid chiral pocket that enforces strict stereocontrol, yielding the hydroxylated product with >99% diastereomeric excess. The use of a cofactor regeneration system, typically involving isopropanol or glucose dehydrogenase, ensures that only catalytic amounts of the expensive cofactor are required, making the process economically viable for large-scale operations. 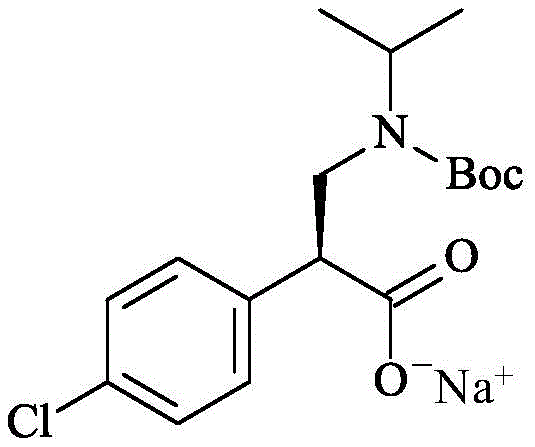
How to Synthesize Pyrimidylcyclopentane Compounds Efficiently
Implementing this advanced synthetic route requires precise control over reaction parameters to maximize yield and stereochemical integrity. The process is divided into three distinct operational phases: the chemical hydrogenation of the amino acid precursor, the enzymatic reduction of the heterocyclic ketone, and the final amide bond formation. Each phase demands specific attention to solvent selection, temperature control, and reagent stoichiometry to ensure reproducibility. The following guide outlines the standardized protocol derived from the patent examples, providing a clear roadmap for technical teams aiming to replicate this high-efficiency manufacturing process. Detailed standardized synthesis steps are provided in the guide below.
- Perform asymmetric hydrogenation of the acrylate precursor using a Ruthenium-BINAP catalyst complex to establish the first chiral center with high enantiomeric excess.
- Execute an enzymatic ketone reduction using engineered ketoreductases (KRED) and a cofactor regeneration system to set the second chiral center with high diastereoselectivity.
- Couple the resulting amino acid salt and hydroxylated heterocycle using propylphosphonic anhydride (T3P) in a green solvent system to form the final amide bond.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible strategic benefits beyond mere technical superiority. The elimination of chiral chromatography represents a massive reduction in solvent usage and processing time, directly impacting the cost reduction in pharmaceutical intermediate manufacturing. By avoiding the use of hazardous reagents like HBTU and solvents like dichloromethane, the process mitigates regulatory risks and lowers the costs associated with environmental compliance and waste treatment. The high efficiency of the catalysts and enzymes means that raw material consumption is optimized, leading to substantial cost savings in the long run. Furthermore, the robustness of the crystallization steps ensures consistent product quality, reducing the risk of batch failures and supply disruptions.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by utilizing high-turnover catalysts that minimize the requirement for expensive precious metals. The direct precipitation of the amino acid salt from the reaction mixture eliminates multiple extraction and concentration cycles, thereby reducing energy consumption and labor costs. Additionally, the switch to safer coupling reagents avoids the need for specialized handling equipment and protective measures required for sensitizing agents, further driving down operational expenditures.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials and robust catalytic systems ensures a stable supply of critical intermediates. The high stereoselectivity of the reactions guarantees consistent optical purity, reducing the variability that often plagues multi-step syntheses. This reliability is crucial for maintaining continuous production schedules for active pharmaceutical ingredients, preventing delays that could impact downstream drug formulation and market availability.
- Scalability and Environmental Compliance: Designed with green chemistry principles in mind, the process utilizes safer solvents and generates water-soluble byproducts that are easily removed via aqueous extraction. This simplifies the waste stream and facilitates compliance with increasingly stringent environmental regulations. The scalability of both the hydrogenation and enzymatic steps has been demonstrated at relevant scales, ensuring that the technology can seamlessly transition from pilot plant to commercial production without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this synthesis platform. These insights are derived directly from the experimental data and embodiments described in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs.
Q: How does this process avoid chiral chromatography?
A: The process utilizes highly stereoselective asymmetric hydrogenation with optimized Ruthenium catalysts and enzymatic reduction, achieving >99% ee and de directly, thus eliminating the need for costly and wasteful chiral separation steps.
Q: What are the safety advantages of the coupling reagent used?
A: Unlike traditional reagents like HBTU which pose allergenic risks, this method employs propylphosphonic anhydride (T3P), a non-toxic coupling agent that generates water-soluble byproducts, significantly improving industrial hygiene and waste management.
Q: Can this synthesis be scaled for commercial production?
A: Yes, the methodology is designed for scalability, utilizing high substrate-to-catalyst ratios (up to 10,000 S/C) and robust enzymatic systems that function effectively at high substrate concentrations, ensuring viable commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidylcyclopentane Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology therapies. Our expertise aligns perfectly with the advanced methodologies described in patent CN110590606B, allowing us to offer superior manufacturing capabilities for pyrimidylcyclopentane compounds. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral purity and residual solvent analysis, guaranteeing that every batch meets the highest international standards.
We invite you to collaborate with us to leverage these innovative synthetic routes for your project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume and timeline needs. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technical excellence can enhance the efficiency and reliability of your supply chain.
